Extrapulmonary Effects of Mechanical Ventilation
Learning Objectives
On completion of this chapter, the reader will be able to do the following:
2 Discuss the three factors that can influence cardiac output during positive-pressure ventilation.
4 Describe how positive-pressure ventilation increases intracranial pressure.
5 Summarize the effects of positive-pressure ventilation on renal and endocrine function.
6 Describe the effects of abnormal arterial blood gases on renal function.
7 Name five ways of assessing a patient’s nutritional status.
8 Describe techniques that can be used to reduce complications associated with mechanical ventilation.
Key Terms
• Cardiac tamponade
• Cardiac transmural pressure
• Oliguria
• Polyneuritis
Effects of Positive-Pressure Ventilation on the Heart and Thoracic Vessels
The physiological effects of mechanical ventilation are well documented. Laboratory and clinical studies have demonstrated that positive-pressure ventilation (PPV) can significantly alter cardiovascular, pulmonary, neurologic, renal, and gastrointestinal function. (See Chapter 17 for information on the pulmonary effects and complications of mechanical ventilation.) As such, every attempt should be made to minimize the adverse effects of PPV. Understanding the physiological effects and potential complications of PPV is therefore essential for clinicians involved with ventilator management.
Adverse Cardiovascular Effects of Positive-Pressure Ventilation
Positive-pressure ventilation can significantly change physiological pressures in the thorax. The extent of these changes depends on the amount of positive pressure applied to the airways and a patient’s cardiopulmonary status (Key Point 16-1).
The Thoracic Pump Mechanism During Normal Spontaneous Breathing and During Positive-Pressure Ventilation
It has been known for several decades that PPV can reduce cardiac output. This phenomenon can be understood in part by comparing intrapleural (i.e., intrathoracic) pressure changes that occur during normal spontaneous or negative pressure breathing with those occurring during PPV.
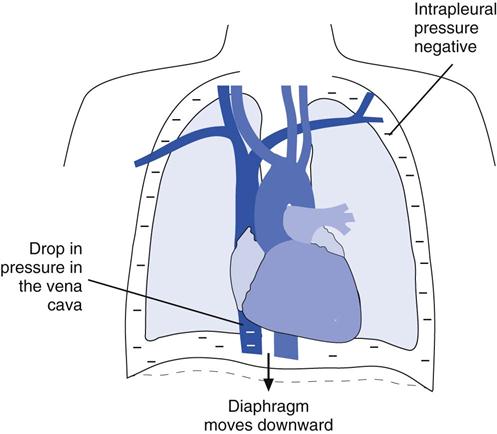
During spontaneous breathing, the fall in intrapleural pressure that draws air into the lungs during inspiration also draws blood into the major thoracic vessels and heart (Fig. 16-1). With this increased return of blood to the right side of the heart and the stretching and enlargement of the right-sided heart volume, the right ventricular preload increases, resulting in an increased right ventricular stroke volume (i.e., Frank-Starling mechanism). Conversely, during a spontaneous (passive) expiration, intrapleural pressure rises (i.e., becomes less negative), causing a reduction in venous return and right ventricular preload, which in turn leads to a decrease in right ventricular stroke volume. Note that these pressure changes affect left-sided heart volumes in a similar fashion.
The effects on intrathoracic pressures and venous return are quite different when positive pressure is applied to the airway (Fig. 16-2). During inspiration, increases in airway pressure are transmitted to the intrapleural space and to the great vessels and other structures in the thorax. As the airway pressure rises, the intrapleural pressure rises and intrathoracic blood vessels become compressed, causing the central venous pressure (CVP) to increase. This increase in CVP reduces the pressure gradient between systemic veins and the right side of the heart, which reduces venous return to the right side of the heart and thus right ventricular filling (preload). As a result, right ventricular stroke volume decreases.1
Notice that vascular pressures within the thorax generally increase in proportion to increases in mean airway pressure (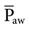 ) and intrapleural pressure (i.e., the higher the
) and intrapleural pressure (i.e., the higher the 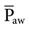 , the greater the effects). This phenomenon is particularly evident when one considers the effect of adding positive end-expiratory pressure (PEEP) during PPV. Because PEEP further increases
, the greater the effects). This phenomenon is particularly evident when one considers the effect of adding positive end-expiratory pressure (PEEP) during PPV. Because PEEP further increases 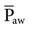 during PPV, it is reasonable to assume that reductions in venous return and cardiac output are greater during PPV with PEEP than with PPV alone. Furthermore, PEEP with assist-control decreases cardiac output more than when PEEP is used with synchronized intermittent mandatory ventilation (SIMV) or continuous positive airway pressure (CPAP) alone.
during PPV, it is reasonable to assume that reductions in venous return and cardiac output are greater during PPV with PEEP than with PPV alone. Furthermore, PEEP with assist-control decreases cardiac output more than when PEEP is used with synchronized intermittent mandatory ventilation (SIMV) or continuous positive airway pressure (CPAP) alone.
Increased Pulmonary Vascular Resistance and Altered Right and Left Ventricular Function
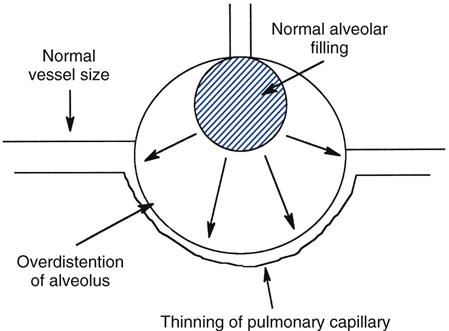
During inspiration with high tidal volumes (VT) or when high levels of PEEP are used, the pulmonary capillaries that interlace the alveoli are stretched and narrowed. As a result, resistance to blood flow through the pulmonary circulation increases (Fig. 16-3). This increases right ventricular (RV) afterload (i.e., pulmonary vascular resistance [PVR] and the resting volume of the RV). In normal healthy individuals, RV stroke volume is maintained in the face of increased PVR because the RV contractile function is not severely impaired. However, in patients with compromised RV function, the RV cannot overcome these increases in PVR, and overdistension of the RV occurs, resulting in a decrease in RV output.
Dilation of the RV can also force the interventricular septum to move to the left. This phenomenon usually occurs when high 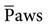 (>15 cm H2O) are used and the patient’s blood volume is depleted.3 When this occurs, the left ventricular end-diastolic volume (LVEDV) is encroached upon and left ventricle (LV) stroke volume may decrease because its ability to fill is limited. Septal shifting can significantly decrease cardiac output in patients with compromised LV function or in patients who are volume depleted.3 In this latter group of patients, intravascular volume expansion may help to restore output from the left side of the heart by returning LV preload to normal.
(>15 cm H2O) are used and the patient’s blood volume is depleted.3 When this occurs, the left ventricular end-diastolic volume (LVEDV) is encroached upon and left ventricle (LV) stroke volume may decrease because its ability to fill is limited. Septal shifting can significantly decrease cardiac output in patients with compromised LV function or in patients who are volume depleted.3 In this latter group of patients, intravascular volume expansion may help to restore output from the left side of the heart by returning LV preload to normal.
The LV output may also be decreased when high VTs are used during PPV because the heart is compressed between the expanding lungs (i.e., cardiac tamponade effect). The distensibility of the left side of the heart appears to be directly related to the transmission of positive pressures to the heart from the lung.2 This effect increases when long inspiratory times and high peak pressures are used.
Coronary Blood Flow with Positive-Pressure Ventilation
In addition to reduced venous return and alteration in ventricular function, lower cardiac output may be caused by myocardial dysfunction associated with reduced perfusion of the myocardium and the resultant myocardial ischemia. The flow of blood into the coronary vessels depends on the coronary perfusion pressure. The coronary artery perfusion pressure gradient for LV is the difference between mean aortic diastolic pressure and left ventricular end-diastolic pressure (LVEDP); the perfusion pressure gradient for the RV is the difference between mean aortic pressure and pulmonary artery systolic pressure.
Reductions in coronary vessel perfusion can result from any factor that decreases this perfusion pressure gradient. Thus, reductions in cardiac output or blood pressure, coronary vasospasms, or direct effect of compression of the coronary vessels caused by increases in intrathoracic pressure during PPV can decrease coronary perfusion and ultimately lead to myocardial ischemia.
Factors Influencing Cardiovascular Effects of Positive-Pressure Ventilation
The level of reduction in cardiac output that occurs with PPV depends on several factors, including lung and chest wall compliance, airway resistance (Raw), and the duration and magnitude of the positive pressure.
Compensation in Individuals with Normal Cardiovascular Function
Because of compensatory mechanisms, systemic hypotension rarely occurs in individuals with normal cardiovascular function receiving PPV. Decreases in stroke volume normally result in an increase in sympathetic tone, which leads to tachycardia and an increase in systemic vascular resistance and peripheral venous pressure from arterial and venous constriction, respectively. Additionally, some peripheral shunting of blood away from the kidneys and lower extremities occurs. The net effect is maintenance of blood pressure even with a decrease in cardiac output (Key Point 16-2).4
It is important to understand that the effectiveness of these compensatory mechanisms in maintaining arterial blood pressure depends on the integrity of the individual’s neuroreflexes. Vascular reflexes can be blocked or impaired in the presence of sympathetic blockade, spinal anesthesia, moderate levels of general anesthesia, spinal cord transection, or severe polyneuritis. In a patient in whom PPV is being initiated or the ventilatory mode is being changed, it is prudent to measure the blood pressure early to ensure that normal vascular reflexes are intact. The presence of normal vascular reflexes increases the probability that the patient will not experience a significant drop in cardiac output and blood pressure if PPV is initiated. For example, it is unusual to see a reduction in cardiac output in normovolemic patients when low levels of PEEP are used (i.e., 5-10 cm H2O of PEEP). However, decreases in cardiac output can occur in this group of patients if higher levels of PEEP are used (>15 cm H2O)4 (Case Study 16-1).
Effects of Lung and Chest Wall Compliance and Airway Resistance
Patients with very stiff lungs, such as those with acute respiratory distress syndrome (ARDS) or pulmonary fibrosis, are less likely to experience hemodynamic changes with high pressures because less of the alveolar pressure (Palv) is transmitted to the intrapleural space. On the other hand, patients with compliant lungs and stiff (noncompliant) chest walls are more likely to have higher intrapleural pressures with PPV and experience more pronounced cardiovascular effects.
In patients with increased Raw, although peak pressures may be very high, much of the pressure is lost to the poorly conductive airways. As a consequence, high peak airway pressures may not be transmitted to the intrapleural space and the alveoli.
Duration and Magnitude of Positive Pressures
One way to reduce the deleterious effects of PPV is to control the amount of pressure exerted in the thorax. Maintaining the lowest possible 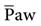 helps to minimize the reductions in cardiac output that can occur during mechanical ventilation. It is therefore important to understand how peak inspiratory pressure (PIP), inspiratory flow, inspiratory-to-expiratory (I : E) ratios, inflation hold, and PEEP affect
helps to minimize the reductions in cardiac output that can occur during mechanical ventilation. It is therefore important to understand how peak inspiratory pressure (PIP), inspiratory flow, inspiratory-to-expiratory (I : E) ratios, inflation hold, and PEEP affect 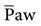 and, ultimately, cardiac output.
and, ultimately, cardiac output.
Beneficial Effects of Positive-Pressure Ventilation on Heart Function in Patients with Left Ventricular Dysfunction
Although the discussion so far has focused on the adverse effects of PPV, it is important to recognize that positive pressure can also be beneficial for patients with LV dysfunction and elevated filling pressures. For example, PEEP may improve cardiac function by raising the PaO2 and improving myocardial oxygenation and performance if the left LV dysfunction is due to hypoxemia. Reductions in venous return decrease the preload to the heart and thus improve length-tension relationships and improve the stroke volume in patients with ventricular overload. Additionally, by raising the intrathoracic pressure, PPV decreases the transmural LV systolic pressure and thus the afterload to the left heart (Critical Care Concept 16-1). Box 16-1 lists some potential effects of PEEP on heart function.5,6
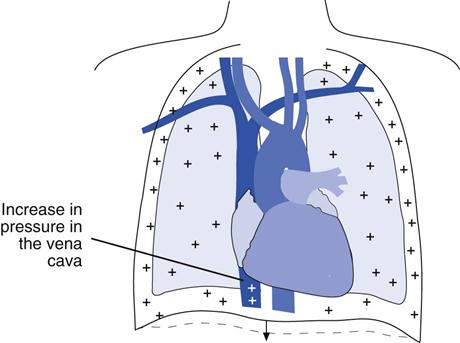
 ) and intrathoracic pressure lead to reduced venous return that can reduce preload to a failing heart and improve function.
) and intrathoracic pressure lead to reduced venous return that can reduce preload to a failing heart and improve function.


