Chapter 78 Arrhythmias Associated with Congenital Heart Disease
Arrhythmia Substrates in Patients with Congenital Heart Disease
Arrhythmia Substrates in Unrepaired Patients with Congenital Heart Disease
Accessory Pathways
Congenital heart disease is relatively common in the general population (~1% of births), and accessory pathways are also somewhat common (1.6 to 3 of 1000 live births). Therefore, one would expect to observe Wolff-Parkinson-White (WPW) syndrome and supraventricular tachycardia (SVT) on the basis of these incidences alone. Indeed, in the Pediatric Radiofrequency Ablation Registry, ablations for accessory pathways have been reported in patients with most types of defects.1 However, it is well known that certain types of congenital cardiac disease are more commonly associated with WPW syndrome. The most prominent of these defects is Ebstein’s anomaly of the tricuspid valve.2,3 In patients with these defects, the prevalence of WPW is approximately 9%. Other defects reported to demonstrate an increased association with WPW include L-transposition of the great vessels (ventricular inversion and congenitally corrected transposition) and hypertrophic cardiomyopathy.4–8 In patients with Ebstein’s anomaly, accessory pathways are often multiple and are generally right sided, although occasionally a posteroseptal pathway location is present. In L-transposition, one sees an increased incidence of Ebstein’s anomaly of the left-sided (systemic) atrioventricular (AV) valve, which is morphologically always a tricuspid valve and is related to a left-sided, morphologic right ventricle. In these patients, it is thought that the increased incidence of WPW is explained by the coexistence of Ebstein’s anomaly.4–6 Finally, WPW has perhaps been overdiagnosed in patients with hypertrophic cardiomyopathy (HCM) because of the pre-existing common QRS abnormality that may resemble pre-excitation. Specifically, some patients actually have a fasciculoventricular pathway of no clinical significance.9 However, SVT mediated by accessory pathways does occur in some patients with HCM; in these patients, SVT may be very poorly tolerated because of a coexisting hemodynamic abnormality.7 Specifically, Danon disease, a form of glyocogen storage disease, is associated with ventricular pre-excitation.10
Other Substrates for Tachycardia
Other arrhythmias are occasionally seen in patients with congenital heart disease who have not had surgical repair. Atrial flutter, when seen, usually is a complication of atrial dilation on hemodynamic grounds. For example, patients with Ebstein’s anomaly or other causes of severe tricuspid regurgitation may have atrial flutter, which, in turn, is most likely caused by right atrial dilation.3 Patients with mitral valve disease, and in particular mitral stenosis, are at risk for atrial fibrillation (AF), depending on their left atrial dilation. Patients with left ventricular failure and those with pulmonary hypertension, suprasystemic right ventricular pressure, or both may also have increased atrial pressures, with consequent atrial arrhythmias, and may also have ventricular ectopy, ventricular tachycardia (VT), or ventricular fibrillation (VF).
Atrioventricular Block
Certain groups of patients are predisposed to the development of complete AV block, independent of attempted surgical repair. First, patients who have L-transposition, also known as ventricular inversion or congenitally corrected transposition of the great vessels, are at risk for the development of complete AV block spontaneously throughout their lives. The yearly incidence has been estimated at approximately 2% per year.11 This is attributed to the abnormalities of the conduction system, in which malalignment exists between the atrial and the ventricular septa, and the normal compact AV node cannot make contact during embryologic development with the distal conducting system. A more anterior AV node forms instead and is thought to be more fragile.
Second, a syndrome of familial AV block associated with various septal defects has recently been described. Heterozygous mutations in NKX2.5, a homeobox transcription factor, lead to the spontaneous development of complete AV block, as well as associated cardiac defects, of which atrial septal defects (ASDs) are the most common.12 Ventricular septal defects (VSDs) may also be seen, particularly those associated with tetralogy of Fallot.
Arrhythmia Substrates Following Surgery for Congenital Heart Disease
Anatomic and Developmental Considerations
During cardiac embryologic development, the right atrium is derived from three sources13: (1) The primitive right atrium forms adjacent to the tricuspid annulus and gives rise to the heavily trabeculated right atrial free wall and right atrial appendage. (2) The sinus venosus is incorporated into the right atrium and provides the origin for the smooth-walled portion of the right atrium (sinus venarum) that exists between the cavae posterior to the primitive right atrial structures. (3) Finally, septation of the primitive common atrium is accomplished by the formation of the atrial septum from the septum primum and the septum secundum. The ostium secundum is a foramen that forms in the septum primum, which is subsequently closed by the septum secundum, which forms a flap over this ostium to create the foramen ovale. In the fetus, the foramen ovale provides a route for right atrial blood to cross to the left atrium. All along the junction between the primitive right atrium and the sinus venosus portion of the right atrium is the crista terminalis (“terminal crest”), which appears as a ridge along the inner surface of the right atrium. The crista terminalis runs superior to inferior along the lateral wall of the right atrium. At its superior edge, near the junction between the superior vena cava (SVC) and the right atrium, is the sinus node pacemaker complex. As it arches inferiorly toward the inferior vena cava (IVC), it gives rise to the eustachian valve ridge (EVR), which appears as more of a flap than a ridge. The EVR is a remnant of the primitive right sinoatrial valve, guarding the ostium between the sinus venosus and the primitive right atrium. The EVR runs anterior to the IVC orifice and posterior to the posterior portion of the tricuspid valve annulus. As such, in the fetal circulation, the EVR acts to direct the IVC flow away from the tricuspid annulus and toward the foramen ovale. As the EVR arches toward the inferior atrial septum, it passes just superior to the ostium of the coronary sinus. It joins with the valve of the coronary sinus to form the tendon of Todaro, which inserts on the atrial septum near the bundle of His. With the coronary sinus ostium and the tricuspid annulus, the tendon of Todaro forms the triangle of Koch, and at the apex of this triangle, the compact AV node is found.
Adult-Type Atrial Flutter
Classic atrial flutter is characterized by atrial rates of up to 300 beats/min, with typical and very characteristic saw-tooth flutter waves visible on the surface electrocardiogram (ECG). This suggests the presence of nearly continuous atrial electrical activity because of the relative lack of a long atrial isoelectric interval in most patients. In typical atrial flutter, the flutter waves are prominent and are negative in leads II, III, and AVF, suggesting inferior to superior atrial activation. Although initially thought to represent re-entry around the caval veins or around the tricuspid valve annulus, the work of multiple investigators has clearly established the actual circuit. Impulses emerge from an isthmus of atrial tissue between the IVC and the tricuspid annulus to spread up the atrial septum, activating the atrium at the site where the bundle of His is recorded, and then down the right atrial free wall to enter the isthmus again.14 This counterclockwise activation has been categorized as typical atrial flutter, whereas atrial flutter that uses the same circuit but in the clockwise order of activation has been categorized as reverse typical atrial flutter.15
In addition, further details have been provided using techniques of entrainment pacing, which depend on the demonstration of equivalence of the postpacing interval (PPI) during entrainment and the tachycardia cycle length (TCL) to establish that any given site is in the circuit (PPI = TCL). These studies have demonstrated the importance of the crista terminalis and the EVR as sites of conduction block during atrial flutter.16–18 Conduction block is suggested by the demonstration that sites along the ridge where double potentials can be recorded are present.17–20 The importance of such areas of conduction block is strengthened by entrainment pacing, demonstrating that the atrial myocardium on one or another side of the line of conduction block is part of the circuit. The critical nature of these lines of block is proved by RF ablation lesions that are designed to bridge from one line of block to another, with resultant abolition of the atrial flutter.21–23 These criteria have been satisfied with both typical and reverse typical atrial flutter, and the features of this arrhythmia circuit now seem well characterized.
As previously described, the wave of activation leaves the region of the tricuspid valve–IVC isthmus to climb the interatrial septum and enter the heavily trabeculated right atrial free wall in the region of the SVC. It then spreads down the right atrial free wall, with the crista terminalis behind and the tricuspid annulus in front, turning counterclockwise around the tricuspid annulus when viewed from below (left anterior oblique view fluoroscopically). As the wave of activation turns posterior, it enters a “funnel,” as described by Nakagawa and colleagues, created because the distance between the crista and the tricuspid annulus becomes progressively shorter.18 The wave is funneled to the isthmus between the IVC and the tricuspid valve annulus, now with the annulus anterior and inferior and the EVR (the extension of the crista terminalis) posterior and superior. It is important to note that at this site, the EVR bisects the isthmus between the IVC and the tricuspid valve and that it is the EVR, not the IVC, that provides the critical site of conduction block. As it enters the interatrial septum, the wave again spreads in a superior fashion along the septum for the next circuit. Atrial flutter can be effectively dealt with using RF ablation either at the septal site of EVR insertion, by lesions that bridge from the tricuspid valve to the EVR, or posterior, from the tricuspid annulus down to the IVC.18,21
Simple Atriotomy-Based Atrial Flutter
When performing surgery to repair a simple secundum ASD, the surgeon typically places a long incision in the right atrial free wall, which is oblique and runs from the right atrial appendage laterally down toward, but not to, the tricuspid annulus or the IVC. Care is taken to avoid the sinus node, and this concern results in the crista terminalis typically not being incised by the surgeon. This incision gives adequate exposure for the repair of ASDs and is also used for the atrial approach to repair VSDs, either alone or as part of tetralogy of Fallot and related defects. The ASD itself is commonly closed by using sutures, but for large defects a patch may be employed. Occasionally, a patent foramen is left open. The occurrence of atrial flutter in patients with repaired tetralogy of Fallot deserves special mention; in some series, it is at least as common as VT, and the presence of right bundle branch block (RBBB) may lead to its being confused with VT.24
This surgical approach clearly creates a long line of permanent conduction block that is entirely in the trabeculated right atrium, anterior to the crista terminalis. This anatomy potentially creates a tunnel of atrial tissue between the crista and the atriotomy and another between the atriotomy and the tricuspid annulus. Such tunnels can easily be imagined as the required protected zones of conduction, mediating IART. Numerous cases of patients who exhibit “incisional” IART in which the atriotomy seems to act as the critical barrier have been reported.25 In such patients, RF application from the atriotomy to the IVC, the tricuspid annulus, or the SVC has been successful in terminating tachycardia and preventing re-induction.
Because the atrial structures that support typical atrial flutter are also present and because these patients often have other risk factors for the development of flutter (atrial dilation, fibrosis, etc.), they may also have typical atrial flutter postoperatively. Furthermore, as has become apparent in patients with otherwise structurally normal hearts, such IART and flutter circuits can run in either direction (counterclockwise or clockwise). Indeed, several series reported a more common occurrence of typical or reverse typical atrial flutter than true incisional flutter in these patients.26,27 In patients who have undergone patch closure of a secundum ASD, the patch itself has been reported to be a possible site of conduction block, mediating tachycardia, although this is less common.21 The potential variability in circuits and rotation creates the possibility for several distinct P-wave morphologies and AT cycle lengths.
Intra-atrial Re-entry Following Atrial Repair of Transposition
The Senning and Mustard procedures, which are similar operations to address the hemodynamic abnormality in transposition, direct systemic return to the left ventricle and pulmonary artery and the pulmonary venous return to the right ventricle and aorta.28,29Although very successful, these operations are rarely performed in the current era, in part because of the success of the arterial switch procedure and in part because of the high incidence of sinus node dysfunction, atrial arrhythmias, and increased risk of sudden cardiac death (SCD). However, interest has recently increased in the so-called double switch procedure as a strategy for managing patients with L-transposition (congenitally corrected transposition). A Senning atrial baffle is constructed in this procedure.30 Thus, this surgical substrate may not, in fact, disappear. In the Mustard procedure, after a long atriotomy anterior to the crista terminalis and the resection of the atrial septum, a baffle is constructed and sewn into place around each caval vein, through the isthmus between the IVC and the tricuspid annulus, and to the posterior wall of the left atrium so that caval flow is direct to the mitral annulus.29 Pulmonary venous flow travels around the baffle and finds the tricuspid annulus. It is important to note that the baffle, where it is sewn into place along the tricuspid annulus, has the same function as the EVR in fetal life, which is to prevent IVC flow from reaching the tricuspid valve. Furthermore, surgical technique is directed at avoiding injury to the sinus node, so the crista terminalis is not disturbed. Finally, various approaches are used to avoid AV block, and often these lead to the coronary sinus being incorporated into the pulmonary venous atrium rather than the systemic venous atrium.31 These details leave the entire right atriotomy as well as the isthmus of atrial tissue between the EVR and tricuspid annulus in the new pulmonary venous atrium.32,33 The one exception is the situation where the coronary sinus drainage is the systemic venous atrium, in which a catheter can reach the flutter isthmus from the IVC.32
In most respects, the Senning procedure is similar electrophysiologically to the Mustard procedure. The Senning procedure was designed to use mostly atrial tissue versus artificial material to construct the baffle.28 In order to accomplish this, two atrial incisions are made. The first is in the right atrium, longitudinal, parallel, and anterior to the crista terminalis. The second, in the left atrium, is parallel to the first and between the right pulmonary veins and the interatrial septum. A U-shaped incision is made in the atrial septum, just above the coronary sinus, leaving the flutter isthmus intact. This flap of atrial septum is sewn to the back of the left atrium, to the left of the left pulmonary veins. The flap of right atrial free wall is sewn into place near or at the site of the EVR, preventing IVC flow from crossing the tricuspid valve. The left atrial incision is closed by sewing to the other edge of the right atrial incision. As in the Mustard procedure, both the flutter isthmus and the right atriotomy are part of the new pulmonary venous atrium.
Intra-atrial Re-entry Following the Fontan Procedure
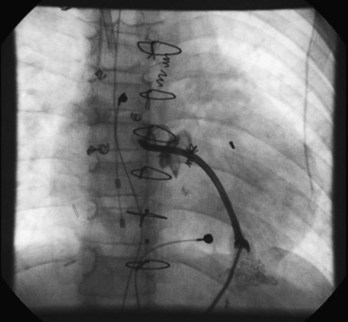
The Fontan procedure has changed many times since its development as a palliative procedure for patients without two functional ventricles, as a way of relieving ventricular volume overload and of normalizing arterial saturations.34 Initially, it was thought that the right atrium could be used as an effective pumping chamber, provided that pulmonary artery pressures were low (atriopulmonary connection). Largely as a result of an extremely high incidence of atrial arrhythmias after such procedures, as well as concerns about hydraulic energy loss in the system and pulmonary venous obstruction, this approach has been abandoned in favor of approaches that bypass the heart entirely (total cavopulmonary connection via the lateral tunnel or via an extracardiac conduit).35–37 Within each of the two categories, many modifications exist. Despite the approach of total cavopulmonary connection, atrial arrhythmias continue to be observed, although some large series now report a lower incidence of arrhythmias with the external conduit Fontan when compared with the lateral tunnel.38 In any case, surgical details are critical in planning mapping and ablation procedures in these patients. In particular, difficulties in access are common and may limit the number of catheters that can be placed in the heart. Novel approaches such as the direct transthoracic approach are reported (Figure 78-1), and one may also consider perforating the baffle or approaching the atrial mass via the SVC and the left pulmonary artery.39–41
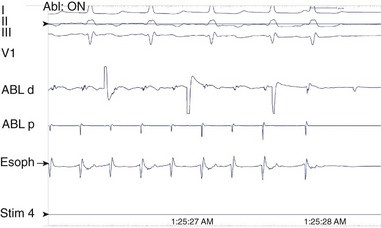
A long atriotomy is placed with the various forms of atriopulmonary connection. In patients who had a conduit from the right atrium to the pulmonary artery and in those in whom the right atrial appendage was connected directly to the pulmonary artery or to the right ventricular outflow tract (RVOT; the Bjork modification), this atriotomy was anterior to the crista terminalis. Often in these patients, patch augmentation of the right atrium was performed, using a piece of pericardium or other material incorporated into the closure. Invariably, closure of a large ASD was also necessary. As in the simpler situation of ASD repair (see earlier), both typical atrial flutter and incisional re-entry around the anterior atriotomy are possible and have been observed. Triedman and colleagues demonstrated slow conduction up the lateral wall in their excellent multiple-site mapping studies using basket catheters42; and this configuration fits the concept of conduction in a long isthmus bounded by the atriotomy and the crista terminalis. Re-entry around the ASD patch is also possible. Finally, patch closure of the tricuspid annulus has been occasionally performed in patients with a single ventricle without tricuspid atresia, potentially creating areas of slow atrial conduction on the other side of the suture line. Entrainment pacing is useful in identifying areas that are in the circuit and might be potential targets for ablation (Figures 78-2 and 78-3).43 Alternatively, the use of dense voltage maps has been reported to be of value in identifying areas of scar and corridors of low-voltage myocardium, which can be targeted for ablation lesions, with some success.44
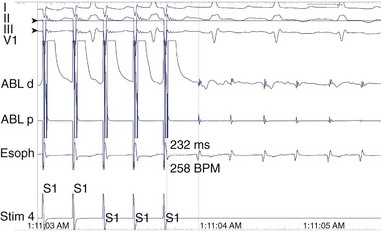
FIGURE 78-2 Surface and intracardiac electrogram tracings during the electrophysiology study in the patient shown in Figure 78-1. Atrial tachycardia is entrained via pacing through the ablation catheter at a candidate site, and the postpacing interval is equivalent to the tachycardia cycle length, demonstrating that this site is in the atrial tachycardia circuit.
Ventricular Tachycardia Following Repair of Tetralogy of Fallot
VT continues to be a difficult problem in the management of patients who have undergone surgical repair of tetralogy of Fallot and other related congenital heart defects. The actual etiology of SCD in patients following tetralogy repair is still somewhat uncertain. However, because of the frequent occurrence of premature ventricular contractions (PVCs), nonsustained and sustained VT in patients who have undergone complete repair of tetralogy of Fallot and related defects such as double-outlet right ventricle, VT has been implicated in the etiology of SCD in this patient group.45–51 It is known that postoperative tetralogy of Fallot is the single most common condition that causes SCD among children between the ages of 1 and 16 years.52
Most of the available information concerning patients with VT and congenital heart disease pertains to tetralogy of Fallot compared with other forms of congenital heart disease. Ventricular arrhythmias do occur but are much rarer in patients with other lesions.53 For the purposes of management, tetralogy of Fallot can be viewed as an archetype for other lesions when patients with other lesions present with ventricular arrhythmias in the setting of ventriculotomy, right ventricular dysfunction, or both.
Patients with tetralogy of Fallot prior to repair have a large VSD with (usually severe) RVOT obstruction, which leads to cyanosis. The placement of a systemic-to-pulmonary artery shunt as a palliative procedure adds the element of potential left ventricular volume overload. Correction of the defect involves patch closure of the VSD with relief of the right ventricular obstruction. In nearly all patients, this requires resection of a significant amount of right ventricular muscle. Early in the experience, this was not done via an atriotomy with retraction of the tricuspid valve but, instead, required a ventriculotomy. Finally, in tetralogy of Fallot, the pulmonary annulus is typically smaller than normal. This has been approached by the placement of a transannular patch, which leads to chronic pulmonic insufficiency. Pulmonic insufficiency may be very severe if it is associated with downstream obstruction related to significant pulmonary arterial stenosis. It has been hypothesized that ventricular arrhythmias are caused by the effects of years of chronic cyanosis, followed by the placement of a ventriculotomy, with elevation of right ventricular pressures arising from inadequate relief of obstruction, and severe pulmonic regurgitation with right ventricular dysfunction and enlargement.45,54–56 Factors such as wall stress and chronic cyanosis, coupled with the passage of time, may lead to myocardial fibrosis and result in the substrate for re-entrant ventricular arrhythmias. This hypothesis is supported by histologic examination of the hearts of patients with tetralogy of Fallot who died suddenly. These studies have shown extensive fibrosis.57 The hypothesis is also supported by the observation of fractionated electrograms and late potentials recorded from the right ventricle at EPS, suggesting the presence of slow conduction.58,59 Despite the presence of a 5% incidence of coronary artery abnormalities in tetralogy of Fallot that put the left anterior descending coronary artery or other large branches at risk at the time of complete repair, such potential damage has not been implicated in the etiology of ventricular arrhythmias or of SCD in most patients.
Careful studies in patients with VT following surgery for tetralogy, as reported by Zeppenfeld et al, have supported the concept that the mechanism of VT is macro–re-entry, which involves a limited number of critical isthmuses in the right ventricle, either at the site of anterior right ventriculotomy or related to the site of a VSD patch.60 Transient entrainment has been documented, with constant fusion at the paced cycle length and progressive fusion at decreasing cycle lengths.61,62 Successful ablation of these isthmuses is effective in preventing the recurrence of VT.
Early reports noted the frequent occurrence of PVCs in patients who had previously undergone repair of tetralogy of Fallot. Gillette and coworkers identified PVCs on routine ECGs in 18% of patients.45 With exercise testing, the incidence may increase—to around 20%, as shown in one study.50 With Holter monitoring, the incidence of ventricular ectopy is reported to be as high as 48%.63 In about half of these patients, ventricular ectopy is complex, defined as multiform beats, couplets, or VT. In the great majority of patients, this ventricular ectopy is entirely asymptomatic.
Many investigators have tried to correlate the incidence of ventricular ectopy with various factors, including age at presentation, age at time of repair, and various hemodynamic features. Four factors seem to be the most important: (1) age at initial repair; (2) time since repair; (3) presence of residual right ventricular obstruction; and (4) presence of significant pulmonic insufficiency. In Chandar and colleagues’ multicenter study, older age at time of repair, especially beyond 10 years of age, was associated with nearly a 100% incidence of ventricular arrhythmias, regardless of the follow-up interval.63 In the same study, time since repair also predicted the occurrence of ventricular ectopy, which occurred in all 4 patients followed up for more than 16 years, despite repair in infancy. Walsh and colleagues, however, showed that in a group of patients who underwent repair at less than 18 months of age, ventricular ectopy was rare on ECG (1%) but more common on Holter monitoring (31%) after an average of 5 years of follow-up.64
Garson and associates, in a study of 488 patients with repaired tetralogy of Fallot, showed that the incidence of ventricular arrhythmias was closely related to right ventricular hemodynamics.65 The incidence of ventricular arrhythmias was significantly higher in those with a right ventricular systolic pressure greater than 60 mm Hg, and in those with a right ventricular end-diastolic pressure greater than 8 mm Hg, suggesting that residual RVOT obstruction and pulmonic insufficiency negatively influence outcome. They also found a relationship to age at surgery, but this was not as important as the follow-up interval. Zahka and coworkers, in a prospective study of 59 patients with tetralogy of Fallot repaired prior to 11 years of age, found that the degree of pulmonary regurgitation was, by far, the most important predictor of the frequency and severity of spontaneously occurring ventricular arrhythmias.54 Although the degree of residual RVOT obstruction was not a predictor in this study, significant residual obstruction was rare in their study group.
Spontaneously occurring sustained VT is, in fact, fairly uncommon among patients with repaired tetralogy, despite the high incidence of ventricular ectopy. The best data in this regard come from the study by Harrison and associates that included patients with repaired tetralogy of Fallot attending an adult congenital heart disease clinic.66 Eighteen of 210 patients (8.6%) had either documented sustained VT, syncope, or near-syncope, with palpitations and inducible sustained monomorphic VT at EPS. VT was closely related to right ventricular hemodynamics, particularly RVOT aneurysms and pulmonic insufficiency. This finding is consistent with the earlier report by Zahka and coworkers, which emphasized the importance of pulmonic insufficiency as a risk factor for ventricular ectopy.54
The occasional but persistent observation of unexpected SCD in this group of patients with repaired congenital heart disease, along with the high incidence of spontaneously occurring ventricular arrhythmias, both simple and complex, has led to the hypothesis that SCD in such patients is caused by VT. During the 1970s and 1980s, it was standard practice to perform EPSs in a large proportion of patients who had undergone repair of tetralogy of Fallot or of related congenital defects. Antiarrhythmic drug therapy was often prescribed on the basis of the results of such studies. This approach has, for the most part, been abandoned because of the lack of strong evidence supporting the proposition that SCD can be prevented with this approach as well as worries about the proarrhythmic effect of the antiarrhythmic medications chosen for treatment. In a large multicenter retrospective review that used a variety of electrophysiological protocols, Chandar and colleagues reported the experience with 359 postoperative patients with tetralogy of Fallot who underwent invasive EPS.63 VT could be induced in 17% of patients but not in any patient who was asymptomatic and had a normal 24-hour ECG. Although late SCD occurred in five patients, none of these patients had inducible VT at EPS.
It is also interesting that the risk of VT can be assessed from QRS duration on the surface ECG. Gatzoulis and associates found that in a group of 48 well-studied postoperative patients with tetralogy of Fallot, those with a QRS duration greater than 180 ms had a greatly increased risk of spontaneous VT, SCD, or both.55 Similarly, Balaji and coworkers showed that a QRS duration greater than 180 ms predicts the finding of inducible sustained monomorphic VT at EPS.67 The cause of this relationship is almost certainly related to the tendency of chronic pulmonic insufficiency to cause right ventricular dilation with more severe right ventricular conduction delay, for which lengthening of the QRS is a marker.
Clear evidence that the risk of SCD may increase at late follow-up exists. In a careful study of 490 survivors of tetralogy of Fallot repair at a single center, Nollert and colleagues constructed actuarial survival curves out to 36 years following surgery.68 In this study, the yearly actuarial mortality rate during the first 25 years was 0.24% per year, but mortality increased dramatically after 25 years to 0.94% per year. Most deaths were from SCD. The mortality risk was also related to date of repair (highest before 1970), the degree of preoperative polycythemia (highest with hematocrit >48), and the use of an RVOT patch (highest with a patch). The last factor is most likely related to the presence of pulmonic insufficiency, as suggested earlier.
The close linkage of pulmonic insufficiency, right ventricular dilation, and VT/SCD suggests that the management of pulmonic insufficiency in patients with tetralogy of Fallot should be more aggressive. However, pulmonary valve placement is problematic in young children with small hearts because of issues related to growth, and valves predictably fail, necessitating reoperation. The earlier the first valve is placed, the more surgeries the patient must undergo over his or her lifetime. However, it is ideal to intervene with pulmonary valve replacement before the development of significant right ventricular dysfunction. While QRS duration is an easily obtained indicator of right ventricular dilation, attention has lately focused on the evaluation of right ventricular volume by cardiac magnetic resonance imaging (MRI). Therrien et al have observed that in adults with tetralogy of Fallot, a right ventricular end-diastolic volume of greater than 170 mL/m2 predicted a failure of the right ventricle to return to normal size following PVR, while all patients with volumes less than 170 mL/m2 had complete normalization.69 Geva has proposed a right ventricular end-diastolic volume of 160 mL/m2 or more as a criterion for PVR, among other criteria and has also recommended an earlier operation for patients with defects originally repaired at age 3 years or later.70 It remains to be seen whether the widespread adoption of these protocols will have an effect on the incidence rates of SCD.71
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree