Fig. 7.1
Relation between dyssynchrony and cardiac output
Typically a widened QRS on the EKG has been considered a surrogate for LV dyssynchrony. Hawkins and their group noted increasing prevalence of dyssynchrony with prolongation of QRS duration [22]. Data from the Vesnarinone (VEST) study has revealed that QRS duration was directly correlated to mortality as noted in Fig. 7.2. Prolongation of QRS (>120 ms) is widely accepted as occurring in about 30 % of patients with HF [23, 24]. It is a significant predictor of LV systolic dysfunction in HF with an inverse correlation between QRS length and left ventricular ejection fraction (LVEF) [24–26]. Xiao et al. showed that early mortality was observed in those with larger increase in QRS duration and the time to death from QRS reaching 160 ms was significantly shorter in those without an implanted pacemaker in comparison to those who had one [27]. Multiple studies have confirmed the higher incidence of sudden cardiac death in HF patients with prolonged QRS complex. In an effort to determine the optimal QRS duration that stratifies HF patients into higher and low risk for increased mortality or need for transplantation, Kalra and his group determined that patients with a QRS duration ≥120 ms were associated with a threefold risk and a significantly low 5-year survival rate [25]. Most large-scale trials have considered QRS length >120 ms as part of their inclusion criteria, thus offering the benefit of application in clinical scenarios.
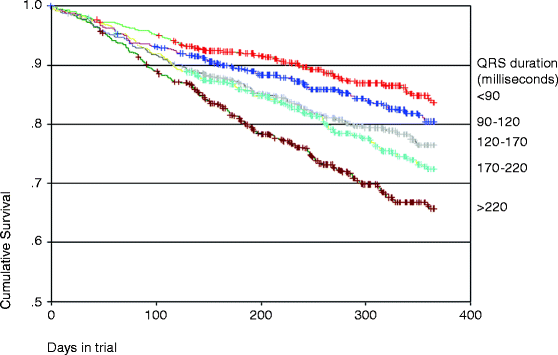

Fig. 7.2
Patient survival stratified by QRS duration (Adapted from Gottipaty V, Krelis S, Lu F, et al. JACC 1999; 33 (2S1):145A)
Rationale for CRT
Given the mounting evidence of dyssynchrony contributing to LV dysfunction, Cazeau and colleagues used CRT to improve the functional status of a patient who presented with HF with conduction abnormalities [28]. This led to further attempts to reverse dyssynchrony with the aid of pacing leads and spurred a new era of therapeutic application. CRT typically involves placement of pacing leads in the right atrium (RA), right ventricle (RV), and the lateral branch of the coronary sinus (Fig. 7.3). This placement in the branch of the coronary sinus is an attempt to pace the lateral wall of the LV, and hence it is often called the LV lead. With this approach, simultaneous and synchronized pacing of the ventricles can be performed to reduce dyssynchrony so as to simulate a physiological depolarization as seen in a healthy normal myocardium. An alternate approach is to pace the LV lateral wall with appropriate AV delay with the intent to merge with the wave originating through intrinsic activation of the RV, thus resulting in a coordinated contractile movement.
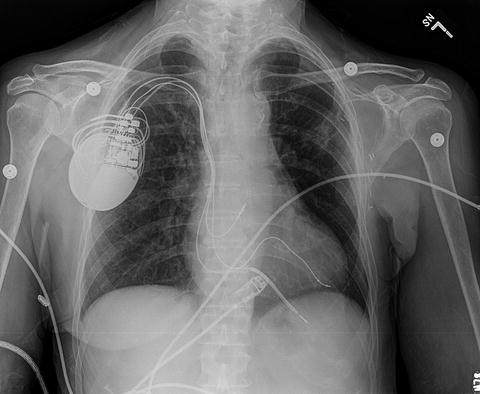

Fig. 7.3
Chest X-ray revealing typical lead placement
The ability to program the device in a manner such that AV interval can be adjusted so as to pace the ventricles without significant delay has been shown to optimize left ventricular filling (therefore preload) and reduction in presystolic mitral regurgitation [29]. The principle of biventricular pacing is to reverse the intraventricular dyssynchrony by timing the RV and LV pacing so as to encourage simultaneous contractility of the septal and lateral wall segments. The added ability to offset the timing of RV and LV pacing seen in newer devices has been proven to be more beneficial than simultaneous pacing of the ventricular leads [30]. Lateral wall is the site preferred for the LV lead placement since coronary sinus provides easy access to the free wall. Compared to other sites, lateral wall provides best response in terms of percentage change in pulse pressure and LV dP/dt and the maximal area where this can be effectively achieved [31, 32].
The acute hemodynamic effects are noted shortly after CRT initiation. Improvements in systolic blood pressure, peak dP/dt, EF, and finally cardiac output with associated decline in pulmonary capillary wedge pressures have been established by invasive hemodynamic monitoring [33]. This is achieved with coordinated contractile function resulting in improvement of ejection fraction as evidenced by improvement in stroke volume and reduction in LV end-systolic volume. The near elimination of functional mitral regurgitation by normalization of mitral valve timing has been known to contribute to reversing the geometrical changes due to disease progression [34, 35]. The clinical effects of these changes were clearly shown in large-scale randomized clinical trials which measured surrogate markers such as NYHA class, QoL, and 6-min walk test [11–13, 16, 35–37].
The long-term benefits of CRT were evident by the progressive reduction in LV cavity volume and LV mass since the initiation of pacing as revealed by the long-term follow-up of several randomized trials (MUSTIC, PATH-CHF, CONTAK-CD, MIRACLE, CARE-HF, REVERSE). The change in hemodynamics with reduction of mitral regurgitation enabled an alteration of LV cavity shape from a globular appearance to more physiological ellipsoid appearance [38]. Importantly, Yu and his group showed the reversal of these changes with discontinuation of CRT thereby highlighting the fact that these changes were independent from that occurs as a result of concomitant use of drugs (β-blockers, angiotensin converting enzyme [ACE] inhibitors, angiotensin receptor blockers [ARB]) [39]. Notably those with depressed EF as a result of nonischemic etiology showed sustained evidence of reversal of ventricular remodeling compared to those with ischemic cardiomyopathy [40]. This is perhaps attributed to the loss of viable myocardium as a result of repeated ischemia. Improvement in these cardiac measures contributed to the reduction in mortality besides the improvement in symptoms that were observed in the COMPANION and CARE-HF trials.
Evidence from the Trials
Since the observation of augmented cardiac performance with CRT, various trials have been undertaken to corroborate those findings. The early observational studies were limited in their sample size where the patient served both as the case as well as the control. The data from the randomized controlled trials have convincingly substantiated the role of CRT in HF management, given that nearly 5,000 patients have been evaluated to date.
A summary of the major trials is noted in Table 7.1, and how they guided standard of care is reviewed below.
Table 7.1
Summary of various randomized controlled trials evaluating the efficacy of cardiac resynchronization therapy
Trials (year of completion) | Design | Enrollment criteria | End points | Results (Parameters in bold attained statistical significance) | |||||
|---|---|---|---|---|---|---|---|---|---|
zNYHA class | EF | QRS (ms) | Rhythm | ICD | Primary | Secondary | |||
PATH-CHF (1998) | Crossover | III, IV | <30a | ≥120 | SR | No | 6 MWD and pVO2 | NYHA class, QoL | Improvement in 6 MWD, pVO 2 , QoL, and NYHA class |
MUSTIC-SR (1999) | Crossover | III | <35 % | >150 | SR | No | 6 MWD | QoL, pVO2, mortality, hospital admissions due to HF | Improvement in 6 MWD, QoL, pVO 2, and reduction in hospitalization |
MUSTIC-AF (1999) | Crossover | III | <35 % | ≥120, Paced QRS ≥ 200 | AFib | No | 6 MWD | QoL, pVO2, mortality, hospital admissions due to HF | Improvement in 6 MWD, QoL, pVO 2, and reduction in hospitalization |
MIRACLE (2000) | Parallel | III, IV | ≤35 % | ≥130 | SR | No | 6 MWD, QoL, NYHA class | EF, pVO2, LV volumes, QRS duration, MR severity, and composite clinical response | Improvement in all end points with statistical significance |
PATH-CHF II (2001) | Crossover | II, III, IV | ≤30 % | ≥120 | SR | No | 6 MWD and pVO2 | NYHA class, QoL | Improvement in QoL, NYHA class, and pVO 2 . Effect more in those with QRS > 150 ms |
CONTAK-CD (2000) | Parallelb | III, IVc | ≤35 % | ≥120 | SR | Yes | Progression of HF, 6 MWD, QoL, and pVO2 | Composite response (HF hospitalization, mortality, VT therapy with ICD), 6 MWD, QoL, and pVO2 | Improvement in QoL, pVO 2, and NYHA class with relative reduction in HF progression |
MIRACLE-ICD (2001) | Parallel | III, IV | ≤35 % | ≥130 | SR | Yes | 6 MWD, QoL, NYHA class | EF, pVO2, LV volumes, neurohormones, and clinical composite response | Improvement in QoL, NYHA class, and pVO 2 and a trend towards better LV volumes and composite score in CRT with no change in 6 MWD |
COMPANION (2002) | Parallel | III, IV | ≤35 % | ≥120 | SR | Yes | Time to death from or hospitalization to any cause | Mortality rate | Improvement in both end points with statistical significance noted in both CRT-P and CRT-D arms |
MIRACLE-ICD II (2002) | Parallel | II | ≤35 % | ≥130 | SR | Yes | pVO2 | NYHA class, EF, 6 MWD, QoL, LV volumes, neurohormones, and clinical composite response | Improvement of significance noted in LV volumes, EF, NYHA class, and clinical composite response but no difference in 6 MWD, QoL, and pVO2 |
CARE-HF (2003) | Parallel | III, IV | ≤35 % | ≥120 | SR | No | Time to death or hospitalization for cardiovascular events | All cause mortality, composite of mortality and HF hospitalization, NYHA class, QoL, LV volumes, and neurohormones | Improvement in all end points with improvement of symptoms and mortality benefit |
REVERSE (2007) | Parallel | I, II | ≤40 % | ≥120 | SR | Yes | HF clinical composite response | LV volume end-systolic volume index | Improvement noted in parameters consistent with reversal of remodeling and time to HF hospitalization in those with NYHA classes I and II |
Pacing Therapies in Congestive Heart Failure (PATH-CHF) Trial: this European study was the first randomized trial which was single blinded with crossover design where moderate to severe HF patients were allocated to receive RV, LV, or biventricular pacing or no pacing. Acute improvement in hemodynamic parameters was noted in the LV and biventricular categories with reduction in LV end-diastolic volume and LV end-systolic volume observed during chronic pacing in the same group. Besides, PATH-CHF also enabled the identification of mid-lateral epicardial lead as the optimal site for LV lead placement. The study was limited by its small sample size and short follow-up [11].
PATH-CHF II trial evaluated the significance of single-site LV pacing and degree of intraventricular conduction delay to clinical benefits. The patients were divided into two categories based on the QRS duration (short: QRS 120–150 ms, long: QRS > 150 ms). The assumption that CRT response may be linked to degree of QRS duration was driven by the observation that the group with prolonged QRS morphology showed improvement in exercise tolerance and QoL [12].
Multisite Stimulation In Cardiomyopathy In Sinus Rhythm (MUSTIC-SR) trial was similar in design to PATH-CHF II except that biventricular pacing was the intervention planned. There was significant improvement in 6-min walking distance (6 MWD) and pVO2 which were the primary end points. Significant improvement was seen in QoL measured as secondary end point in the treatment arm. Besides, there was alteration in the structural geometry due to reduction of LV cavity size. The degree of remodeling was more pronounced in HF patients whose etiology was related to nonischemic causes compared to ischemic ones [13].
Multisite Stimulation In Cardiomyopathy In Atrial Fibrillation (MUSTIC-AF) trial was an attempt to extrapolate similar results in patients with atrial fibrillation whose paced QRS > 200 ms. Although the effect was less pronounced than in the sinus rhythm group, it was significant enough to conclude that biventricular pacing was a reasonable option in HF patients with concomitant atrial fibrillation who become pacer dependent due to bradycardia either as a result of ablation or intrinsic conduction disease [14]. The intervention arm in both MUSTIC study groups preferred biventricular pacing and showed reduced hospitalizations in the 12-month follow-up period.
Multicenter InSync Randomized Clinical Evaluation (MIRACLE) trial was the first double-blind randomized control trial to validate the efficacy of CRT. Hence sufficient targets such as 6 MWD, NYHA class, and QoL were included as the primary end points. The patients were divided into two arms and assigned to 6 months of optimal medical therapy with biventricular pacing or medical therapy alone. The CRT group had significant improvement in all primary end points compared to the control. A noteworthy observation was that in those who underwent CRT, 67 % demonstrated improvement in clinical composite end point of NYHA class compared to 39 % in the control [35]. This study paved the way for the US Food and Drug Administration (FDA) to approve the use of InSync® device (Medtronic, Minneapolis, MN) in 2001.
Cardiac Resynchronization Therapy for the Treatment of Heart Failure in Patients With Intraventricular Conduction Delay and Malignant Ventricular Tachyarrhythmias: VENTAK CHF/CONTAK CD Biventricular Pacing Trial: the objective of this study was to assess the safety and effectiveness of CRT in combination with an ICD in patients with HF class II–IV symptoms. The study was unique since HF patients with history of ventricular tachyarrhythmia who were candidates for ICD placement were considered in the inclusion criteria. The primary end point was progression of HF, defined as all cause mortality along with HF hospitalizations and ventricular tachycardia or ventricular fibrillation requiring device intervention. Despite a 15 % reduction in the primary end point, no statistical significance was attained between the two groups. However improvement in secondary end points such as 6 MWT, pVO2, and EF was noted in NYHA classes III and IV but not so much in class II. Echocardiography demonstration of remodeling effect was evident in the reduction of ventricular dimensions across all NYHA classes studied [41].
MIRACLE-ICD trial was configured to compare the effect of CRT with ICD against ICD alone in terms of QoL and functional capacity. Despite an improvement in NYHA class and QoL, the primary end points and pVO2, and the secondary end point, no significant difference was noted in terms of 6 MWD. The study established that in those with an indication for ICD, addition of CRT offers symptomatic relief [36].
MIRACLE-ICD II trial was a randomized substudy carried out in HF patients who fit the criteria for MIRACLE-ICD study with the exception of NYHA class being II. Though there was no significant improvement in pVO2, 6 MWT, and QoL, improvement was noted in LVEF and cardiac dimensions, which are surrogate markers for ventricular remodeling [42].
COMPANION trial is the largest randomized CRT trial to date designed to compare optimal medical therapy against CRT alone and CRT with defibrillator (CRT-D) in advanced heart failure patients who otherwise had no indication for pacemaker or ICD at baseline. The primary end point was all cause mortality or hospitalization, while secondary end points were both all cause mortality and exercise performance. This was the first trial that was powered to evaluate the mortality benefit in a prospective manner. The primary composite end point was reduced by 19 % in the CRT/CRT-D arms with a reduction of total mortality by 15 % and 40 % in the CRT and CRT-D arm, respectively. The benefit offered in terms of reduction in sudden death noted in the CRT-D arm provided compelling evidence for the FDA to approve the CRT-D device for patients who are candidates for CRT implantation [16].
Cardiac Resynchronization in Heart Failure (CARE-HF) trial was a European study designed with the intent to observe morbidity and mortality benefit of CRT in HF patients whose QRS duration was more than 120 ms. The sample was again subdivided based on the QRS width into those with QRS duration more than 150 ms and the other group with QRS duration between 120 and 150 ms. The study was distinctive in its design by requiring to demonstrate the evidence of LV dyssynchrony by echocardiogram (ECHO) in the latter group. The primary end point was all cause mortality or any cardiovascular event requiring hospitalization and secondary end point being all cause mortality. CRT was shown to reduce risk of death, intraventricular mechanical delay, mitral regurgitation, and LV end-systolic volume and improve EF, symptoms, and QoL [17]. An extension of the CARE-HF study beyond the initial 29.4 months showed the extension of mortality benefits with long-term follow-up [43].
A recent meta-analysis of pooled data (Fig. 7.5) from 14 randomized controlled trials evaluating the benefit of CRT over 6 months revealed an improvement by at least one NYHA class from baseline in 59 % of patients who underwent CRT implantation in comparison to 37 % among controls with no resynchronization therapy [44]. An augmentation in other parameters such as LVEF, 6 MWD, and QoL was also observed in this study by McAlister and his group. A higher percentage ranging from 63 % to 82 % was noted when 97 observational studies were pooled for analysis by the same group. Only 19 % required hospitalization in the device group compared to 27 % in those without CRT. A notable decrease in all cause mortality was evident in the CRT group (13.2 % in CRT as against 15.5 % with no CRT), and this was expected to increase with duration after CRT. This was in consensus with results of CARE-HF data pertaining to determination of the number needed to prevent one death, which decreased from 13 patients at 2 years to nine patients in 3 years [44]. Figures below demonstrate the response to clinical end points from various randomized controlled trials (Figs. 7.5, 7.6, 7.7, 7.8, 7.9, 7.10 and 7.11).
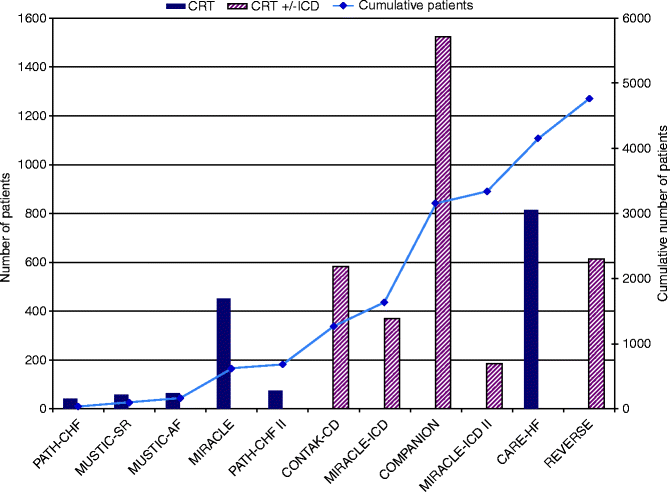
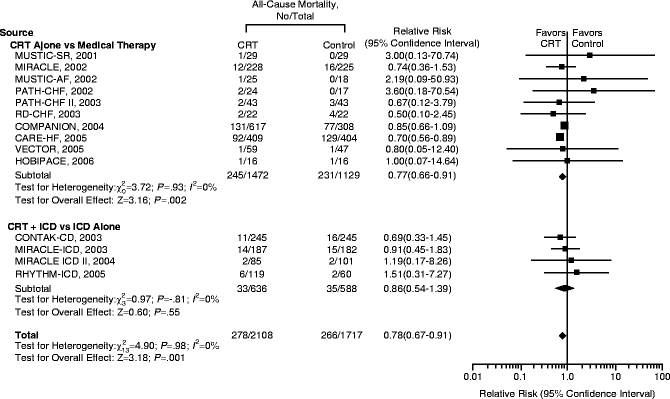
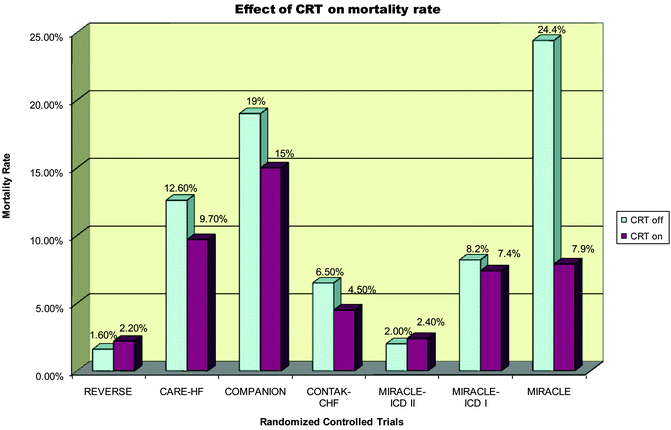
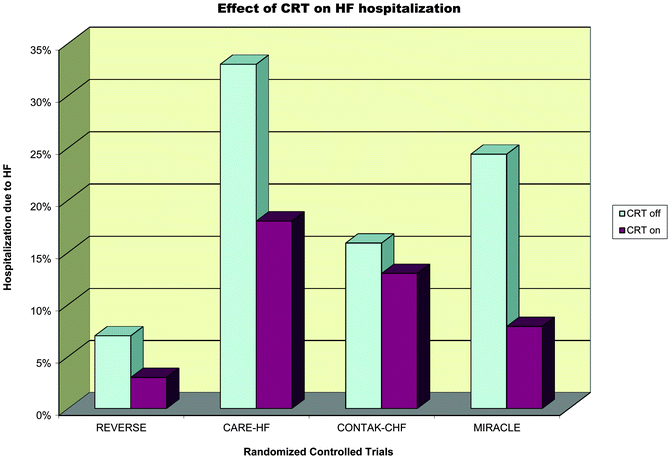
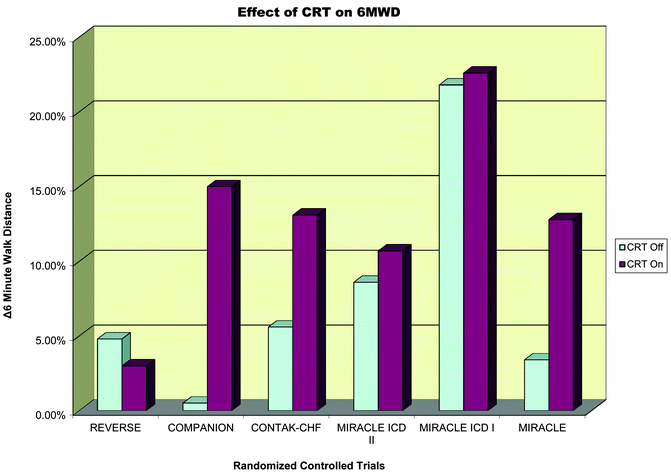
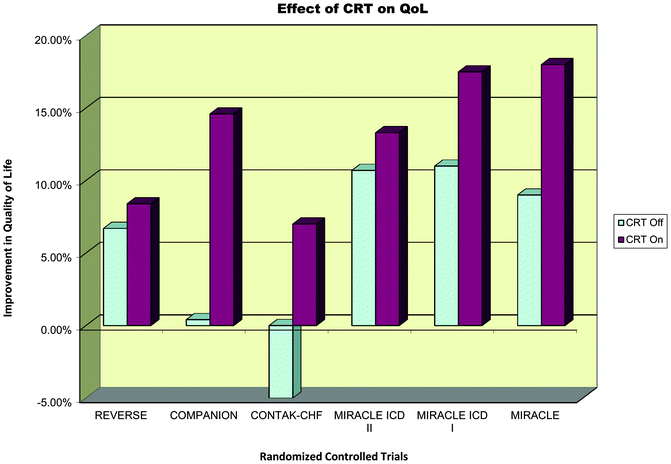
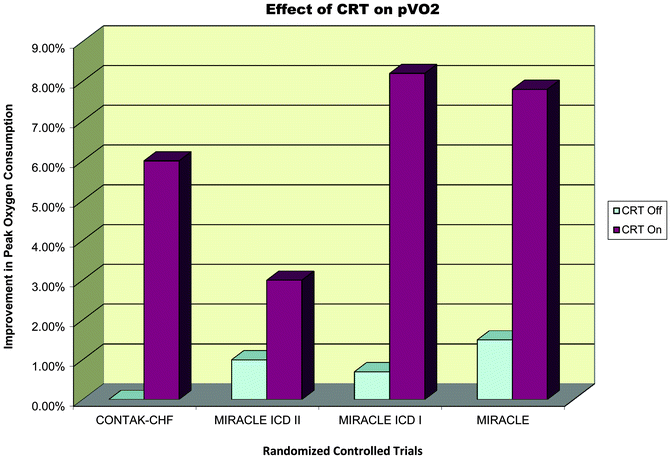
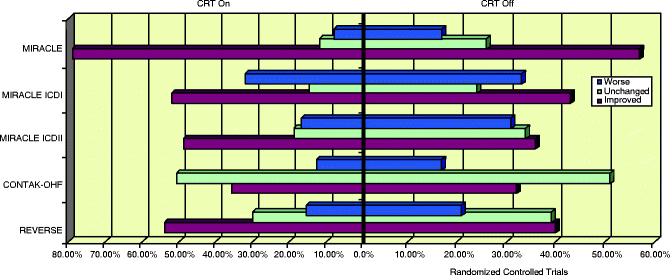

Fig. 7.4
Data spread of various randomized controlled trials on CRT

Fig. 7.5
Effect of CRT on all cause mortality (Reprinted with permission from McAlister FA, Ezekowitz J, Hooton N, Vandermeer B, Spooner C, Dryden DM, et al. Cardiac resynchronization therapy for patients with left ventricular systolic dysfunction: a systemic review. JAMA. 2007;297(22):2502–2514). Copyright © (2007) American Medical Association. All rights reserved

Fig. 7.6
Effect of CRT on mortality rate

Fig. 7.7
Effect of CRT on HF hospitalization

Fig. 7.8
Effect of CRT on 6-min walk distance

Fig 7.9
Effect of CRT on quality of life

Fig. 7.10
Effect of CRT on pVO2

Fig. 7.11
Effect of CRT on HF clinical composite response
Given the insurmountable evidence provided by these trials, major cardiology societies (American Heart Association/American College of Cardiology (AHA/ACC), European Society of Cardiology) have stated CRT as class I indications in heart failure patients who are optimized on medical management and fulfill all of the following:
A documented EF ≤ 35 %
In sinus rhythm
A functional class of NYHA class III or ambulatory NYHA class IV
Evidence of cardiac dyssynchrony as evidenced by a QRS ≥ 120 ms [45]
Despite these guidelines, a recent study by Hernandez et al. revealed that one in ten patients with CRT implantation has received the devices outside its current recommendations [46]. The researchers hypothesized that a small number of this could be possibly explained by the prophylactic implantation of biventricular pacemaker instead of RV pacing with the intent to prevent systolic dysfunction among those with EF > 35 % as supported by the post AV nodal ablation evaluation (PAVE) trial [47]. Since the update to the AHA/ACC guidelines, results from the “resynchronization reverses remodeling in systolic left ventricular dysfunction” (REVERSE) trial have shed light into the benefit of CRT to patients with HF who are either asymptomatic or have mild symptoms. This randomized control trial evaluated the benefit of CRT (with or without a defibrillator) in patients with systolic HF on optimum drug therapy with an EF ≤ 40 % and QRS duration ≥120 ms who belong to NYHA class I or II. The primary end point in this study was clinical composite response for HF stated as worsened, unchanged, or improved. Due to the inclusion of asymptomatic patients, a proportional analysis was performed as a marker for efficacy. Twenty-one percent in the group with no CRT worsened in comparison to 16 % in the CRT group, but this did not achieve statistical significance. However the time to hospitalization was delayed in the CRT group with added evidence of reversal of LV remodeling noted increasingly in this arm as evidenced by marked improvement in the LV end-systolic volume index [48]. Though this study was not devised to analyze mortality, one can speculate that the evidence of reversal of remodeling may contribute to reduction in mortality. Ongoing trials such as Multicenter Automatic Defibrillator Implantation Trial with CRT (MADIT-CRT) and Resynchronization/Defibrillation for Ambulatory Heart Failure Trial (RAFT) are designed to address mortality benefit in mild HF patients.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


