Chapter 5 Autonomic Nervous System and Cardiac Arrhythmias
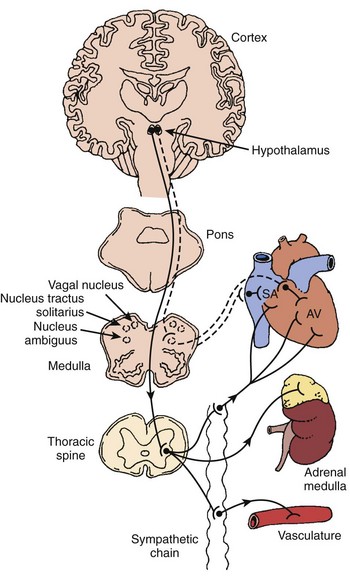
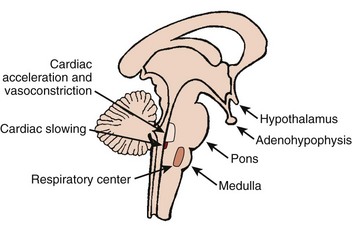
The autonomic nervous system (ANS) comprises the portion of the central nervous system that provides moment-to-moment regulation of the function of the cardiovascular system as well as that of all other organ systems. The ANS continuously monitors afferent neural signals from vascular beds and organ systems and coordinates efferent neural traffic to modify the responses of heart and blood vessels to ever-changing physiological and metabolic requirements. In this context, the sympathetic and parasympathetic components of the ANS are the dominant players (Figures 5-1 and 5-2).1 However, ANS cardiovascular control also incorporates actions of cardiac and extracardiac neurohumoral agents, intracardiac reflex arcs, and the contributions of certain less well-understood agents such as vasoactive intestinal peptide (VIP), neuropeptide Y, transmitters released by the so-called purinergic nerve endings, serotonin, inflammatory cytokines, vasopressin, and nitric oxide.2 Further, with respect to cardiovascular control, the ANS collaborates with the hypothalamic-pituitary-adrenal (HPA) axis. For its part, the HPA-axis, governed from the hypothalamus, participates by prompting the release of glucocorticoids, mainly cortisol and, to a lesser extent, mineralocorticoids. The HPA theater of operation therefore includes inflammatory, immune, metabolic, and pressor effects.3 Both systems (ANS and HPA) are involved in stress responses.2,3
It is not unexpected that any disturbance of ANS function, given its wide-ranging impact, may lead to clinically important consequences. In terms of cardiac electrophysiology and arrhythmias, common clinical conditions in which ANS effects are evident include acute myocardial ischemia, heart failure, and neurally mediated reflex syncope (particularly the vasovagal faint). Furthermore, it is now widely acknowledged that the nervous system has the capacity to injure the heart acutely (e.g., stress-induced cardiomyopathy); serious acute cerebral disorders such as subarachnoid hemorrhage, intracerebral bleeds, infections, and seizures may induce electrocardiographic changes, myocardial damage, arrhythmias, and even sudden death.3–7 Perhaps the most publicized direct cardiac effects of presumed autonomic “storms” are the immediate, apparently stress-triggered, increases in the number of cardiovascular events; these include acute myocardial infarctions, sudden cardiac deaths, and presumed stress-induced cardiomyopathy (Table 5-1).3–5
Table 5-1 Epidemiologic Associations: Stress and Increased Cardiovascular Event Rates
MI, Myocardial infarction; SCD, sudden cardiac death; ICD, implantable cardioverter-defibrillator.
References
a Stalnikowicz R, Tsafrir A: Acute psychosocial stress and cardiovascular events, Am J Emerg Med 20:488–491, 2002.
b Brown DL: Disparate effects of the 1989 Loma Prieta and 1994 Northridge earthquakes on hospital admissions for acute myocardial infarction: Importance of superimposed triggers, Am Heart J 137:830–836, 1999.
c Watanabe H, Kodama M, Okura Y, et al: Impact of earthquakes on Takotsubo cardiomyopathy. JAMA 294:305–306, 2005.
d Zhang XQ, Chen M, Yang Q, et al: Effect of the Wenchuan earthquake in China on hemodynamically unstable ventricular tachyarrhythmia in hospitalized patients, Am J Cardiol 103(7):994–997, 2009.
e Wilbert-Lampen U, Leistner D, Greven S, et al: Cardiovascular events during World Cup soccer. N Engl J Med 358:475–483, 2008.
f Katz E, Metzker J-T, Marazzi A, Kappenberger L: Increased sudden cardiac deaths in Switzerland during the 2002 FIFA World Cup, Int J Cardiol 107:132–133, 2006.
g Brotman DJ, Golden SH, Wittstein IS: The cardiovascular toll of stress, Lancet 376: 1089–1100, 2007.
h Meisel SR, Kutz I, Dayan KI, et al: Effect of Iraqi missile war on incidence of acute myocardial infarction and sudden death in Israeli civilians, Lancet 338:660–661, 1991.
i Tofler GH, Muller JE: Triggering of acute cardiovascular disease and potential preventive strategies, Circulation 114:1863–1872, 2006.
Anatomic Nervous System and Cardiac Conduction System Physiology
Sinus Node, Atrioventricular Node, and His-Purkinje System
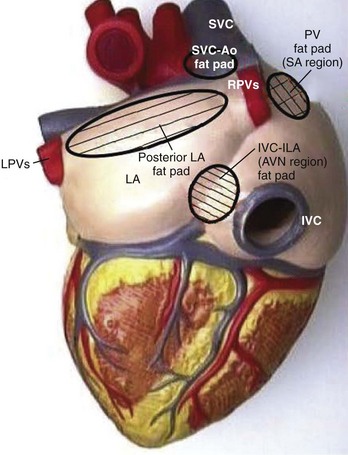
The sinus node (SN) and the atrioventricular (AV) node appear to be represented by separate cells within the nucleus ambiguus. However, it is uncertain whether the nodes are coordinated centrally; in fact, it seems increasingly likely that local circuits, often positioned within the epicardial fat pads of the heart, participate in the coordination of these structures (Figure 5-3).4,8
Atrioventricular Node and Cardiac Conduction System
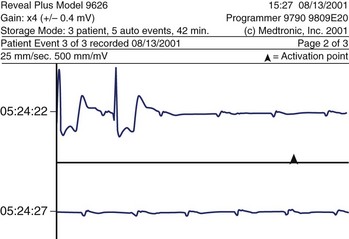
As a rule, AV nodal dromotropic responsiveness in the resting patient is under relatively balanced sympathetic and parasympathetic neural influence. However, this situation is readily altered by physiological events (e.g., exercise, sleep), the impact of disease states, drug effects, or during cardiac electrophysiology procedures when certain atrial regions are stimulated. Any tendency toward parasympathetic predominance markedly enhances the decremental properties of the AV node; in the extreme, this can be associated with transient complete AV nodal block (Figure 5-4). The latter is, in fact, a relatively common finding in sleeping patients and in very fit resting subjects. The relationship between ANS control of SN rate and AV conduction properties appears to foster both the maintenance of 1 : 1 AV conduction and a relatively optimal AV conduction interval.
Ventricular Myocardium
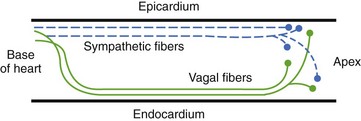
Ventricular sympathetics tend to lie within the subepicardial layer and follow the large coronary vessels as they spread out over the myocardium.9,10 The parasympathetics, in contrast, tend to penetrate the myocardium after crossing the AV groove and thereafter are subendocardial in location (Figure 5-5). The parasympathetic vagal efferents to the mycardium terminate not on the muscle cells themselves but on intracardiac ganglia. Evidence suggests that these ganglia not only form relay stations but also subserve certain local integrative functions, including the intracardiac reflex activity discussed earlier.