Chapter 97 Surgery for Ventricular Arrhythmias
Surgical Methods for Controlling Ventricular Tachyarrhythmias
Indirect Methods
The first reported surgical cure of VT resulted from excision of a left ventricular aneurysm. When aneurysmectomy was used more widely as a treatment for VT, however, it became apparent that this procedure had low efficacy and high mortality rates (often because of intractable VT). CABG was also used in an attempt to control VT, with similarly poor results. These methods generally failed because although aneurysms are frequently present in patients with VT, the excised portion of the aneurysm does not contain critical portions of VT circuits, and VT episodes usually do not result from acute ischemia (which could be corrected by coronary bypass). CABG has definite application in relatively rare cases of polymorphic VT or VF that clearly occurs in the setting of acute ischemia. CABG and aneurysmectomy are both still performed in some cases of VT but only as adjunctive methods along with one of the more direct techniques discussed below. Neural modification, typically with left or bilateral stellate ganglionectomy, has benefit in selected patients with VTAs: patients with long QT syndrome (LQTS) who have recurrent syncope or cardiac arrest despite β-blocker and ICD therapies; patients with recurrent episodes of VF in the absence of SHD (idiopathic VF); and patients with catecholaminergic polymorphic VT. A variety of approaches have been used for the destruction or removal of the left or bilateral stellate ganglia (cervical, thoracotomy incisions). Thoracoscopic techniques are being more widely used for approaching the stellate ganglion, rather than open surgical exposure.1
Direct Methods
After the failure of CABG and aneurysmectomy to reliably affect VT recurrence, several investigators began using intraoperative electrophysiological mapping studies to understand more about where VT was originating. The rationale was that a site in the ventricle from which VT arose, or exited from a circuit, should precede the onset of each surface QRS complex by greater than 40 ms, or even occur in mid-diastole. Epicardial mapping during VT usually did not reveal sites of activation that met this criterion; endocardial mapping, after ventriculotomy, routinely showed sites activated prior to the QRS during VT (Figure 97-1). These areas were typically near the border between the aneurysm or the dense infarct and more normal muscle. Several means of therapy targeting these regions were devised in the late 1970s, including excision (endocardial resection) an isolating incision (encircling ventriculotomy), and cryoablation. Additional modalities have been used, including intraoperative laser photoablation. Because these hearts have already suffered extensive damage from the infarction that caused the arrhythmia, a cardinal principle of surgical therapy is to remove or destroy only as much tissue as is necessary to eliminate the VT while damaging as little remaining normal myocardium as possible. Mapping tools and techniques have aided in the discrimination between normally and abnormally functioning tissue.
Ventricular Tachycardia
Preoperative Studies
Because most patients undergoing surgery for VT have prior infarction from coronary atherosclerosis, cardiac catheterization with coronary arteriography is an important part of the preoperative evaluation. Coronary stenoses that should undergo bypass grafting can thus be identified and any possible valvular lesions identified. Ventriculography (assessing overall left ventricular function) can be helpful in estimating operative risk. In the presence of coronary stenoses of uncertain flow-limiting significance, stress testing with some form of perfusion imaging can help determine whether bypass grafting of particular arteries would be beneficial. Preoperative electrophysiological study (EPS) is almost always indicated (except in cases of severe left main stenosis or three-vessel disease, in which the rapid heart rates encountered during induced VT could possibly be detrimental). This study can provide information as to how readily inducible VT is as a guide to how vigorous intraoperative stimulation will need to be. Endocardial catheter mapping can also be performed at this time to help focus attention on certain areas that need to be ablated during surgery. However, if mapping is performed in this setting, it is almost always accompanied by ablation attempts; if the ablation successfully eliminates inducibility of one or more morphologies of VT, they no longer need surgical attention. If ablation is not successful, the validity of the mapping information may be called into question. Thus preoperative mapping may have a more limited role than was once thought. Percutaneous access to the pericardial space has been used for epicardial mapping and ablation; this is more frequently necessary in cardiomyopathic cases than in post-MI VT. Recently, a hybrid procedure has been developed in which epicardial catheter mapping has been facilitated by surgical exposure of the pericardial space using a small subxiphoid incision. This is particularly helpful when prior cardiac surgery has resulted in extensive pericardial adhesions that would significantly restrict catheter mobility and access to all areas of the epicardial surface.2
Principles of Cardiac Mapping
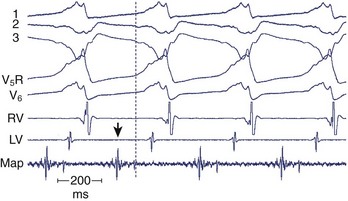
If mapping is to be performed, several pieces of equipment are necessary beyond the usual requirements for open-heart surgery. These include electrodes, a mapping system (consisting of amplifiers and a recording and analysis system), and a stimulator with which to initiate VT. Several commercial mapping systems are available, capable of recording from 64 to 256 electrodes simultaneously, online analysis of activation times, and generation of isochronal maps. Most record onto magnetic or optical media for data archiving. At the time of surgery, the electrocardiogram (ECG) leads from the patient are connected to the mapping system. The heart is exposed through a median sternotomy, and a pericardial cradle is formed. Cannulae are placed for cardiopulmonary bypass, and reference electrodes are inserted and tested. The heart is inspected for regions of infarct or aneurysm, through which the ventricle may be entered without damaging the viable myocardium; sometimes, this area is only evident after the patient is on cardiopulmonary bypass and the left ventricle (LV) has been vented. Cardiopulmonary bypass is then established while maintaining a perfusate temperature of 37° C to 38° C. This is necessary because cardiac electrical activity deteriorates at cooler temperatures and arrhythmias may not be inducible or mappable. Radiant heat loss of the heart surface during mapping may necessitate even slightly higher perfusate temperatures. Epicardial mapping during sinus rhythm or VT can be performed with the heart closed, but this is usually omitted in the interests of time unless specific circumstances suggest its usefulness. In most cases, once the heart-lung machine is running well, the LV is then opened through the previously identified infarct or aneurysm. The endocardial surface is inspected for the presence of any adherent thrombus, which is then removed. An electrode or multipolar electrode array is placed on the endocardial surface in an area of obvious scar tissue. At this time, sinus rhythm mapping can be performed to designate areas with abnormal electrograms indicating damaged myocardium; this step usually adds little and is omitted except in special situations. In most cases, VT is then initiated using previously placed electrodes, and endocardial mapping is performed during VT. Some tissue is activated at every instant during a re-entrant arrhythmia, even during the diastolic interval between discrete QRS complexes; sites from which diastolic activation is recorded during VT are of particular interest, since these regions are often in “protected” corridors that are critical for continued re-entry. Computerized mapping systems are able to quickly display these areas of diastolic activation (Figure 97-2). Because most patients with post-infarct VT have more than one ECG morphology of VT that may arise from the same or different regions, attempts are made to initiate and map other morphologies of VT. Because of time limitations, rigorous entrainment studies (overdrive pacing during VT) to validate the activation mapping data are often not performed. Once all inducible morphologies of VT have been mapped, the electrode array is removed and the ablation portion of the procedure begins. Several different procedures have been used for this purpose (Figure 97-3), including:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree