Chapter 4 Mechanisms of Re-entrant Arrhythmias
The most deadly cardiac arrhythmias result from re-entry, that is, electrical waves that rotate at a high frequency, in a self-sustaining manner. These waves give rise to electrical activity that propagates throughout the ventricles in complex ways. Spontaneous re-entry often occurs as a consequence of a wavebreak produced by the interaction of a propagating wavefront with a functional or anatomic obstacle.1 It is important to note, however, that the waves that break and initiate re-entry may be generated by electrical pacemaker discharges, triggered activity (i.e., early or delayed after-depolarizations), or another re-entry circuit. The objective of this chapter is to briefly examine the mechanisms of re-entrant arrhythmias. First, the chapter provides a brief historical perspective on the mechanisms of initiation and maintenance of reentry, including those mechanisms thought to underlie tachycardia and fibrillation. Emphasis is placed on the concepts derived from the theory of nonlinear wave of propagation in generic excitable media. This is followed by a summary of the work from the authors’ laboratory pertaining to functional re-entry in numerical simulations, engineered mouse models, and larger animals, as well as two-dimensional monolayers of neonatal rat ventricular myocytes. The aim of the chapter is to provide the interested reader with information to enhance didactic, clinical, or research endeavors.
What Is Re-entry?
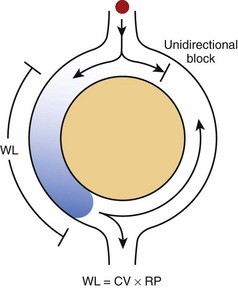
In its simplest form, re-entry is the circulation of the cardiac impulse around an obstacle; this leads to repetitive excitation of the heart at a frequency that depends on the conduction velocity of the circulating impulse and the perimeter of the obstacle (Figure 4-1).2 According to the original description by George Mines, which was published in The Journal of Physiology in 1913, re-entry occurs around a fixed anatomic obstacle, and the physical disruption of the surrounding circuit will interrupt the activity. As illustrated in Figure 4-1, the initiation of the re-entrant activity depends on the occurrence of a unidirectional block in which activation takes place in only one direction within the circuit.
It is clear from Figure 4-1 that the rotation time around the circuit should be longer than the recovery period of all segments of the circuit. The extra time required for the impulse to successfully complete a rotation may result from a relatively large circuit, a relatively slow conduction velocity of the impulse, or the relatively short duration of the refractory period. Hence, the “wavelength,” which may be calculated roughly as the product of the refractory period and the conduction velocity, must be shorter than the perimeter of the circuit. An excitable region will separate the front of the impulse from its own refractory tail (i.e., excitable gap) and re-excitation will ensue.
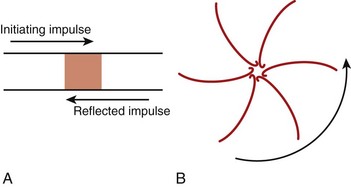
The classic model of anatomically determined re-entry depicted in Figure 4-1 is directly applicable to specific cases of tachyarrhythmias. These include supraventricular tachycardias occurring within the AV node or those using accessory pathways, as well as bundle branch re-entrant tachycardia. However, other types of re-entrant arrhythmias require somewhat different explanations for their mechanisms. For example, the cellular basis of closely coupled ventricular extrasystoles initiated somewhere in the Purkinje fiber network can be explained by the so-called reflection mechanism described by Antzelevitch et al in 1980 (Figure 4-2, A).3 Recently, reflection to and from an accessory pathway was shown to be a potential mechanism for the initiation of atrial fibrillation in patients with manifest (pre-excited) Wolff-Parkinson-White (WPW) syndrome.4
In the absence of a predetermined obstacle or circuit (Figure 4-2, B), however, many tachyarrhythmias that originate in the myocardium (atrial or ventricular) require mechanisms whereby re-entrant activation may occur as vortices of electrical excitation rotating around an area of myocardium. Accordingly, the impulse must circulate around a region of quiescence. In 1977, Allessie et al5 explained such functionally determined re-entry by proposing the so-called leading circle hypothesis, with its two variants of “anisotropic” re-entry described in 1988 by Dillon et al6 and “figure-of-8” re-entry proposed by El-Sherif7 in 1985. A somewhat different postulate for vortex-like re-entry, the “spiral wave re-entry” hypothesis, was put forth by Davidenko et al8 in 1992 and is derived from the theory of wave propagation in excitable media. Spiral wave re-entry attempts to provide a unifying explanation for the mechanisms of monomorphic and polymorphic ventricular tachycardias as well as the mechanism of fibrillation.
Circus Movement Re-entry
Functionally Determined Re-entry
In 1924, Garrey9 presented in an article in Physiological Reviews the first description of re-entrant excitation in the absence of anatomic obstacles in experimental studies on circus movement in the turtle heart. Garrey’s observations suggested that point stimulation of the atrium was sufficient to initiate a regular wave of rotation around the stimulus site. Subsequently, in 1946, Wiener and Rosenblueth10 developed the first mathematical model of circus movement re-entry, which supported waves of rotation around a sufficiently large barrier, but they could not demonstrate re-entry in the absence of an obstacle. This prompted Wiener and Rosenblueth to suggest that perhaps Garrey may have unwittingly produced a transient artificial obstacle near the stimulation site.
The “Leading Circle” Model
According to the leading-circle concept of Allessie and colleagues, in the absence of an anatomic obstacle, the dynamics of re-entry are determined by the smallest possible loop in which the impulse can continue to circulate. Under these conditions, the wavefront must propagate through relatively refractory tissue, in which case no “fully excitable gap” will be present and the wavelength will be very close to the length of the circuit. The leading circle idea paved the way for major advances in the understanding of functional re-entry. It served as a platform for developing a unifying hypothesis (spiral wave re-entry) that explains most of the major properties of functionally determined re-entry, which are commonly observed in normal cardiac muscle in experiments. This includes the phenomenon of re-entry “drift” described by Davidenko et al8 in 1992 and Pertsov et al11 in 1993. Re-entry drift results in beat-to-beat changes in the location of the rotation center (see Drifting Vortices and Ventricular Fibrillation).
Anisotropic Re-entry
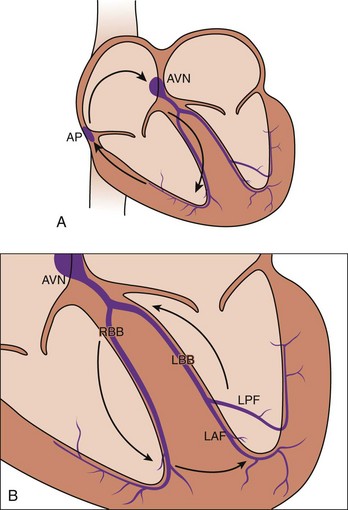
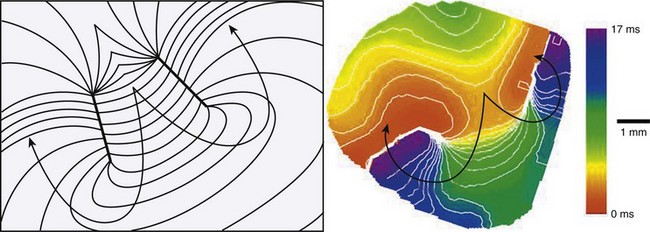
In 1986, Spach and Dolber12 implicated microscopic structural complexities of the cardiac muscle in the mechanism of re-entrant activation in both atria and ventricles, particularly in relation to the orientation of myocardial fibers, the manner in which the fibers and fiber bundles are connected to each other, and the effective electrical resistivities that depend on fiber orientation. Because of these structural properties, propagation velocity in the cardiac muscle is three to five times faster in the longitudinal axis of the cells than along the transverse axis. Mapping studies performed by Peters and Wit13 in 1998, using multiple extracellular electrodes have shown that, in the setting of myocardial infarction, re-entry may occur in the survival epicardial rim of tissue. Under such conditions, the wave circulates around a functionally determined elongated region of block, the so-called line of conduction block (Figure 4-4, A). Based on the orientation of the line of block, it was thought that anisotropic propagation played a major role both in the initiation as well as in the maintenance of re-entry in ventricular tissue surviving a myocardial infarction (Figure 4-4, B). In addition, propagation velocity is exceedingly slow at the edges of the lines of block, which has also been attributed to anisotropic propagation.
Figure-of-8 Re-entry
Figure-of-8 re-entry, described in 1987 by El-Sherif, Gough, and Restivo, has been recognized as an important pattern of re-entry in the late stages of myocardial infarction. In most cases, two counter-rotating waves coexist at a relatively short distance from each other (Figure 4-5). As described for the case of single re-entrant circuits, each wave of the figure-of-8 re-entry circulates around a thin line or arc of block. The region separating the lines of block is called the common pathway. A detailed description of the common pathway is of great practical importance, since there is evidence that it could be a strategic region for surgical or catheter ablation in this type of re-entry. In fact, unlike other forms of functionally determined re-entry, figure-of-8 re-entry may, indeed, be interrupted by physical disruption of the circuit. Several studies have attempted to describe the characteristics of propagation in the common pathway. However, the properties of the common pathway are still not clearly defined. It effectively behaves like an isthmus limited by two functionally determined barriers. In addition, there are two wavefronts that interact in the common pathway. As a result, propagation may be determined by a combination of factors other than those analyzed in most experimental studies such as anisotropy. The study of propagation across an isthmus and the influence of wavefront curvature may have significant implications in understanding the properties of the common pathway, as discussed in the next section.
Spiral Wave Re-entry
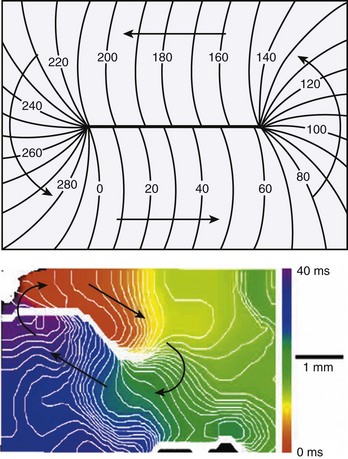
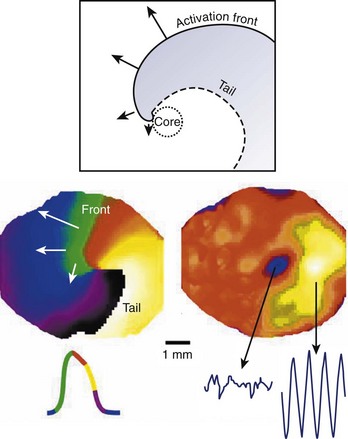
According to traditional concepts proposed by Allessie and colleagues, circus movement re-entry may be initiated in the heart because block is predetermined by the inhomogeneous functional characteristics of the tissue, whereas spiral waves could be formed in the heart even if cardiac muscle was completely homogeneous in its functional properties as shown by Davidenko et al8 in 1992, Pertsov et al11 in 1993, and Winfree14 in 1998. This is because the initiation of rotating activity may depend solely on transient local conditions (e.g., the conditions created by cross-field stimulation).1 Moreover, according to the traditional concept of re-entry, the circulation of the activity occurs around an anatomically or functionally predetermined circuit, and the rotating activity cannot drift. In other words, the circuit gives rise to and maintains the rotation. However, spiral waves occur due to initial curling of the wavefront; in fact, the curvature of the wavefront determines the size and shape of the region, called the core, around which activity rotates (Figure 4-6). Importantly, the core remains unexcited by the extremely curved activation front and it is readily excitable.1 This explains the mechanism underlying the drift of spirals.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree