UNSTABLE ANGINA AND NON-ST-SEGMENT ELEVATION MYOCARDIAL INFARCTION
Patients with ischemic heart disease fall into two large groups: patients with chronic coronary artery disease (CAD) who most commonly present with stable angina (Chap. 33) and patients with acute coronary syndromes (ACSs). The latter group, in turn, is composed of patients with acute myocardial infarction (MI) with ST-segment elevation on their presenting electrocardiogram (ECG) (STEMI; Chap. 35) and those with unstable angina (UA) and non-ST-segment elevation MI (UA/NSTEMI; Fig. 35-1). Every year in the United States, approximately 1 million patients are admitted to hospitals with UA/NSTEMI as compared with ~300,000 patients with acute STEMI. The relative incidence of UA/NSTEMI compared to STEMI appears to be increasing. More than one-third of patients with UA/NSTEMI are women, while less than one-fourth of patients with STEMI are women.
DEFINITION
The diagnosis of UA is based largely on the clinical presentation. Stable angina pectoris is characterized by chest or arm discomfort that may not be described as pain but is reproducibly associated with physical exertion or stress and is relieved within 5–10 min by rest and/or sublingual nitroglycerin (Chap. 4). UA is defined as angina pectoris or equivalent ischemic discomfort with at least one of three features: (1) it occurs at rest (or with minimal exertion), usually lasting >10 min; (2) it is severe and of new onset (i.e., within the prior 4–6 weeks); and/or (3) it occurs with a crescendo pattern (i.e., distinctly more severe, prolonged, or frequent than previously). The diagnosis of NSTEMI is established if a patient with the clinical features of UA develops evidence of myocardial necrosis, as reflected in elevated cardiac biomarkers.
PATHOPHYSIOLOGY
UA/NSTEMI is most commonly caused by a reduction in oxygen supply and/or by an increase in myocardial oxygen demand superimposed on a lesion that causes coronary arterial obstruction, usually an atherothrombotic coronary plaque. Four pathophysiologic processes that may contribute to the development of UA/NSTEMI have been identified: (1) plaque rupture or erosion with a superimposed nonocclusive thrombus, believed to be the most common cause; in such patients, NSTEMI may occur with downstream embolization of platelet aggregates and/or atherosclerotic debris; (2) dynamic obstruction (e.g., coronary spasm, as in Prinzmetal’s variant angina [PVA]); (3) progressive mechanical obstruction (e.g., rapidly advancing coronary atherosclerosis or restenosis following percutaneous coronary intervention [PCI]); and (4) UA secondary to increased myocardial oxygen demand and/or decreased supply (e.g., tachycardia, anemia). More than one of these processes may be involved.
Among patients with UA/NSTEMI studied at angiography, approximately 5% have stenosis of the left main coronary artery, 15% have three-vessel CAD, 30% have two-vessel disease, 40% have single-vessel disease, and 10% have no apparent critical epicardial coronary artery stenosis; some of the latter may have obstruction of the coronary microcirculation. The “culprit lesion” may show an eccentric stenosis with scalloped or overhanging edges and a narrow neck on angiography. Angioscopy has been reported to show “white” (platelet-rich) thrombi, as opposed to “red” (fibrin- and cell-rich) thrombi; the latter are more often seen in patients with acute STEMI. Patients with UA/NSTEMI frequently have multiple plaques at risk of disruption (vulnerable plaques).
CLINICAL PRESENTATION
History and physical examination
The clinical hallmark of UA/NSTEMI is chest pain, typically located in the substernal region or sometimes in the epigastrium, that radiates to the neck, left shoulder, and/or the left arm (Chap. 4). This discomfort is usually severe enough to be described as frank pain. Anginal “equivalents” such as dyspnea and epigastric discomfort may also occur, and these appear to be more frequent in women. The physical examination resembles that in patients with stable angina (Chap. 33) and may be unremarkable. If the patient has a large area of myocardial ischemia or a large NSTEMI, the physical findings can include diaphoresis; pale, cool skin; sinus tachycardia; a third and/or fourth heart sound; basilar rales; and, sometimes, hypotension, resembling the findings of large STEMI.
Electrocardiogram
In UA, ST-segment depression, transient ST-segment elevation, and/or T-wave inversion occur in 30% to 50% of patients. In patients with the clinical features of UA, the presence of new ST-segment deviation, even of only 0.05 mV, is an important predictor of adverse outcome. T-wave changes are sensitive for ischemia but less specific, unless they are new, deep T-wave inversions (≥0.3 mV).
Cardiac biomarkers
Patients with UA/NSTEMI who have elevated biomarkers of necrosis, such as CK-MB and troponin (a much more specific and sensitive marker of myocardial necrosis), are at increased risk for death or recurrent MI. Elevated levels of these markers distinguish patients with NSTEMI from those with UA. There is a direct relationship between the degree of troponin elevation and mortality. However, in patients without a clear clinical history of myocardial ischemia, minor troponin elevations have been reported and can be caused by congestive heart failure (CHF), myocarditis, or pulmonary embolism, or they may be false-positive readings. Thus, in patients with an unclear history, small troponin elevations may not be diagnostic of an ACS.
DIAGNOSTIC EVALUATION
(See also Chap. 4) Approximately 6 million persons per year in the United States present to hospital emergency departments (EDs) with a complaint of chest pain or other symptoms suggestive of ACS. A diagnosis of an ACS is established in 20% to 25% of such patients. The first step in evaluating patients with possible UA/NSTEMI is to determine the likelihood that CAD is the cause of the presenting symptoms. The American College of Cardiology/American Heart Association (ACC/AHA) guidelines include, among the factors associated with a high likelihood of ACS, a prior history typical of stable angina, a history of established CAD by angiography, prior MI, CHF, new ECG changes, or elevated cardiac biomarkers.
Diagnostic pathways
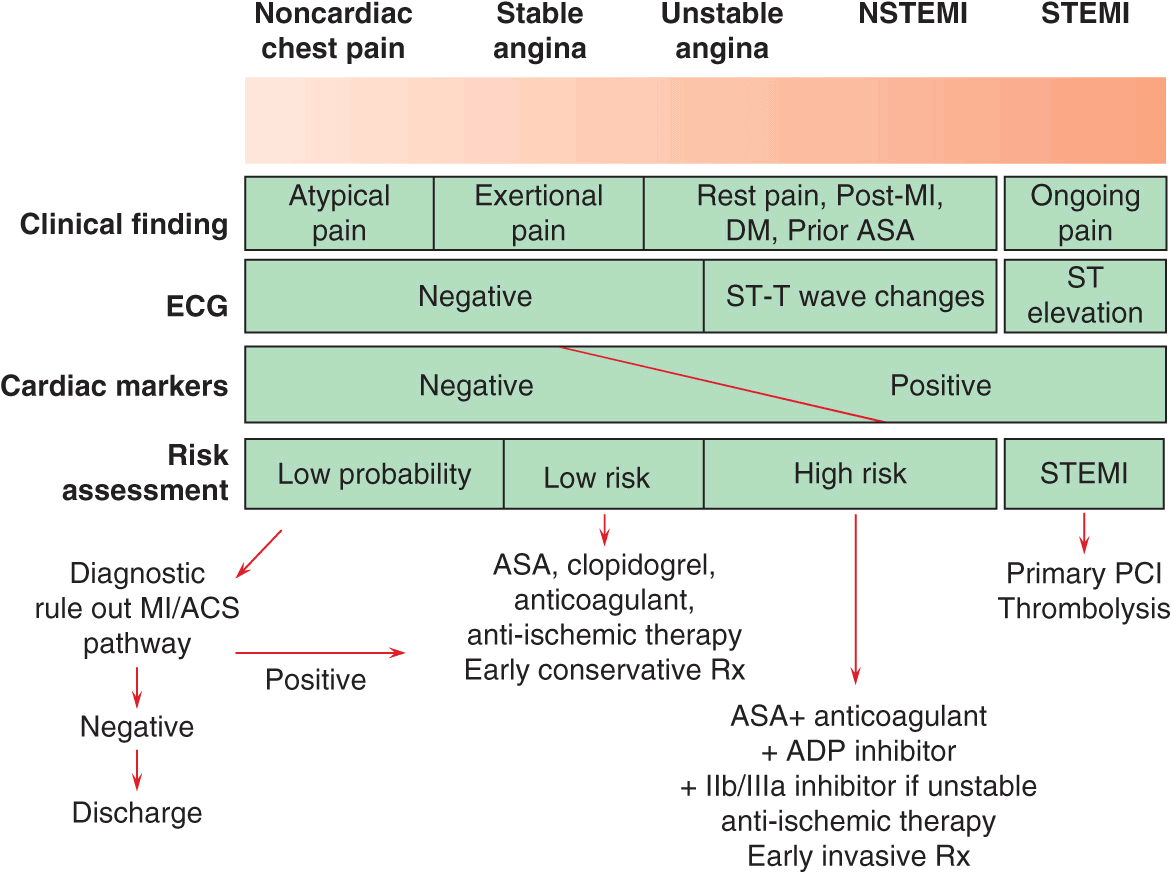
Four major diagnostic tools are used in the diagnosis of UA/NSTEMI in the ED: clinical history, the ECG, cardiac markers, and stress testing (coronary imaging is an emerging option). The goals are to: (1) recognize or exclude MI (using cardiac markers), (2) evaluate for rest ischemia (using serial or continuous ECGs), and (3) evaluate for significant CAD (using provocative stress testing). Patients with a low likelihood of ischemia are usually managed with an ED-based critical pathway (which, in some institutions, is carried out in a “chest-pain unit” Fig. 34-1). Evaluation of such patients includes clinical monitoring for recurrent ischemic discomfort, serial ECGs, and cardiac markers, typically obtained at baseline and at 4–6 h and 12 h after presentation. If new elevations in cardiac markers or ECG changes are noted, the patient should be admitted to the hospital. If the patient remains pain free and the markers are negative, the patient may proceed to stress testing. CT angiography is used with increasing frequency to exclude obstructive CAD (Chap. 12).
FIGURE 34-1
Algorithm for risk stratification and treatment of patients with suspected coronary artery disease. Using the clinical history of the type of pain and medical history, the ECG, and cardiac markers, one can identify patients who have a low likelihood of UA/NSTEMI, for whom a diagnostic “ruleout myocardial infarction (MI) or acute coronary syndrome (ACS)” is warranted. If this is negative, the patient may be discharged, but if positive, the patient is admitted and treated for UA/NSTEMI. On the other end of the spectrum, patients with acute ongoing pain and ST-segment elevation are treated with percutaneous coronary intervention (PCI) or fibrinolysis (Chap. 35). For those with UA/NSTEMI, risk stratification is used to identify patients at medium to high risk, for whom an early invasive strategy is warranted. Antithrombotic therapy should include aspirin, an anticoagulant, an ADP antagonist (clopidogrel or prasugrel), with GP IIb/IIIa inhibition considered for use during PCI. For patients at low risk, treatment with aspirin, clopidogrel, an anticoagulant such as unfractionated or low molecular–weight heparin (LMWH) or fondaparinux and anti-ischemic therapy with beta blockers and nitrates, and a conservative strategy are indicated. ASA, aspirin; DM, diabetes mellitus; ECG, electrocardiogram; MI, myocardial infarction; Rx, treatment; STEMI, ST-segment elevation myocardial infarction. (Adapted from CP Cannon, E Braunwald, in Braunwald’s Heart Disease: A Textbook of Cardiovascular Medicine, 9th ed, R Bonow et al [eds]. Philadelphia, Saunders, 2011.)
RISK STRATIFICATION AND PROGNOSIS
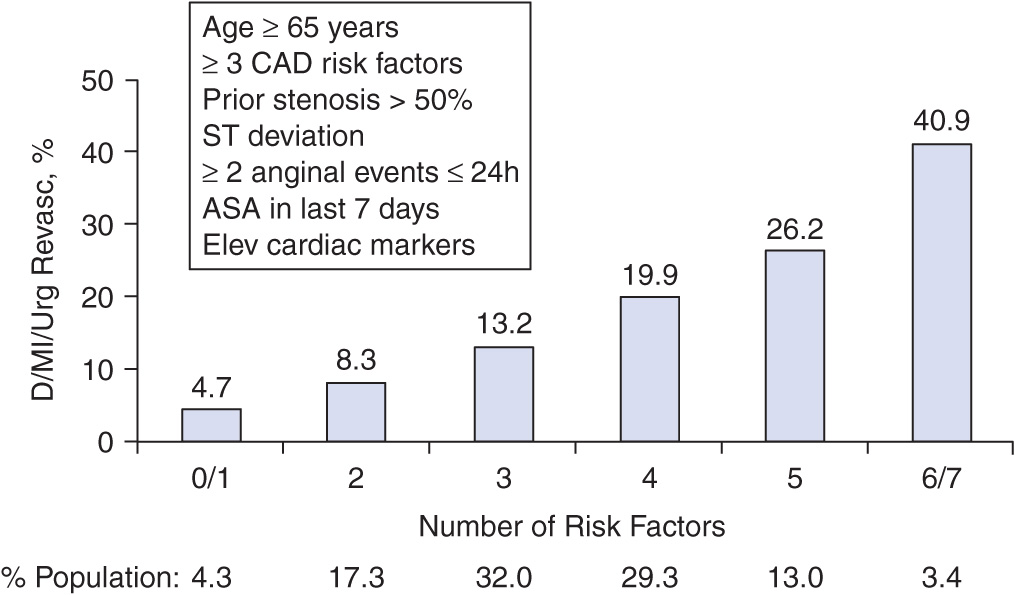
Patients with documented UA/NSTEMI exhibit a wide spectrum of early (30 days) risk of death, ranging from 1% to 10%, and of new or recurrent infarction of 3–5% or recurrent ACS (5-15%). Assessment of risk can be accomplished by clinical risk scoring systems such as that developed from the Thrombolysis in Myocardial Infarction (TIMI) Trials, which includes seven independent risk factors: age ≥65 years, three or more risk factors for CAD, documented CAD at catheterization, development of UA/NSTEMI while on aspirin, more than two episodes of angina within the preceding 24 h, ST deviation ≥0.5 mm, and an elevated cardiac marker (Fig. 34-2). Other risk factors include diabetes mellitus, left ventricular dysfunction, renal dysfunction and elevated levels of brain natriuretic peptides and C-reactive protein. Multimarker strategies involving several biomarkers are now gaining favor, both to define more fully the pathophysiologic mechanisms underlying a given patient’s presentation and to stratify the patient’s risk further. Early risk assessment (especially using troponin, ST-segment changes, and/or a global risk-scoring system) is useful both in predicting the risk of recurrent cardiac events and in identifying those patients who would derive the greatest benefit from antithrombotic therapies more potent than unfractionated heparin, such as low molecular–weight heparin (LMWH) and glycoprotein IIb/IIIa inhibitors, and from an early invasive strategy. For example, in the TACTICSTIMI 18 Trial, an early invasive strategy conferred a 40% reduction in recurrent cardiac events in patients with a positive troponin level, whereas no benefit was observed in those without detectable troponin.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree