APPROACH TO THE PATIENT WITH A HEART MURMUR
INTRODUCTION
The differential diagnosis of a heart murmur begins with a careful assessment of its major attributes and responses to bedside maneuvers. The history, clinical context, and associated physical examination findings provide additional clues by which the significance of a heart murmur is established. Accurate bedside identification of a heart murmur can inform decisions regarding the indications for noninvasive testing and the need for referral to a cardiovascular specialist. Preliminary discussions can be held with the patient regarding antibiotic or rheumatic fever prophylaxis, the need to restrict various forms of physical activity, and the potential role for family screening.
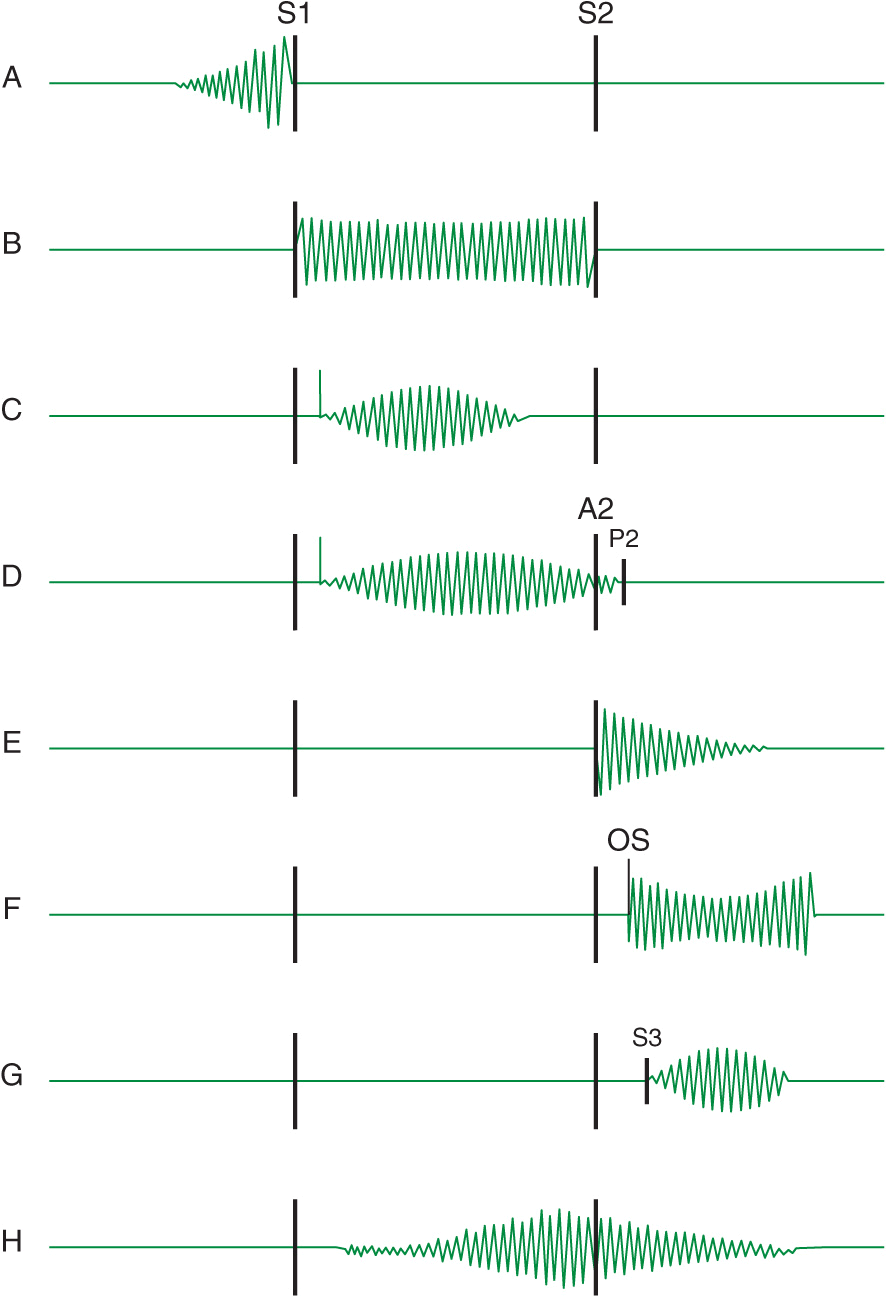
Heart murmurs are caused by audible vibrations that are due to increased turbulence from accelerated blood flow through normal or abnormal orifices, flow through a narrowed or irregular orifice into a dilated vessel or chamber, or backward flow through an incompetent valve, ventricular septal defect, or patent ductus arteriosus. They traditionally are defined in terms of their timing within the cardiac cycle (Fig. 10-1). Systolic murmurs begin with or after the first heart sound (S1) and terminate at or before the component (A2 or P2) of the second heart sound (S2) that corresponds to their site of origin (left or right, respectively). Diastolic murmurs begin with or after the associated component of S2 and end at or before the subsequent S1. Continuous murmurs are not confined to either phase of the cardiac cycle but instead begin in early systole and proceed through S2 into all or part of diastole. The accurate timing of heart murmurs is the first step in their identification. The distinction between S1 and S2 and, therefore, systole and diastole is usually a straightforward process but can be difficult in the setting of a tachyarrhythmia, in which case the heart sounds can be distinguished by simultaneous palpation of the carotid upstroke, which should closely follow S1.
FIGURE 10-1
Diagram depicting principal heart murmurs. A. Presystolic murmur of mitral or tricuspid stenosis. B. Holosystolic (pansystolic) murmur of mitral or tricuspid regurgitation or of ventricular septal defect. C. Aortic ejection murmur beginning with an ejection click and fading before the second heart sound. D. Systolic murmur in pulmonic stenosis spilling through the aortic second sound, pulmonic valve closure being delayed. E. Aortic or pulmonary diastolic murmur. F. Long diastolic murmur of mitral stenosis after the opening snap (OS). G. Short mid-diastolic inflow murmur after a third heart sound. H. Continuous murmur of patent ductus arteriosus. (Adapted from P Wood, Diseases of the Heart and Circulation, London, Eyre & Spottiswood, 1968. Permission granted courtesy of Antony and Julie Wood.)
Duration
The duration of a heart murmur depends on the length of time over which a pressure difference exists between two cardiac chambers, the left ventricle and the aorta, the right ventricle and the pulmonary artery, or the great vessels. The magnitude and variability of this pressure difference, coupled with the geometry and compliance of the involved chambers or vessels, dictate the velocity of flow; the degree of turbulence; and the resulting frequency, configuration, and intensity of the murmur. The diastolic murmur of chronic aortic regurgitation (AR) is a blowing, high-frequency event, whereas the murmur of mitral stenosis (MS), indicative of the left atrial–left ventricular diastolic pressure gradient, is a low-frequency event, heard as a rumbling sound with the bell of the stethoscope. The frequency components of a heart murmur may vary at different sites of auscultation. The coarse systolic murmur of aortic stenosis (AS) may sound higher pitched and more acoustically pure at the apex, a phenomenon eponymously referred to as the Gallavardin effect. Some murmurs may have a distinct or unusual quality, such as the “honking” sound appreciated in some patients with mitral regurgitation (MR) due to mitral valve prolapse (MVP).
The configuration of a heart murmur may be described as crescendo, decrescendo, crescendo-decrescendo, or plateau. The decrescendo configuration of the murmur of chronic AR (Fig. 10-1E) can be understood in terms of the progressive decline in the diastolic pressure gradient between the aorta and the left ventricle. The crescendo-decrescendo configuration of the murmur of AS reflects the changes in the systolic pressure gradient between the left ventricle and the aorta as ejection occurs, whereas the plateau configuration of the murmur of chronic MR (Fig. 10-1B) is consistent with the large and nearly constant pressure difference between the left ventricle and the left atrium.
Intensity
The intensity of a heart murmur is graded on a scale of 1–6 (or I–VI). A grade 1 murmur is very soft and is heard only with great effort. A grade 2 murmur is easily heard but not particularly loud. A grade 3 murmur is loud but is not accompanied by a palpable thrill over the site of maximal intensity. A grade 4 murmur is very loud and is accompanied by a thrill. A grade 5 murmur is loud enough to be heard with only the edge of the stethoscope touching the chest, whereas a grade 6 murmur is loud enough to be heard with the stethoscope slightly off the chest. Murmurs of grade 3 or greater intensity usually signify important structural heart disease and indicate high blood flow velocity at the site of murmur production. Small ventricular septal defects (VSDs), for example, are accompanied by loud, usually grade 4 or greater, systolic murmurs as blood is ejected at high velocity from the left ventricle to the right ventricle. Low-velocity events, such as left-to-right shunting across an atrial septal defect (ASD), are usually silent. The intensity of a heart murmur also may be diminished by any process that increases the distance between the intracardiac source and the stethoscope on the chest wall, such as obesity, obstructive lung disease, and a large pericardial effusion. The intensity of a murmur also may be misleadingly soft when cardiac output is reduced significantly or when the pressure gradient between the involved cardiac structures is low.
Location and radiation
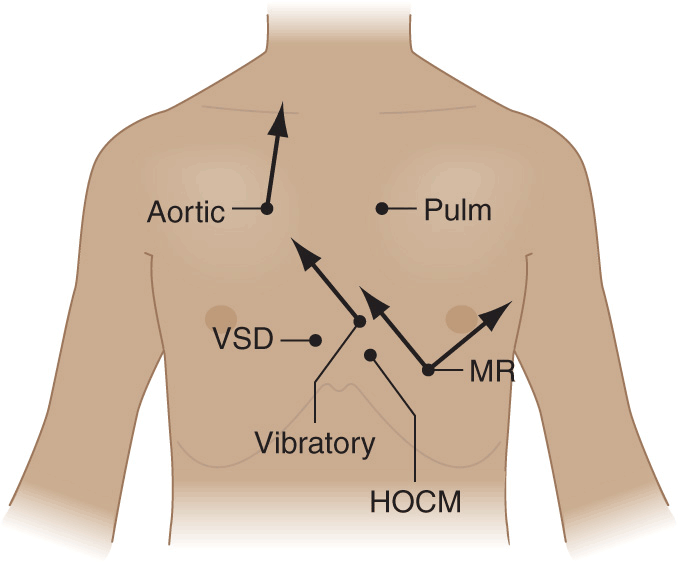
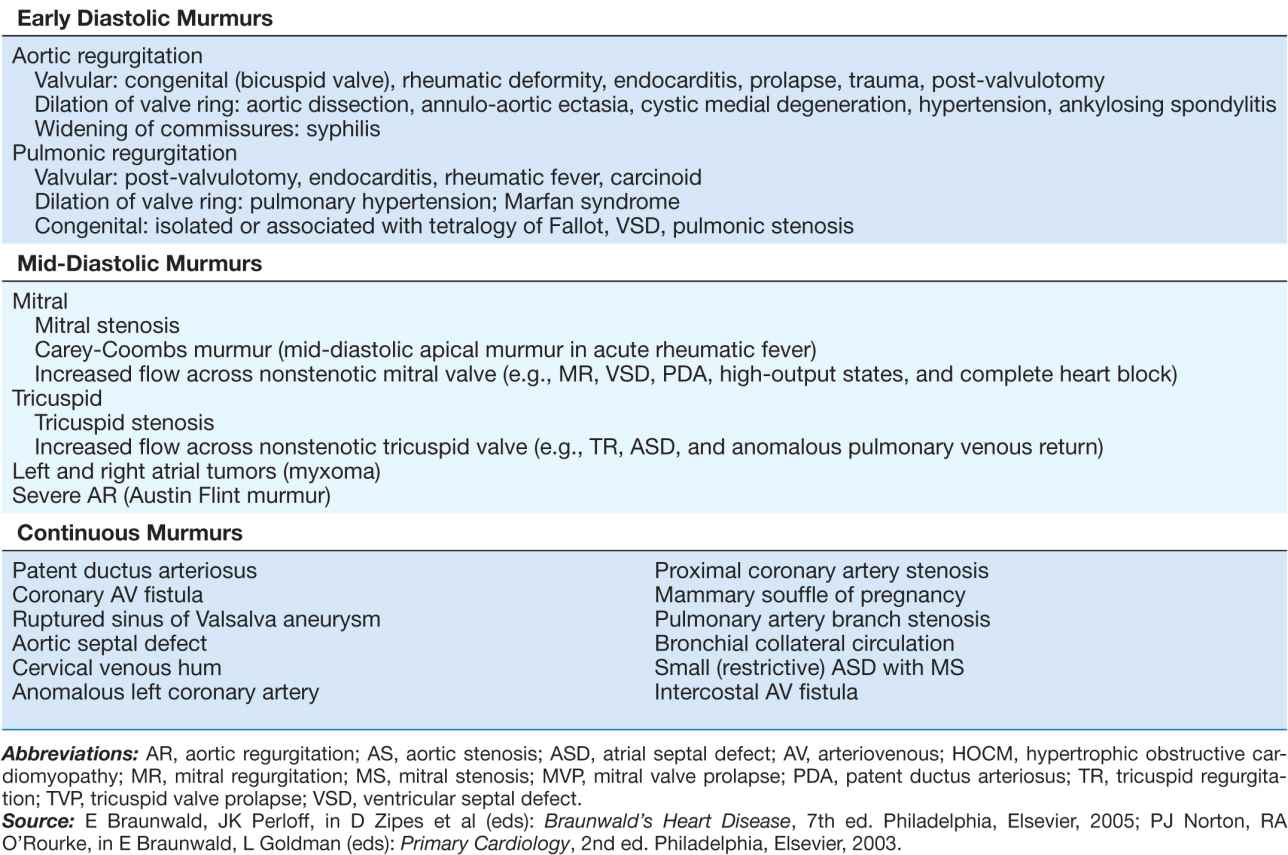
Recognition of the location and radiation of the murmur helps facilitate its accurate identification (Fig. 10-2). Adventitious sounds, such as a systolic click or diastolic snap, or abnormalities of S1 or S2 may provide additional clues. Careful attention to the characteristics of the murmur and other heart sounds during the respiratory cycle and the performance of simple bedside maneuvers complete the auscultatory examination. These features, along with recommendations for further testing, are discussed later in the context of specific systolic, diastolic, and continuous heart murmurs (Table 10-1).
FIGURE 10-2
Maximal intensity and radiation of six isolated systolic murmurs. HOCM, hypertrophic obstructive cardiomyopathy; MR, mitral regurgitation; Pulm, pulmonary stenosis; Aortic, aortic stenosis; VSD, ventricular septal defect. (From JB Barlow, Perspectives on the Mitral Valve. Philadelphia, FA Davis, 1987, p 140.)
TABLE 10-1
PRINCIPAL CAUSES OF HEART MURMURS

SYSTOLIC HEART MURMURS
Early systolic murmurs
Early systolic murmurs begin with S1 and extend for a variable period, ending well before S2. Their causes are relatively few in number. Acute, severe MR into a normalsized, relatively noncompliant left atrium results in an early, decrescendo systolic murmur best heard at or just medial to the apical impulse. These characteristics reflect the progressive attenuation of the pressure gradient between the left ventricle and the left atrium during systole owing to the rapid rise in left atrial pressure caused by the sudden volume load into an unprepared chamber and contrast sharply with the auscultatory features of chronic MR. Clinical settings in which acute, severe MR occur include (1) papillary muscle rupture complicating acute myocardial infarction (MI) (Chap. 35), (2) rupture of chordae tendineae in the setting of myxomatous mitral valve disease (MVP, Chap. 20), (3) infective endocarditis (Chap. 25), and (4) blunt chest wall trauma.
Acute, severe MR from papillary muscle rupture usually accompanies an inferior, posterior, or lateral MI and occurs 2–7 days after presentation. It often is signaled by chest pain, hypotension, and pulmonary edema, but a murmur may be absent in up to 50% of cases. The posteromedial papillary muscle is involved 6 to 10 times more frequently than the anterolateral papillary muscle. The murmur is to be distinguished from that associated with post-MI ventricular septal rupture, which is accompanied by a systolic thrill at the left sternal border in nearly all patients and is holosystolic in duration. A new heart murmur after an MI is an indication for transthoracic echocardiography (TTE) (Chap. 12), which allows bedside delineation of its etiology and pathophysiologic significance. The distinction between acute MR and ventricular septal rupture also can be achieved with right heart catheterization, sequential determination of oxygen saturations, and analysis of the pressure waveforms (tall v wave in the pulmonary artery wedge pressure in MR). Post-MI mechanical complications of this nature mandate aggressive medical stabilization and prompt referral for surgical repair.
Spontaneous chordal rupture can complicate the course of myxomatous mitral valve disease (MVP) and result in new-onset or “acute on chronic” severe MR. MVP may occur as an isolated phenomenon, or the lesion may be part of a more generalized connective tissue disorder as seen, for example, in patients with Marfan syndrome. Acute, severe MR as a consequence of infective endocarditis results from destruction of leaflet tissue, chordal rupture, or both. Blunt chest wall trauma is usually self-evident but may be disarmingly trivial; it can result in papillary muscle contusion and rupture, chordal detachment, or leaflet avulsion. TTE is indicated in all cases of suspected acute, severe MR to define its mechanism and severity, delineate left ventricular size and systolic function, and provide an assessment of suitability for primary valve repair.
A congenital, small muscular VSD (Chap. 19) may be associated with an early systolic murmur. The defect closes progressively during septal contraction, and thus, the murmur is confined to early systole. It is localized to the left sternal border (Fig. 10-2) and is usually of grade 4 or 5 intensity. Signs of pulmonary hypertension or left ventricular volume overload are absent. Anatomically large and uncorrected VSDs, which usually involve the membranous portion of the septum, may lead to pulmonary hypertension. The murmur associated with the left-to-right shunt, which earlier may have been holosystolic, becomes limited to the first portion of systole as the elevated pulmonary vascular resistance leads to an abrupt rise in right ventricular pressure and an attenuation of the interventricular pressure gradient during the remainder of the cardiac cycle. In such instances, signs of pulmonary hypertension (right ventricular lift, loud and single or closely split S2) may predominate. The murmur is best heard along the left sternal border but is softer. Suspicion of a VSD is an indication for TTE.
Tricuspid regurgitation (TR) with normal pulmonary artery pressures, as may occur with infective endocarditis, may produce an early systolic murmur. The murmur is soft (grade 1 or 2), is best heard at the lower left sternal border, and may increase in intensity with inspiration (Carvallo’s sign). Regurgitant “c–v” waves may be visible in the jugular venous pulse. TR in this setting is not associated with signs of right heart failure.
Mid-systolic murmurs
Mid-systolic murmurs begin at a short interval after S1, end before S2 (Fig. 10-1C), and are usually crescendo-decrescendo in configuration. Aortic stenosis is the most common cause of a mid-systolic murmur in an adult. The murmur of AS is usually loudest to the right of the sternum in the second intercostal space (aortic area, Fig. 10-2) and radiates into the carotids. Transmission of the midsystolic murmur to the apex, where it becomes higher pitched, is common (Gallavardin effect; see earlier).
Differentiation of this apical systolic murmur from MR can be difficult. The murmur of AS will increase in intensity, or become louder, in the beat after a premature beat, whereas the murmur of MR will have constant intensity from beat to beat. The intensity of the AS murmur also varies directly with the cardiac output. With a normal cardiac output, a systolic thrill and a grade 4 or higher murmur suggest severe AS. The murmur is softer in the setting of heart failure and low cardiac output. Other auscultatory findings of severe AS include a soft or absent A2, paradoxical splitting of S2, an apical S4, and a late-peaking systolic murmur. In children, adolescents, and young adults with congenital valvular AS, an early ejection sound (click) is usually audible, more often along the left sternal border than at the base. Its presence signifies a flexible, noncalcified bicuspid valve (or one of its variants) and localizes the left ventricular outflow obstruction to the valvular (rather than sub- or supravalvular) level.
Assessment of the volume and rate of rise of the carotid pulse can provide additional information. A small and delayed upstroke (parvus et tardus) is consistent with severe AS. The carotid pulse examination is less discriminatory, however, in older patients with stiffened arteries. The electrocardiogram (ECG) shows signs of left ventricular hypertrophy (LVH) as the severity of the stenosis increases. TTE is indicated to assess the anatomic features of the aortic valve, the severity of the stenosis, left ventricular size, wall thickness and function, and the size and contour of the aortic root and proximal ascending aorta.
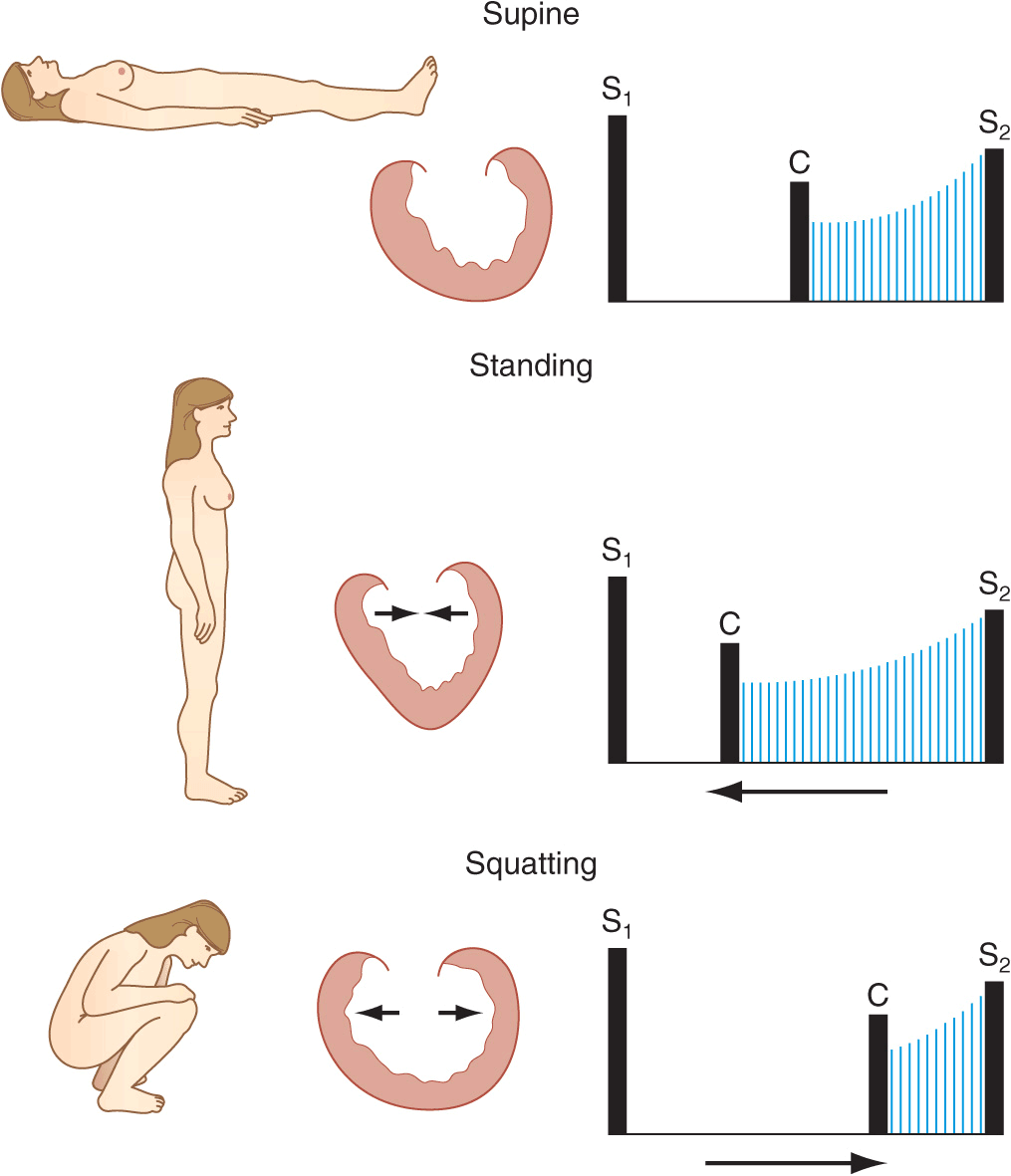
The obstructive form of hypertrophic cardiomyopathy (HOCM) is associated with a mid-systolic murmur that is usually loudest along the left sternal border or between the left lower sternal border and the apex (Chap. 21, Fig. 10-2). The murmur is produced by both dynamic left ventricular outflow tract obstruction and MR, and thus, its configuration is a hybrid between ejection and regurgitant phenomena. The intensity of the murmur may vary from beat to beat and after provocative maneuvers but usually does not exceed grade 3. The murmur classically will increase in intensity with maneuvers that result in increasing degrees of outflow tract obstruction, such as a reduction in preload or afterload (Valsalva, standing, vasodilators), or with an augmentation of contractility (inotropic stimulation). Maneuvers that increase preload (squatting, passive leg raising, volume administration) or afterload (squatting, vasopressors) or that reduce contractility (β-adrenoreceptor blockers) decrease the intensity of the murmur. In rare patients, there may be reversed splitting of S2. A sustained left ventricular apical impulse and an S4 may be appreciated. In contrast to AS, the carotid upstroke is rapid and of normal volume. Rarely, it is bisferiens or bifid in contour (see Fig. 9-2D) due to mid-systolic closure of the aortic valve. LVH is present on the ECG, and the diagnosis is confirmed by TTE. Although the systolic murmur associated with MVP behaves similarly to that due to HOCM in response to the Valsalva maneuver and to standing/squatting (Fig. 10-3), these two lesions can be distinguished on the basis of their associated findings, such as the presence of LVH in HOCM or a nonejection click in MVP.
FIGURE 10-3
A mid-systolic nonejection sound (C) occurs in mitral valve prolapse and is followed by a late systolic murmur that crescendos to the second heart sound (S2). Standing decreases venous return; the heart becomes smaller; C moves closer to the first heart sound (S1), and the mitral regurgitant murmur has an earlier onset. With prompt squatting, venous return increases; the heart becomes larger; C moves toward S2, and the duration of the murmur shortens. (From JA Shaver, JJ Leonard, DF Leon, Examination of the Heart, Part IV, Auscultation of the Heart. Dallas, American Heart Association, 1990, p 13. Copyright, American Heart Association.)
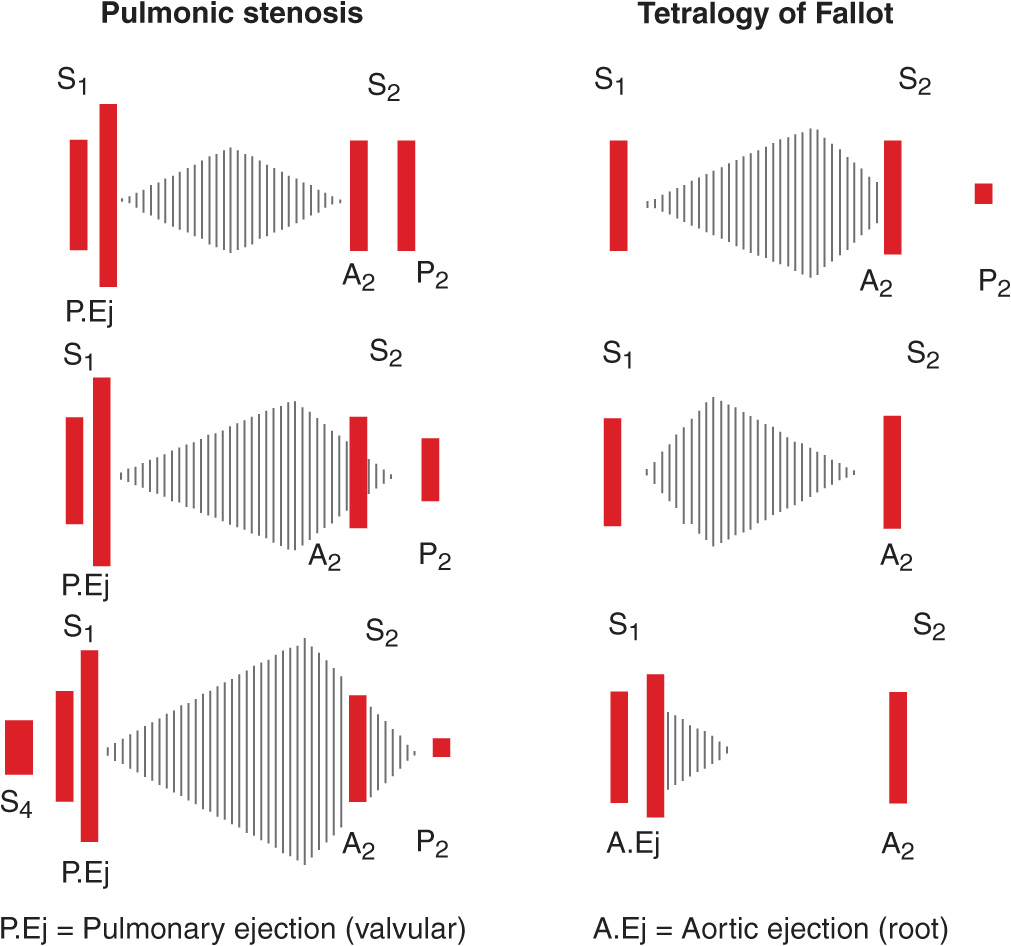
The mid-systolic, crescendo-decrescendo murmur of congenital pulmonic stenosis (PS, Chap. 19) is best appreciated in the second and third left intercostal spaces (pulmonic area) (Figs. 10-2 and 10-4). The duration of the murmur lengthens and the intensity of P2 diminishes with increasing degrees of valvular stenosis (Fig. 10-1D). An early ejection sound, the intensity of which decreases with inspiration, is heard in younger patients. A parasternal lift and ECG evidence of right ventricular hypertrophy indicate severe pressure overload. If obtained, the chest x-ray may show poststenotic dilation of the main pulmonary artery. TTE is recommended for complete characterization.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree