Physical Examination
Kanu Chatterjee
Overview
A systematic approach to the bedside examination of a patient is essential in determining the significance of an abnormal physical finding, such as decreased or increased intensity of the first heart sound (S1), a pathologic third heart sound (S3), or a systolic or diastolic murmur. Assessment of the abnormalities of the systemic venous pressure and pulse, arterial pulse, precordial impulse, heart sound, and murmurs provides clues not only for the diagnosis of the anatomic abnormalities, but also for the determination of the severity of the hemodynamic abnormalities.
Furthermore, a careful, systematic, and methodical bedside clinical evaluation—including analysis of clinical history—can provide enough information to decide on appropriate further investigations to establish the diagnosis.
Inspection
General
During general inspection (1,2), the stature, body habitus, and presence or absence of obesity are noted. An unusually short stature is seen in patients with osteogenesis imperfecta, which is associated with aortic and mitral regurgitation and calcification of the arterial system. Congenital heart disease, such as Noonan syndrome, occurs in association with short stature, web neck, dental malocclusion, antimongoloid slanting of the eyes, mental retardation, and hypogonadism. Pulmonary valve stenosis and obstructive and nonobstructive cardiomyopathy are also observed. Dwarfism is an essential phenotypic feature of the various types of mucopolysaccharidosis, which can be associated with various valvular and myocardial dysfunctions. An unusually tall stature is seen in patients with Marfan syndrome, which is associated with aortic aneurysm, aortic regurgitation, and mitral regurgitation. When severe chest or back pain is the presenting symptom in a patient with Marfan syndrome, acute aortic dissection should be suspected. A tall stature, long extremities, and eunuchoid appearance is observed in patients with Klinefelter syndrome, which can be associated with congenital heart diseases such as ventricular septal defects, patent ductus arteriosus, and tetralogy of Fallot. During initial cardiac evaluation, it is customary to notice any obvious musculoskeletal deformity, such as kyphoscoliosis, straight back, and pectus deformity. A higher incidence of mitral valve prolapse is observed in the presence of such musculoskeletal deformities. An increased incidence of mitral valve prolapse is also observed in females with smaller breasts. Muscular dystrophies such as
pseudohypertrophic, fascioscapular humoral, or limb girdle varieties can be suspected during general inspection. These muscular dystrophies may be associated with various cardiovascular abnormalities, including myocardial disease, mitral valve prolapse, ventricular septal defect, and dysrhythmias.
pseudohypertrophic, fascioscapular humoral, or limb girdle varieties can be suspected during general inspection. These muscular dystrophies may be associated with various cardiovascular abnormalities, including myocardial disease, mitral valve prolapse, ventricular septal defect, and dysrhythmias.
Gait
Neurologic deficits resulting from cardioembolic strokes or hypertensive cerebrovascular disease may be associated with abnormalities of gait. A parkinsonian gait may indicate Shy-Drager syndrome, which may be associated with orthostatic hypotension and supine hypertension (3). Certain metabolic disorders (e.g., hyperthyroidism, hypothyroidism, Cushing syndrome, and acromegaly) can be suspected during inspection, and these metabolic diseases may be associated with various cardiovascular abnormalities, including systemic and pulmonary hypertension and myocardial and pericardial diseases.
Body Habitus
Marked weight loss, wasting, and cachexia are manifestations of severe, chronic congestive heart failure (CHF). Cardiac cachexia (4) does not correlate with cardiac output as usually perceived, but to a marked neuroendocrine abnormality characterized by an activated renin–angiotensin system and increased levels of cytokines, including tumor necrosis factor and interleukins. Furthermore, cardiac cachexia is associated with poor prognosis. The presence and severity of obesity should be noted; various cardiovascular abnormalities can be associated with obesity. Obesity, particularly abdominal and truncal types, is one of the components of the metabolic syndrome X, which comprises hypertension, hyperlipidemia, and insulin resistance (5,6). In patients with metabolic syndrome, there is increased risk of atherosclerotic macrovascular disease, including coronary artery disease. In markedly obese patients, there is increased incidence of sleep apnea, hypertension, and dilated cardiomyopathy. Truncal obesity, if associated with buffalo hump or moon facies, should raise the suspicion of Cushing syndrome, which may be complicated by hypertension and hypertensive heart disease. In African Americans, severe obesity and marked increase in body mass index and diabetes are more important predictors of primary diastolic heart failure than hypertension or coronary artery disease.
Respiration
During inspection, presence of respiratory distress and types of altered respiration should be observed. Inability to lie down or a dry, irritating cough with dyspnea in the supine position usually indicates pulmonary venous congestion. Sleep-disordered breathing is common in chronic CHF (7). Typical Cheyne-Stokes respiration with central sleep apnea is observed in 20% to 30% of patients with moderate or severe systolic CHF. Cheyne-Stokes respiration with central sleep apnea is a risk factor for increased mortality (8). Also, patients with central sleep apnea are likely to benefit from continuous positive airway pressure therapy (9). Frequent sighing respirations and a restless, anxious look are more frequently encountered in patients with anxiety and neurocirculatory asthenia. The “blue bloater” and “pink puffer” appearances, which can be detected during inspection, usually indicate chronic obstructive pulmonary disease (COPD). During inspection, it is desirable to note any changes in the color of the skin. A bluish discoloration of the skin may indicate cyanosis, which can be either peripheral or central. Peripheral cyanosis is detected in exposed skin (e.g., lips, nose, and earlobes, and extremities) and indicates impaired peripheral perfusion. A bluish discoloration of the tongue, uvula, and buccal mucus membrane suggests central cyanosis, which results from intrapulmonary or intracardiac right-to-left shunt. When differential cyanosis (i.e., cyanosis in the inferior extremities without cyanosis in the superior extremity) is observed along with differential clubbing, Eisenmenger syndrome associated with patent ductus arteriosus should be suspected.
Edema
Generalized edema can be detected during inspection, which usually results from nephrotic syndrome and sepsis, and rarely from severe heart failure. Dependent edema, on the other hand, is associated with right heart failure. The presence of ascites, which also may be a manifestation of right heart failure, can be suspected from the protuberant abdomen. Ascites in the absence of edema of the lower extremities is more frequently a manifestation of liver disease, such as cirrhosis, rather than of heart failure. However, in patients with constrictive pericarditis and restrictive cardiomyopathy, disproportionately large ascites with little or no edema of the lower extremities can be observed (10).
Skin
Slate or bronze pigmentation of the skin may suggest hemochromatosis, which may be associated with various cardiac complications (e.g., restrictive or dilated cardiomyopathy, arrhythmias, and conduction disturbances). It should be appreciated that patients on chronic amiodarone therapy develop similar discoloration of the skin with exposure to sunlight (11). Discoloration of the skin similar to marked suntan is also seen in patients with carcinoid syndrome. Mild jaundice may be observed in patients with heart failure with congestive hepatopathy. Prosthetic valve malfunction should be suspected if jaundice is obvious in a patient with artificial heart valves. The livido reticulares with cyanosis of the toes and preserved peripheral pulses (blue toes syndrome) suggest cholesterol emboli (12). Acrosclerosis with taut, thickened, or edematous skin bound tightly to subcutaneous tissue in the hands and fingers suggests systemic sclerosis, which may be associated with pulmonary hypertension, pericarditis, right heart failure, systemic hypertension, restrictive cardiomyopathy, and dilated cardiomyopathy. If malar flush is detected, mitral stenosis with pulmonary hypertension should be suspected. Malar flush, however, can also occur in patients with severe precapillary pulmonary hypertension and may be confused with butterfly rash of lupus erythematosus. Transverse or diagonal earlobe creases in a relatively young person (<45 years of age), along with corneal arcus—a circumferential light ream around the iris that begins inferiorly—is associated with increased risk of atherosclerotic coronary artery disease. Arcus is also frequently associated with hypercholesterolemia and xanthelasma (deposits of cholesterol on the eyelids). When telangiectasia of the lips, tongue, and buccal mucosa are detected, Osler-Weber-Rendu disease, which is associated with pulmonary arteriovenous fistulae, should be suspected. Exfoliative dermatitis, purpura, and petechial rashes may indicate drug reactions. Although an erythema marginatum is characteristic of acute rheumatic fever, erythema nodosum is a nonspecific finding and occurs in many systemic diseases. In patients with suspected bacterial endocarditis, searches should be made for conjunctival hemorrhages, skin purpuric, and petechial lesions, as well as for splinter hemorrhages. It should be realized that splinter hemorrhages in the nail beds may occur in normal patients after mild trauma and in patients with trichinosis or hemorrhagic disorders.
During examination of the skin, it is customary to check for the presence of cutaneous and subcutaneous nodular lesions, which may suggest some systemic and metabolic diseases. The rheumatic nodules associated with acute rheumatic fever are small, nontender, and most frequently are present on the
knuckles, extensor surface of the elbows, and suboccipital regions. Rheumatoid nodules, on the other hand, are large, nontender, and characteristically localized over points of pressure or friction—most commonly the extensor surfaces of the proximal forearms. If rheumatoid nodules are detected, attention should be directed to detect the cardiovascular complications of rheumatoid arthritis (e.g., pericarditis, aortic and mitral valvular disease, conduction disturbances, and, rarely, cardiomyopathy). Xanthomas—cholesterol-filled nodules—occur in different types of abnormalities of lipoprotein metabolism, and recognition of the distribution of xanthomas aids in the diagnosis of these disorders. Tendon xanthomas, xanthomas occurring on digital extensors, and tuberous xanthomas occur in familial hypercholesterolemia (13). Eruptive xanthomas are small, yellow papules in the skin surrounded by an erythematous halo. They occur in patients with primary familial hypertriglyceridemia that results from lipoprotein lipase deficiency and is associated with recurrent episodes of pancreatitis, but not with premature coronary artery disease. Eruptive xanthomas also occur in patients with endogenous and mixed hypertriglyceridemia, which can be associated with ischemic vascular disease. In patients with suspected bacterial endocarditis, it is desirable to search for Osler nodes and Janeway lesions. Osler nodes are most frequently observed on the palms, soles of the feet, and pads of the fingers or toes. These nodes are tender, nodular, erythematous skin lesions and result from emboli. Janeway lesions are nontender, raised, hemorrhagic nodules that usually occur on the palms of the hands and soles of the feet. These lesions were initially thought to be due to vasculitis, but they also appear to be of embolic origin. Café-au-lait spots—sometimes detected only in the axilla—occur in neurofibromatosis, which occasionally is associated with hypertrophic cardiomyopathy.
knuckles, extensor surface of the elbows, and suboccipital regions. Rheumatoid nodules, on the other hand, are large, nontender, and characteristically localized over points of pressure or friction—most commonly the extensor surfaces of the proximal forearms. If rheumatoid nodules are detected, attention should be directed to detect the cardiovascular complications of rheumatoid arthritis (e.g., pericarditis, aortic and mitral valvular disease, conduction disturbances, and, rarely, cardiomyopathy). Xanthomas—cholesterol-filled nodules—occur in different types of abnormalities of lipoprotein metabolism, and recognition of the distribution of xanthomas aids in the diagnosis of these disorders. Tendon xanthomas, xanthomas occurring on digital extensors, and tuberous xanthomas occur in familial hypercholesterolemia (13). Eruptive xanthomas are small, yellow papules in the skin surrounded by an erythematous halo. They occur in patients with primary familial hypertriglyceridemia that results from lipoprotein lipase deficiency and is associated with recurrent episodes of pancreatitis, but not with premature coronary artery disease. Eruptive xanthomas also occur in patients with endogenous and mixed hypertriglyceridemia, which can be associated with ischemic vascular disease. In patients with suspected bacterial endocarditis, it is desirable to search for Osler nodes and Janeway lesions. Osler nodes are most frequently observed on the palms, soles of the feet, and pads of the fingers or toes. These nodes are tender, nodular, erythematous skin lesions and result from emboli. Janeway lesions are nontender, raised, hemorrhagic nodules that usually occur on the palms of the hands and soles of the feet. These lesions were initially thought to be due to vasculitis, but they also appear to be of embolic origin. Café-au-lait spots—sometimes detected only in the axilla—occur in neurofibromatosis, which occasionally is associated with hypertrophic cardiomyopathy.
Funduscopy
In patients with established or suspected systemic hypertension, funduscopic examination should be performed. Several grades of changes may be observed (14). Grade 1 consists of minimal irregularity of the arterial lumen and narrowing with increased light reflex. Grade 2 changes consist of arteriovenous nicking, more marked narrowing and irregularity of the arterioles, and distention of the veins. Grade 3 changes are characterized by the presence of flame-shaped hemorrhages and fluffy “cotton wool” exudates in addition to arterial changes. Hard exudates may also be present. Grade 4 funduscopic changes are characterized by the presence of papilledema and any other changes in grades 1 through 3. Generally, grade 1 and 2 changes are present in benign hypertension, whereas grade 3 or 4 changes are seen in accelerated or malignant hypertension. Funduscopic examination should also be performed in patients with suspected bacterial endocarditis and may reveal vascular occlusions and hemorrhagic areas with white centers (Roth spots), which result from emboli in the nerve fiber retinal layer. Roth spots, however, are not diagnostic of bacterial endocarditis and can be seen in hemorrhagic disorders, including leukemia. Mycotic aneurysms resulting from large emboli occasionally may be discovered in retinal vessels during funduscopic examination. Identification of such lesions should lead to further evaluation for the source of emboli, and appropriate investigations—including transthoracic and, occasionally, transesophageal echocardiography—must be undertaken. Occasionally, funduscopic examination may reveal unusual findings such as beading of the retinal artery (hypercholesterolemia), microinfarction in the peripheral retina (sickle cell disease), or angioid streaks (pseudoxanthoma elasticum). These disorders may be associated with cardiovascular complications. A wreath-like arteriovenous anastomosis around the optic disk is a characteristic of Takayasu syndrome. In adult cardiology practice, the abnormal findings that are often detected during inspection, along with their significance, are summarized in Table 16.1.
TABLE 16.1 Inspection | ||||||
|---|---|---|---|---|---|---|
|
Examination of the Arterial Pulse
During initial evaluation, all accessible arterial pulses should be examined; in the inferior extremities, dorsales pedes, posterior tibials, and femoral pulses should be examined bilaterally. In the upper extremities, both brachial and radial pulses should be examined, and, in special circumstances, the ulnar pulses and axillary arterial pulses should also be examined. Temporal arteries are examined when temporal arteritis is suspected in patients with headache and jaw claudication. Carotid arteries should be examined sequentially in all patients. Diminished or absent dorsalis pedis pulses is associated with increased incidence of atherosclerotic coronary artery disease. Loss of or decreased femoral pulse unilaterally or bilaterally most frequently suggests local obstructive lesions due to atherosclerotic disease. However, diminished amplitudes of the lower extremity pulses, including femoral, popliteals, posterior tibials, and dorsales pedes arterial pulsations, also occur from isolated aortoiliac diseases, such as Leriche syndrome (15), postsubclavian coarctation of the aorta, aortic dissection, descending thoracic and abdominal aortic aneurysms, and abdominal aortic disease such as giant cell arteritis. When coarctation of the
aorta is suspected, the radial and femoral pulses should be examined simultaneously to assess radial/femoral delay.
aorta is suspected, the radial and femoral pulses should be examined simultaneously to assess radial/femoral delay.
In a normal adult, the pulse transmission time from aorta to the radial artery is approximately 75 ms and to the femoral artery is approximately 70 ms. Thus, in the presence of coarctation, delay in the onset of femoral pulse compared to that of radial pulse can be detected. The radial/femoral delay is rarely observed in patients with Leriche syndrome and abdominal coarctation. Also in pseudocoarctation, the degree of obstruction is not severe enough to decrease the amplitude of the femoral pulse or to cause delay in the onset of femoral pulse compared with that of radial pulse (16). In patients with radial/femoral delay and suspected coarctation, blood pressure should be recorded in the upper and lower extremities. In the supine position, the inferior extremity pressure is normally slightly higher than that of the upper extremity arterial pressure. In coarctation, inferior extremity pressure is lower than upper extremity pressure.
While examining the peripheral arterial pulses, it is desirable to assess the rigidity and elasticity of the arteries. The rigidity of the arterial pulses are best appreciated by examining the femoral, radial, brachial, and carotid pulses. In clinical practice, the thickness and firmness of the arterial walls are examined by rolling the vessel, usually the radial artery, against underlying tissue. The more rigid the artery, the less it is compressible. The appreciation of nonelastic, rigid peripheral arteries may indicate the presence of systolic hypertension. If the significant rigidity of the peripheral arteries is observed, it is desirable to perform Osler maneuver. Osler maneuver is performed by elevating the cuff pressure to obliterate the radial pulse; if, after obliteration of the pulse, the radial artery is easily palpable and appears rigid (positive Osler sign), then there might be a significant difference between indirect measurement of arterial pressure by cuff method and directly determined intraarterial pressure.
The peripheral arterial pulses are also examined at the bedside for detection of arrhythmias. If the pulse rate is regular but slow, sinus bradycardia, junctional rhythm, or complete atrioventricular block is suspected. Occasionally, bigeminy may produce an irregular slow pulse because of the nonconducted pulse associated with the ectopic beat. Careful examination of the venous pulse and simultaneous auscultation may be helpful for the differential diagnosis of slow regular pulse. If regular cannon waves with each cardiac cycle are recognized, junctional rhythm is suspected. In the presence of a slow regular pulse, if irregular cannon waves and changing intensity of the S1 are observed, atrioventricular dissociation owing to complete atrioventricular block is the most likely diagnosis. Bigeminy can be diagnosed easily by auscultation, which demonstrates the postectopic compensatory pause. Atrial fibrillation can be suspected if irregularly irregular pulses are appreciated. However, frequent premature beats or multifocal atrial tachycardia can also produce an irregularly irregular pulse. If atrial fibrillation is suspected, it is desirable to determine the ventricular rate by simultaneous auscultation to assess the degree of pulse deficit. The difference between the heart rate by auscultation and the pulse rate is the pulse deficit. The rapid ventricular responses are the cause of the hemodynamic abnormalities of atrial fibrillation. A fast and regular pulse rate (≥150 beats) should raise the possibility of supraventricular or ventricular tachycardia. It is mandatory to do electrocardiographic evaluation of every patient with suspected arrhythmia.
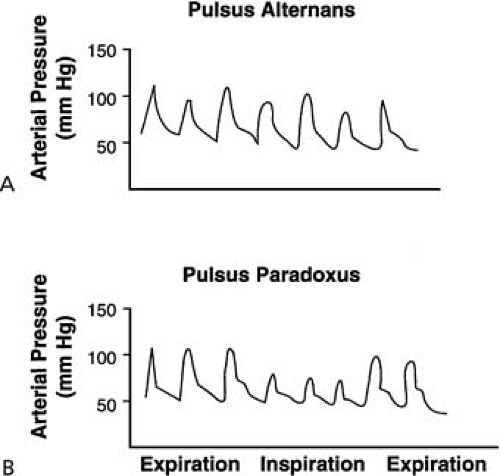
Peripheral arterial pulses are also examined to detect any alteration of the character of the pulse, which can provide important diagnostic clues. Pulsus alternans is suspected when strong and weak amplitude pulses are appreciated with alternate beats in the presence of a regular pulse. Pulsus alternans can be confirmed by measuring blood pressure by sphygmomanometer. When the cuff pressure is slowly released, phase I Korotkoff sound is heard initially only during the alternate strong beats; with further release of cuff pressure, the softer sounds of the weak beat also appear. The most important cause of pulsus alternans is left ventricular systolic failure. Pulsus alternans is rarely encountered in patients with cardiac tamponade with pulsus paradoxus, if the respiratory rate is half of the heart rate. However, in these patients, when respiration is held transiently, the pulsus alternans is resolved. Pulsus alternans (mechanical alternans) can also occur occasionally in aortic stenosis, in hypertrophic obstructive cardiomyopathy, with sudden increase of afterload, with ischemia, with abrupt fall of preload, and with the onset of a tachyarrhythmia. Rapid atrial pacing may induce pulsus alternans in the presence of normal left ventricular systolic function. However, clinically detectable sustained pulsus alternans almost always indicates left ventricular systolic dysfunction.
In experimental and clinical studies, alternating changes in preload, afterload, and contractility have been suggested as potential mechanisms for mechanical alternans (17); ventricular dyssynchrony with alternating contraction of the interventricular septum, incomplete relaxation in the alternate beats, partial asystole of the left ventricle, alternating changes in action potential duration, and alternating amounts of Ca2+ sparks from the sarcoplasmic reticulum are the other proposed mechanisms (18). The failing heart is sensitive to altered afterload and resistance to left ventricular ejection. The increased arterial pressure associated with a strong beat increases the resistance to left ventricular ejection for the following beat, which is thus associated with decreased forward stroke volume and decreased arterial pressure. This reduction in arterial pressure lowers the resistance to left ventricular ejection, which allows increase in forward stroke volume and, therefore, increase in arterial pressure. These changes in arterial pressure, reflecting changes in the ejection impedance with alternate beats, can perpetuate pulsus alternans (Fig. 16.1).
A substantial reduction in the amplitude of the arterial pulse during inspiration may suggest pulsus paradoxus. Determination of arterial pressure by sphygmomanometry during inspiration and expiration should be performed to confirm. Systolic arterial pressure normally falls during inspiration; however, the
magnitude of fall in arterial pressure during inspiration usually does not exceed 8 to 12 mm Hg. A more marked inspiratory decrease in arterial pressure exceeding 12 to 15 mm Hg is regarded as pulsus paradoxus. When the cuff pressure is slowly released, the systolic pressure at expiration is noted. With further slow deflation of the cuff, the systolic pressure during inspiration can also be detected. The difference between the pressure during expiration and inspiration is the magnitude of the pulsus paradoxus. The abnormal pulsus paradoxus is an important physical finding of cardiac tamponade (19). The marked inspiratory decrease in arterial pressure in tamponade results from the marked inspiratory decline of left ventricular stroke volume due to a decreased end-diastolic volume. In cardiac tamponade during inspiration, there is an increase in venous return to the right atrium and the right ventricle. Because of increased intrapericardial pressure, the intraventricular septum shifts toward the left ventricle during inspiration, which decreases left ventricular preload. There is also an expected decrease in venous return to the left ventricle during inspiration because of increased pulmonary venous reservoir capacity during inspiration. In clinical practice, besides cardiac tamponade, pulsus paradoxus is observed in patients with COPD. It should be emphasized that pulsus paradoxus is a rare finding in patients with constrictive pericarditis. Pulsus paradoxus is rarely observed in pulmonary embolism, pregnancy, marked obesity, and partial obstruction of the superior vena cava. In patients with significant aortic regurgitation and atrial septal defect, pulsus paradoxus may not occur despite cardiac tamponade. In hypertrophic obstructive cardiomyopathy, arterial pressure occasionally increases during inspiration (reversed pulsus paradoxus) (20). The precise mechanism for this phenomenon is not clear.
magnitude of fall in arterial pressure during inspiration usually does not exceed 8 to 12 mm Hg. A more marked inspiratory decrease in arterial pressure exceeding 12 to 15 mm Hg is regarded as pulsus paradoxus. When the cuff pressure is slowly released, the systolic pressure at expiration is noted. With further slow deflation of the cuff, the systolic pressure during inspiration can also be detected. The difference between the pressure during expiration and inspiration is the magnitude of the pulsus paradoxus. The abnormal pulsus paradoxus is an important physical finding of cardiac tamponade (19). The marked inspiratory decrease in arterial pressure in tamponade results from the marked inspiratory decline of left ventricular stroke volume due to a decreased end-diastolic volume. In cardiac tamponade during inspiration, there is an increase in venous return to the right atrium and the right ventricle. Because of increased intrapericardial pressure, the intraventricular septum shifts toward the left ventricle during inspiration, which decreases left ventricular preload. There is also an expected decrease in venous return to the left ventricle during inspiration because of increased pulmonary venous reservoir capacity during inspiration. In clinical practice, besides cardiac tamponade, pulsus paradoxus is observed in patients with COPD. It should be emphasized that pulsus paradoxus is a rare finding in patients with constrictive pericarditis. Pulsus paradoxus is rarely observed in pulmonary embolism, pregnancy, marked obesity, and partial obstruction of the superior vena cava. In patients with significant aortic regurgitation and atrial septal defect, pulsus paradoxus may not occur despite cardiac tamponade. In hypertrophic obstructive cardiomyopathy, arterial pressure occasionally increases during inspiration (reversed pulsus paradoxus) (20). The precise mechanism for this phenomenon is not clear.
The amplitude of the peripheral arterial pulse may provide some information about stroke volume, systemic vascular resistance, and compliance of the arteries. A small-amplitude, rapid pulse usually indicates hypotension, reduced stroke volume, and increased systemic vascular resistance. A large-amplitude, arterial pulse suggests large stroke volume or decreased compliance. A large-volume, bounding pulse is noted after exercise; in high-output states such as chronic anemia, hyperthyroidism, and aortic regurgitation; and in patients with bradycardia (e.g., complete heart block). Decreased compliance and increased rigidity of the peripheral arteries may also increase the amplitude of the arterial pulse, as seen in elderly patients with systolic hypertension. In elderly patients, when the carotid pulse amplitude is decreased, local carotid disease or aortic stenosis should be suspected.
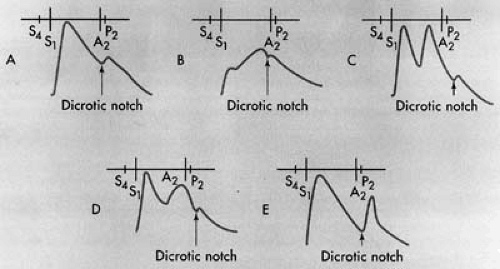
Changes in the contour of the arterial pulses (Fig. 16.2) should also be noted. In patients with significant aortic valve stenosis, a delayed upstroke of the ascending limb of the carotid pulse (so-called pulsus tardus), an anacrotic character of the carotid pulse, delayed peak, and small amplitude (pulsus parvus) of the carotid pulse are frequently appreciated. In patients with aortic stenosis, a thrill over the carotid pulse may also be detected. These abnormalities are best appreciated in the central pulse. In the central aortic ascending pressure pulse, normally there is a notch on the ascending limb: the anacrotic notch. On the upstroke of the carotid pulse, however, the anacrotic notch is not normally appreciated. An anacrotic carotid pulse gives the impression of interruption of the ascending limb or the upstroke of the carotid pulse. When the anacrotic notch is felt immediately after the onset of the upstroke, aortic stenosis is likely to be hemodynamically significant. An anacrotic radial pulse also suggests moderate to severe aortic stenosis. At the bedside, the delayed peak of the carotid pulse is appreciated by simultaneous auscultation of the duration of the systole. Normally, the peak of the carotid pulse is closer to the S1. In the presence of significant aortic stenosis, the peak of the carotid pulse is delayed and is closer to the second heart sound (S2). With increasing severity of aortic stenosis, the peak of the carotid pulse is not only further delayed and closer to the S2, but the amplitude of the carotid pulse is also substantially reduced. It needs to be emphasized, however, that these changes in the contour of the carotid pulse may not be present in elderly patients with aortic stenosis with decreased and noncompliant carotid arteries. When aortic stenosis is suspected, whether in young or older patients, an echocardiographic evaluation is highly desirable.
Pulsus bisferiens is appreciated by the presence of two positive impulses near the peak of the arterial pulse. These two positive impulses represent accentuated percussion and tidal waves, which can be recorded in the carotid arterial pulse tracing and the central aortic pulse waveform, even in normal subjects. The percussion wave results from the rapid left ventricular ejection, and a second, usually smaller, peak represents the tidal wave that results from a reflected wave from the periphery. Normally, radial and femoral pulse tracings demonstrate a single sharp peak. In pulsus bisferiens, percussion and tidal waves are accentuated. At the bedside, it is often difficult to distinguish between anacrotic, pulsus bisferiens, and dicrotic pulse. The anacrotic pulse, however, is characterized by a positive palpable wave during the ascending limb of the arterial pulse. Thus, it should be easily distinguished from pulsus bisferiens or the dicrotic pulse. However, if the two positive waves are felt near where the maximum amplitude of the pulse wave occurs, it is difficult to distinguish between pulsus bisferiens and dicrotic pulse. In these circumstances, one has to rely on detection of the etiologic conditions that can be associated with pulsus bisferiens or dicrotic pulse (21). The conditions most frequently associated with pulsus bisferiens are isolated hemodynamically significant aortic regurgitation, mixed aortic stenosis and regurgitation with predominant aortic regurgitation, and obstructive hypertrophic cardiomyopathy. Pulsus bisferiens is rarely appreciated in patients with large patent ductus arteriosus with large left-to-right shunt, multiple arteriovenous fistulas, and complete heart block. If these etiologies are excluded at the bedside, then one
should consider dicrotic pulse if double-picked arterial pulse is appreciated. The arterial pressure waveform, when recorded, reveals a single percussion wave and a prominent, accentuated dicrotic wave. At the bedside, however, it is difficult to differentiate between dicrotic pulse and pulsus bisferiens. The dicrotic pulse is appreciated in some patients with severe heart failure, for example, due to dilated cardiomyopathy, low cardiac output, and increased systemic vascular resistance. On the other hand, dicrotic pulse is also appreciated in patients with septic shock with high cardiac output and low systemic vascular resistance. However, a dicrotic pulse is also appreciated in patients after aortic valve replacement and usually indicates impaired left ventricular systolic function. In patients with aortic valve replacement, if a double-picked carotid pulse with sharp initial upstroke is appreciated, it is desirable to evaluate prosthetic valve and left ventricular function by echocardiography to exclude paraprosthetic leak.
should consider dicrotic pulse if double-picked arterial pulse is appreciated. The arterial pressure waveform, when recorded, reveals a single percussion wave and a prominent, accentuated dicrotic wave. At the bedside, however, it is difficult to differentiate between dicrotic pulse and pulsus bisferiens. The dicrotic pulse is appreciated in some patients with severe heart failure, for example, due to dilated cardiomyopathy, low cardiac output, and increased systemic vascular resistance. On the other hand, dicrotic pulse is also appreciated in patients with septic shock with high cardiac output and low systemic vascular resistance. However, a dicrotic pulse is also appreciated in patients after aortic valve replacement and usually indicates impaired left ventricular systolic function. In patients with aortic valve replacement, if a double-picked carotid pulse with sharp initial upstroke is appreciated, it is desirable to evaluate prosthetic valve and left ventricular function by echocardiography to exclude paraprosthetic leak.
TABLE 16.2 Altered Characters of the Arterial Pulse and their Clinical Significance | |
|---|---|
|
Corrigan pulse, or water-hammer pulse, is typically observed in patients with significant chronic aortic regurgitation and is characterized by an abrupt, very rapid upstroke of the peripheral pulse followed by rapid collapse. It is best appreciated by raising the arm abruptly and feeling the changes in the radial pulse. The significance of altered character of the arterial pulse in adult patients is summarized in Table 16.2.
Examination of Jugular Venous Pulse and Pressure
Examination of jugular venous pulse and pressure (1,2) is essential to assess hemodynamic changes in the right side of the heart. Jugular venous pressure and pulses usually are examined with the patient in a 45-degree, semirecumbent position. However, if, in this position, the venous pulsations are not recognized, the examination of venous pressure and pulse should be done with the patient in a supine position or even with the head and neck tilted below the level of the chest. Occasionally, if the venous pressure is normal or low, the legs need to be raised to increase the venous return and increase right-sided venous pressure. When the venous pressure is extremely elevated and the veins appear to be distended, it is preferable to examine the jugular venous pulses with the patient in an upright position or even in a standing position. It is preferable to examine the internal jugular venous pressure and pulse. In adults, particularly in elderly patients, the external jugular venous pressure may be elevated because of partial obstruction at the level of the external jugular venous bulb owing to partial thrombosis or even obstruction by the platysma muscle. The internal jugular venous pulse is located medial to the mandibular portion of the sternomastoid muscle. The proximal internal jugular venous pulse is located in the supraclavicular area between the two proximal heads of the sternomastoid muscle. Both right and left jugular venous pulsations need to be examined, because sometimes disparity between left and right internal jugular venous pressures can be recognized. Occasionally in elderly patients, the left internal jugular venous pressure is higher than the right internal jugular venous pressure because of the partial obstruction of the left innominate vein by unfolded aorta. In these circumstances, with inspiration and descent of the diaphragm, partial obstruction of the left innominate vein is relieved, and the pressures in the right and left internal jugular veins become equal.
At the bedside, venous pulsation needs to be differentiated from carotid artery pulsation. By inspection, the venous pulse is characterized by a sharp inward movement, whereas the arterial pulse is characterized by a sharp outward movement. During inspection, the venous pulse is also recognized by its double undulation character in sinus rhythm. In the presence of atrial fibrillation, the double undulation character of the venous pulse is lost because of the absence of an ‘a’ wave associated with atrial systole. The venous pressure and the amplitude of the venous pulse can be decreased or increased by appropriate maneuvers. The pressure and amplitude can be decreased by raising the level of the head and trunk above the level of the right atrium (in a sitting or standing position), or they can be increased by enhancing the venous return to the right side of the heart by raising the legs or by abdominal compression. In the presence of low systemic venous pressure, when a patient is examined in a head-down position, it is possible to recognize the venous pulsation in the neck. When a pulsation in the neck is recognized, a gentle to moderate compression—by the fingers or stethoscope tubes—at the root of the neck obliterates the venous pulse, but the arterial pulse remains visible. This maneuver is extremely useful for differentiating between arterial and venous pulsations. Normally during inspection, the jugular venous pulse amplitude decreases during inspiration, whereas the arterial pulse amplitude does not change during respiration.
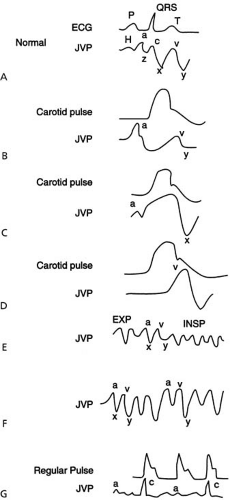
After recognizing the venous pulse, the venous pressure is estimated by noting the height of the oscillating top of the venous pulse above the sternal angle. Right atrial pressure is approximated by adding 5 cm to the height of the venous column, because it is assumed that the right atrium is located about 5 cm below the sternal angle. The normal right atrial pressure is less than 9 cm of water. If the jugular venous pressure is increased in the absence of obvious pulsation, it is desirable to exclude superior vena cava obstruction, which does not permit transmission of right atrial pulsation to the internal jugular veins. If pulsation of the neck veins can be recognized, the estimated venous pressure can be used to estimate right atrial pressure. The character of the venous pulse and other physical findings need to be incorporated to assess the cause of elevation of systemic venous and right atrial pressures. The normal jugular venous pulse wave or right atrial pressure wave recordings usually consist of three positive waves (‘a,’ ‘c,’ and ‘v’) and two negative waves (‘x’ and ‘y’ descents) (Fig. 16.3). The ‘a’ wave is caused by transmitted right atrial pressure to the jugular veins during right atrial systole. The ‘a’ wave in the jugular venous pulse is appreciated by its occurrence just prior to the left ventricular ejection, which is recognized by simultaneous palpation of the carotid pulse upstroke. At the bedside, simultaneous palpation of the carotid pulse upstroke and inspection of the venous pulse give the impression that the ‘a’ wave of the venous pulse and the carotid pulse upstroke occur out of phase. On the other hand, the prominent
‘v’ wave in the venous pulse occurs more or less simultaneously with the carotid pulse upstroke. Atrial relaxation initiates the descent of the ‘a’ wave. Rarely, when the PR interval is markedly prolonged, the descent may continue until a plateau is reached, the ‘z’ point, which occurs just prior to the ventricular systole. The descent after atrial systole usually is interrupted by the ‘c’ wave. In the right atrial pressure pulse, the ‘c’ wave is recognized with the onset of right ventricular systole and occurs from bulging of the tricuspid valve into the right atrium. It should be emphasized, however, that the ‘c’ wave in the jugular venous pulse probably results from transmission of the carotid artery pulsation and not from the transmission of the right atrial ‘c’ wave. The ‘v’ wave is caused by the rise in right atrial and jugular venous pressure due to continued inflow of blood to the venous system during right ventricular systole when the tricuspid valve is still closed. Although in right atrial and jugular venous pressure tracings, the peak of the normal ‘v’ wave occurs immediately after ventricular systole; at the bedside, the normal or abnormal ‘v’ wave coincides with the carotid pulse upstroke and downstroke. Although the regurgitant wave of tricuspid regurgitation (a prominent ‘v’ wave) occurs earlier and coincides with the beginning of left ventricular ejection, at the bedside the tricuspid regurgitant wave is appreciated simultaneously with the carotid pulse upstroke (see Fig. 16.3).
‘v’ wave in the venous pulse occurs more or less simultaneously with the carotid pulse upstroke. Atrial relaxation initiates the descent of the ‘a’ wave. Rarely, when the PR interval is markedly prolonged, the descent may continue until a plateau is reached, the ‘z’ point, which occurs just prior to the ventricular systole. The descent after atrial systole usually is interrupted by the ‘c’ wave. In the right atrial pressure pulse, the ‘c’ wave is recognized with the onset of right ventricular systole and occurs from bulging of the tricuspid valve into the right atrium. It should be emphasized, however, that the ‘c’ wave in the jugular venous pulse probably results from transmission of the carotid artery pulsation and not from the transmission of the right atrial ‘c’ wave. The ‘v’ wave is caused by the rise in right atrial and jugular venous pressure due to continued inflow of blood to the venous system during right ventricular systole when the tricuspid valve is still closed. Although in right atrial and jugular venous pressure tracings, the peak of the normal ‘v’ wave occurs immediately after ventricular systole; at the bedside, the normal or abnormal ‘v’ wave coincides with the carotid pulse upstroke and downstroke. Although the regurgitant wave of tricuspid regurgitation (a prominent ‘v’ wave) occurs earlier and coincides with the beginning of left ventricular ejection, at the bedside the tricuspid regurgitant wave is appreciated simultaneously with the carotid pulse upstroke (see Fig. 16.3).
The descending limb of the ‘v’ wave—the ‘y’ descent—is caused by the opening of the tricuspid valve and the rapid inflow of blood to the right ventricle from the right atrium. The ‘y’ descent is almost always recognized in the jugular venous pulse, and it follows the ‘v’ wave.
During examination of the jugular venous pulse at the bedside, if a prominent ‘a’ wave is appreciated, conditions associated with increased resistance to right atrial emptying during atrial systole should be considered. The ‘a’ waves need to be distinguished from regular cannon waves, which occur in junctional rhythm or ventricular tachycardia with retrograde ventriculoatrial conduction. Cannon waves occur during atrial systole with closed tricuspid valve. Cannon waves, therefore, occur concurrently with the onset of ventricular systole (i.e., concurrently with carotid pulse upstroke). Cannon waves are distinguished from ‘v’ waves by lack of obvious ‘y’ descents that follow ‘v’ waves or regurgitant waves. In the presence of a regular pulse, if irregular cannon waves are recognized by a sudden appearance of a large positive wave coincident with ventricular systole (with carotid upstroke), atrioventricular dissociation should be suspected. Regular cannon waves also
occur in patients with a prolonged PR interval when atrial systole occurs during the preceding ventricular systole. When a prominent ‘a’ wave with large amplitude is appreciated, it is desirable to exclude tricuspid valve obstruction; this can be done by noting absence of a mid-diastolic rumble along the lower left sternal border, which increases in intensity during inspiration. Although in severe tricuspid stenosis the ‘y’ descent is abbreviated and presystolic hepatic pulsations can be observed, such a degree of tricuspid stenosis is rarely encountered. It should also be emphasized that isolated tricuspid stenosis is almost never encountered in rheumatic heart disease, and rheumatic tricuspid stenosis occurs almost always in the presence of mitral or aortic valve disease and in patients who are usually in atrial fibrillation. In adult cardiac patients, if isolated tricuspid stenosis is recognized with a prominent ‘a’ wave with a mid-diastolic rumble along the lower left sternal border that increases in intensity during inspiration (Carvello sign), right atrial myxoma or carcinoid heart disease should be considered in the differential diagnosis. However, the most common cause of a prominent ‘a’ wave in adults is right ventricular hypertrophy, which offers increased resistance to right atrial emptying during right atrial systole. Right ventricular hypertrophy may result from a right ventricular outflow tract obstruction, such as pulmonary valve stenosis or precapillary or postcapillary pulmonary hypertension. Significant pulmonary valve stenosis can be easily diagnosed by noting a long ejection systolic murmur, which is heard best over the left second intercostal space, a pulmonary ejection sound, and a widely split S2 with reduced intensity of the pulmonary component of the S2. Pulmonary hypertension is also easily recognized at the bedside by noting the increased intensity of the pulmonic component of the S2 and its transmission to the cardiac apex (mitral area).
occur in patients with a prolonged PR interval when atrial systole occurs during the preceding ventricular systole. When a prominent ‘a’ wave with large amplitude is appreciated, it is desirable to exclude tricuspid valve obstruction; this can be done by noting absence of a mid-diastolic rumble along the lower left sternal border, which increases in intensity during inspiration. Although in severe tricuspid stenosis the ‘y’ descent is abbreviated and presystolic hepatic pulsations can be observed, such a degree of tricuspid stenosis is rarely encountered. It should also be emphasized that isolated tricuspid stenosis is almost never encountered in rheumatic heart disease, and rheumatic tricuspid stenosis occurs almost always in the presence of mitral or aortic valve disease and in patients who are usually in atrial fibrillation. In adult cardiac patients, if isolated tricuspid stenosis is recognized with a prominent ‘a’ wave with a mid-diastolic rumble along the lower left sternal border that increases in intensity during inspiration (Carvello sign), right atrial myxoma or carcinoid heart disease should be considered in the differential diagnosis. However, the most common cause of a prominent ‘a’ wave in adults is right ventricular hypertrophy, which offers increased resistance to right atrial emptying during right atrial systole. Right ventricular hypertrophy may result from a right ventricular outflow tract obstruction, such as pulmonary valve stenosis or precapillary or postcapillary pulmonary hypertension. Significant pulmonary valve stenosis can be easily diagnosed by noting a long ejection systolic murmur, which is heard best over the left second intercostal space, a pulmonary ejection sound, and a widely split S2 with reduced intensity of the pulmonary component of the S2. Pulmonary hypertension is also easily recognized at the bedside by noting the increased intensity of the pulmonic component of the S2 and its transmission to the cardiac apex (mitral area).
If a prominent ‘v’ wave in the jugular venous pulse is recognized at the bedside, tricuspid regurgitation should be suspected, and physical findings to confirm the diagnosis should be sought. In tricuspid regurgitation, jugular venous pulse reveals a sharp ‘y’ descent after a prominent ‘v’ wave. In severe tricuspid regurgitation, lateral pulsatile motions of the earlobes coincident with each cardiac cycle are observed in many patients. There usually is a systolic murmur either early or pansystolic in duration, which is heard best over the left third and fourth intercostal spaces, along the left sternal border. Tricuspid regurgitation murmur frequently radiates to the epigastrium and the right side of the sternum. It may be heard even over the jugular venous pulse. The tricuspid regurgitation murmur also increases in intensity after inspiration. When the diagnosis of tricuspid regurgitation is confirmed, it is desirable to assess carefully the intensity of the pulmonic component of the S2 to distinguish between primary and secondary tricuspid regurgitation. Secondary tricuspid regurgitation is defined when tricuspid regurgitation occurs owing to right ventricular dilatation and failure secondary to pulmonary hypertension.
Occasionally, a prominent ‘v’ wave is detected in patients with atrial septal defect in the absence of pulmonary arterial hypertension and tricuspid regurgitation (22). The mechanism of the prominent ‘v’ wave in atrial septal defect is not clear; however, a concomitant increase in systemic venous return and left-to-right shunt during ventricular systole may cause a rapid increase in right atrial pressure—hence, a prominent ‘v’ wave. It should be emphasized that a prominent ‘v’ wave in patients with atrial septal defect is not followed by a sharp ‘y’ descent.
Although a sharp ‘y’ descent most frequently occurs in tricuspid regurgitation, it can be observed in constrictive pericarditis and restrictive cardiomyopathy. In constrictive pericarditis and restrictive cardiomyopathy, systemic venous pressure is elevated, and inspection of the jugular venous pulse may reveal a sharp ‘y’ descent of brief duration. In constrictive pericarditis or restrictive cardiomyopathy, the jugular venous pressure does not fall appropriately or even increase during inspiration (Kussmaul sign) (23) (see Fig. 16.3). It should be recognized that the Kussmaul sign is not diagnostic of constrictive pericarditis or restrictive cardiomyopathy. Kussmaul sign can be observed in patients with right ventricular infarction (24), primary severe right ventricular failure resulting from any cause, primary or secondary severe tricuspid regurgitation, partial obstruction of the vena cava, or right atrial and right ventricular tumors; in some patients with severe CHF without tricuspid regurgitation; and, occasionally, in patients with tricuspid stenosis. The Kussmaul sign is observed in some elderly patients without any obvious pathology (personal observation). The Kussmaul sign is uncommon and seldom noted in cardiac tamponade. The mechanism of the Kussmaul sign has not been adequately studied; however, increased resistance to right atrial filling during inspiration seems to be a common contributory factor. For practical purposes, in patients with chronic CHF, a presence of the Kussmaul sign should raise the possibility of constrictive pericarditis or restrictive cardiomyopathy, and appropriate evaluations should be considered. The presence of physical findings of pulmonary hypertension (e.g., a sustained systolic left parasternal lift, a loud pulmonic component of the S2, and severe tricuspid regurgitation with pulsatile hepatic impulse) favor the diagnosis of restrictive cardiomyopathy. In contrast, lack of findings suggestive of pulmonary hypertension, a quiet precordium or presence of a prominent diastolic left parasternal impulse, and absence of hepatic pulsation favor the diagnosis of constrictive pericarditis. In constrictive pericarditis, a pericardial “knock” (similar to right-sided S3 gallop) and, rarely, a mid-diastolic murmur are heard. Once constrictive pericarditis or restrictive cardiomyopathy are suspected, appropriate investigations should be performed (see Chapter 54).
In patients with suspected CHF, it is desirable to perform the hepatojugular reflux test (25,26). With the patient breathing normally and in the semirecumbent position, firm pressure is applied with the palm of the hand to the upper right quadrant of the abdomen for at least 10 seconds. In normal patients, there may be a transient increase in jugular venous pressure with rapid return to or near baseline in less than 10 seconds. The abnormal hepatojugular reflux is defined when there is a rapid increase in jugular venous pressure that remains elevated by 4 cm or more until abdominal compression is released. During abdominal compression with increased intraabdominal pressure, there is an increase in venous return to the right atrium and right ventricle. Concurrently, there is an increase in right ventricular afterload owing to upward movement of the diaphragm, which reduces the intrathoracic volume capacity. The normally functioning right ventricle handles this increase in preload and afterload, and systemic venous pressure remains normal. The dysfunctioning right ventricle, however, fails to accept this increase in preload and afterload; therefore, there is a persistent elevation of systemic venous pressure. A positive hepatojugular reflux is most frequently associated with CHF resulting from left heart failure (27). In these circumstances, an abnormal sustained elevation of right atrial pressure during hepatojugular reflux indicates incipient right heart failure or abnormal compliance of the right ventricle in the presence of intact pericardium (Fig. 16.4). It should be emphasized that a positive hepatojugular reflux is noted in patients with isolated right heart failure due to precapillary pulmonary hypertension or right ventricular infarction. The abnormalities of the venous pressure and pulse and associated suspected cardiovascular disorders are summarized in Table 16.3.
Examination of the Precordial Pulsations
Inspection and palpation of precordial pulsation is best performed in patients in supine position with head and trunk
elevated (45 degrees) (1,2). Normally, a slight, abrupt, inward pulsation can be seen occasionally over the lower left parasternal area, particularly in children and thin-chested patients. Epigastric and subxyphoid pulsations are usually abnormal, although they can be seen in patients with COPD. A pronounced epigastric or subxyphoid pulsation should raise the possibility of right ventricular failure or abdominal aortic aneurysm. A visible pulsation over the right second intercostal space or right sternoclavicular joint may indicate aneurysm of the ascending aorta. Aneurysm of the arch of the aorta may also cause suprasternal pulsation. The most common cause of right supraclavicular pulsation is a kinked, tortuous right carotid artery. A visible pulsation over the left second or third interspace may be due to dilated pulmonary artery, which may result from increased flow, such as with atrial septal defect or increased pressure, as in patients with precapillary or postcapillary pulmonary hypertension. Systolic outward parasternal and left ventricular outward movements are better appreciated by palpation. However, in many patients, an abnormal left ventricular apical impulse is visible and may result from a pronounced, sustained, outward movement or hyperdynamic left ventricular apical impulse. When cardiac pulsations are visible lateral to the left midclavicular line, cardiac enlargement should be suspected. Leftward displacement of the cardiac apex may occur due to fibrosis of the left lung, right-sided tension pneumothorax, or massive left pleural effusion. Absent left pericardium (a congenital anomaly) and thoracic deformity may also cause visible pulsations beyond the midclavicular line. Occasionally in patients with adhesive pericarditis, retraction of the ribs in the left axilla (Broadbent sign) are recognized. In patients with severe dilated congestive cardiomyopathy, a double or triple impulse over the left ventricular apex can be recognized and represents a sustained left ventricular outward movement, a prominent atrial filling wave, and early diastolic filling impulse.
elevated (45 degrees) (1,2). Normally, a slight, abrupt, inward pulsation can be seen occasionally over the lower left parasternal area, particularly in children and thin-chested patients. Epigastric and subxyphoid pulsations are usually abnormal, although they can be seen in patients with COPD. A pronounced epigastric or subxyphoid pulsation should raise the possibility of right ventricular failure or abdominal aortic aneurysm. A visible pulsation over the right second intercostal space or right sternoclavicular joint may indicate aneurysm of the ascending aorta. Aneurysm of the arch of the aorta may also cause suprasternal pulsation. The most common cause of right supraclavicular pulsation is a kinked, tortuous right carotid artery. A visible pulsation over the left second or third interspace may be due to dilated pulmonary artery, which may result from increased flow, such as with atrial septal defect or increased pressure, as in patients with precapillary or postcapillary pulmonary hypertension. Systolic outward parasternal and left ventricular outward movements are better appreciated by palpation. However, in many patients, an abnormal left ventricular apical impulse is visible and may result from a pronounced, sustained, outward movement or hyperdynamic left ventricular apical impulse. When cardiac pulsations are visible lateral to the left midclavicular line, cardiac enlargement should be suspected. Leftward displacement of the cardiac apex may occur due to fibrosis of the left lung, right-sided tension pneumothorax, or massive left pleural effusion. Absent left pericardium (a congenital anomaly) and thoracic deformity may also cause visible pulsations beyond the midclavicular line. Occasionally in patients with adhesive pericarditis, retraction of the ribs in the left axilla (Broadbent sign) are recognized. In patients with severe dilated congestive cardiomyopathy, a double or triple impulse over the left ventricular apex can be recognized and represents a sustained left ventricular outward movement, a prominent atrial filling wave, and early diastolic filling impulse.
TABLE 16.3 Abnormalities of the Venous Pressure and Pulse and their Clinical Significance | |
|---|---|
|
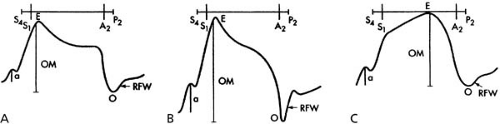
Palpation of left ventricular impulse and left parasternal impulse provide useful information to assess changes in cardiac dynamics and function. The left ventricular apical impulse is best palpated when the patient lies in a partial left lateral decubitus position. The outward movement of the left ventricular apical impulse is normally brief and localized and does not extend significantly into the left ventricular ejection phase (Fig. 16.5). The beginning of left ventricular ejection at the bedside is appreciated by the onset of the carotid pulse upstroke. The normal outward movement of the left ventricular apical impulse recedes from the chest wall and becomes impalpable with the onset of ejection after the upstroke of the carotid pulse and during the downstroke of the carotid pulse (28). Thus, at the bedside, when carotid pulse and left ventricular apical impulse are examined concurrently, these two impulses appear out of phase or asynchronous. The sustained apical impulse is characterized by a prolonged duration of the outward movement extending into the ejection phase of the left ventricle. Usually, this outward movement is diffuse and occupies more than one intercostal space. When the carotid pulse upstroke and duration are evaluated simultaneously with the left ventricular outward movement, the sustained apical impulse appears in phase with the carotid pulse upstroke and remains palpable during the downstroke of the carotid pulse.
With a hyperdynamic apical impulse, the amplitude of the outward movement is increased, but the sequence between the onset of the carotid pulse and the outward movement remains normal. The hyperdynamic apical impulse is appreciated as a thrust of large amplitude that immediately disappears from the palpating fingers. When the duration of the ejection phase is estimated from the carotid pulse upstroke and downstroke or from the interval between the S1 and S2, the hyperdynamic apical impulse does not extend throughout systole. The sustained outward movement usually is felt as a heave. The duration of the outward movement of the apical impulse when it is sustained extends throughout the ejection phase. A sustained apical impulse usually is found when there is significant impairment of left ventricular systolic function (i.e., reduced ejection fraction) (29,30). Another cause of sustained apical impulse is marked left ventricular hypertrophy resulting from systemic hypertension or left ventricular outflow tract obstruction. In patients with known coronary artery disease or previous myocardial infarction and dilated cardiomyopathy, a sustained apical impulse almost invariably indicates reduced left ventricular ejection fraction. Systolic bulges occasionally may be appreciated in the absence of previous myocardial infarction and probably result from myocardial ischemia. At the bedside, however, these transient abnormal systolic motions are difficult to appreciate. Occasionally in patients with mitral valve prolapse, a deep notch in the systolic portion of the left ventricular apical impulse, which coincides with the midsystolic click, is recorded in the apex cardiogram, but it is difficult to recognize at the bedside. The hyperdynamic apical impulse occurs in conditions associated with an increased stroke volume or volume overload. Hyperdynamic impulse is found in patients with hypermetabolic state, such as hyperthyroidism, anemia, primary mitral regurgitation, and aortic regurgitation, and in some patients
with a patent ductus arteriosus and ventricular septal defect with a large left-to-right shunt. A hyperdynamic impulse, in general, suggests preserved left ventricular ejection fraction.
with a patent ductus arteriosus and ventricular septal defect with a large left-to-right shunt. A hyperdynamic impulse, in general, suggests preserved left ventricular ejection fraction.
While examining the left ventricular apical impulse, particularly with the patient in the left lateral decubitus position, a double impulse is felt and almost always represents a palpable ‘a’ wave (presystolic wave) and a prominent outward movement. The palpable ‘a’ wave is related to an accentuated atrial filling wave and is most frequently observed in patients with noncompliant left ventricle, such as in patients with aortic stenosis, hypertensive heart disease, or ischemic heart disease. The hemodynamic correlate of palpable ‘a’ wave is increased left ventricular end-diastolic pressure. Occasionally, a palpable, rapid filling spike, which coincides with a prominent S3 gallop, is appreciated during palpation of left ventricular apical impulse. At the bedside, this palpable early diastolic rapid filling spike is appreciated after the outward movement. The hemodynamic correlate of palpable S3 gallop is also increased left ventricular diastolic pressure. In occasional patients with obstructive hypertrophic cardiomyopathy, the apical impulse may have a bifid outward movement and if there is a prominent ‘a’ wave, a triple impulse may be felt (31,32). The characteristics of the left ventricular outward movement, along with changes in the filling waves, are illustrated in Fig. 16.5.
Right ventricular impulse is appreciated by palpation of the lower left parasternal area. In children and in some adults with thin chest walls, a brief, gentle impulse may be palpable over the left third and fourth interspaces. This impulse usually is in diastole. A prolonged left parasternal impulse extending during the ejection phase (i.e., throughout the carotid pulse upstroke and downstroke or between S1 and S2) is distinctly abnormal and reflects right ventricular failure or right ventricular hypertrophy. In the presence of significant mitral regurgitation, left atrial expansion during left ventricular systole may also produce a sustained systolic left parasternal impulse. In patients with right ventricular hypertrophy and failure, a sustained epigastric impulse is also appreciated. An inward systolic movement and an outward diastolic movement are sometimes appreciated in some patients with constrictive pericarditis (33). The diastolic movement usually coincides in timing with the pericardial knock. The precise explanation for these unusual precordial impulses in constrictive pericarditis has not been clarified. It has been suggested that this unusual outward movement during isovolumic systole is inhibited by the constriction, and the outward movement during early diastole becomes accentuated. In patients with severe chronic constrictive pericarditis, however, the precordium usually is quiet, and no obvious precordial impulses are appreciated at the bedside. In patients with Ebstein anomaly, the precordium may also be quiet. In some patients with Ebstein anomaly, a right parasternal systolic outward movement resulting from a large, ventricularized right atrium is appreciated. A hyperdynamic left parasternal impulse may be palpable in volume overloaded right ventricle as with an atrial septal defect or tricuspid regurgitation (34).
A left ventricular aneurysm can be associated with a systolic impulse in unusual locations, such as over the midprecordium. Pulsation in the left second interspace usually reflects an enlarged pulmonary artery resulting from chronic severe pulmonary arterial hypertension or from increased pulmonary flow, as in atrial septal defect. Occasionally, a higher frequency impulse is appreciated coincident with the downstroke of the carotid pulse in the left second interspace and usually reflects an accentuated pulmonary component of the S2, as in patients with severe chronic pulmonary hypertension. A pulsatile mass in the suprasternal notch may indicate aneurysm of the ascending aorta. Aneurysm of the ascending aorta is sometimes associated with the tracheal tug, which is appreciated when the trachea is slightly pulled upward and the pulsation in the trachea is recognized with each cardiac cycle. A pulsatile mass in the right supraclavicular area is most frequently caused by a kinked right carotid artery. Such an abnormal impulse may indicate the presence of atherosclerotic peripheral vascular disease.
Auscultation
A systematic and careful cardiac auscultation (1,35) is essential for the diagnosis of a cardiovascular abnormality. Auscultation should be performed whenever possible with the patient in the left lateral decubitus, supine, and sitting positions. Auscultation can begin over the cardiac apex—the so-called mitral area—and one can proceed counterclockwise to the left fourth interspace (tricuspid area), left third interspace, left second interspace (pulmonic area), and right second interspace (aortic area), sometimes, over the right fourth interspace adjacent to the sternal borders, and, finally, over the epigastrium. It is customary to assess the intensity and splitting of the S1 and S2 and the presence of S3 and the fourth heart sound (S4). It is also desirable to listen for the presence of abnormal early systolic, midsystolic, early diastolic, and late diastolic heart sounds.
After the analysis of the normal and abnormal heart sounds, heart murmurs, if present, are analyzed. When certain diagnoses are suspected, areas of auscultation should include the left axilla, thoracic and lumber spine, vortex (mitral regurgitation), axilla and back (pulmonary artery branch stenosis), posterior chest (coarctation of the aorta), left infraclavicular area (patent ductus arteriosus), and sternoclavicular joints (venous hum).
After the analysis of the normal and abnormal heart sounds, heart murmurs, if present, are analyzed. When certain diagnoses are suspected, areas of auscultation should include the left axilla, thoracic and lumber spine, vortex (mitral regurgitation), axilla and back (pulmonary artery branch stenosis), posterior chest (coarctation of the aorta), left infraclavicular area (patent ductus arteriosus), and sternoclavicular joints (venous hum).
First Heart Sound
Intensity
For the analysis of the S1, the diaphragm is used, and auscultation is performed over the mitral and tricuspid areas. The intensity of the S1 is determined primarily by the intensity of mitral component (M1) of the S1. Several factors contribute to the intensity of M1 (36,37). The position of the mitral valve at the onset of systole, the rate of mitral valve closure, the mobility of the mitral valve, the PR interval, and the dP/dT of the left ventricle—all are important factors that influence the intensity of M1. The position of the atrioventricular valves at the beginning of the ventricular systole, and the velocity of closure seem to be the major determinants of the intensity of the S1. When the mitral valve remains fully open until the very end of diastole and then closes rapidly, the intensity of M1 is increased. The greater the distance that the mitral valve leaflets have to travel from the open to closed positions and the greater the velocity of closure of the mitral valve, the louder the S1. If a substantial increase in the intensity of the S1 is recognized, an increase in transmitral valvular pressure gradient (e.g., in mitral stenosis), increased transvalvular flow (e.g., in the presence of large left-to-right shunt owing to ventricular septal defect or a patent ductus arteriosus), a very short PR interval (e.g., in preexcitation syndrome), or markedly shortened left ventricular diastole (e.g., in tachycardia) should be suspected. When mitral valve obstruction and short PR interval are excluded, increased intensity of the S1 may reflect an increased left ventricular dP/dT, as during adrenergic stimulation. The increased intensity of the tricuspid valve closure sound (T1), which is appreciated over the left third and fourth interspaces along the lower sternal border, usually occurs when the transtricuspid valve pressure gradient is increased, as in patients with tricuspid stenosis or right atrial myxoma. A substantial increase in diastolic flow across the tricuspid valve, as in large atrial septal defect, may also increase the intensity of the T1. Intermittent increased intensity of the S1 in the presence of regular pulse and heart rhythms indicates atrioventricular dissociation, results from varying PR intervals, and is associated with variable rates of closure of atrioventricular valves. An increase in intensity of the S1 has been observed in some patients with mitral valve prolapse, despite mitral regurgitation, and probably reflects increased adrenergic activity (38).
A decrease in intensity of the S1 can result from a substantial loss of the tissue mass of the atrioventricular valves, as occurs in severe rheumatic tricuspid and mitral valve diseases and bacterial endocarditis. This decrease can also result from the marked restriction of the movement of the atrioventricular valves because of calcification or sclerosis of the valve leaflets, which occasionally occurs in patients with rheumatic mitral valve stenosis and may be associated with decreased—rather than increased—intensity of the S1. When the PR interval is prolonged (>200 ms), the intensity of the S1 decreases because semiclosure of the mitral valve occurs after atrial systole and before ventricular systole begins. In severe aortic regurgitation, premature closure of the mitral valve may occur owing to a rapid rise in left ventricular diastolic pressure, and the mitral valve may be virtually closed at the onset of ventricular systole, resulting in a markedly decreased intensity of the S1. In patients with acute severe aortic regurgitation associated with marked increase in left ventricular diastolic pressure, the S1 may be inaudible and should be regarded as an indication for surgical intervention (39). A substantial increase in left ventricular diastolic pressure is an important mechanism for decreased intensity of the S1 in the absence of prolonged PR interval and restricted mitral valve mobility. Impaired contractile function, as in dilated cardiomyopathy, is contributory to reduced intensity of the S1. In these patients, left ventricular diastolic pressure is also frequently elevated. Occasionally, decreased intensity of S1 is observed in isolated left bundle branch block, probably reflecting impaired left ventricular function (40).
A variable intensity of the S1 is common in atrial fibrillation. Auscultatory alternans—in which the S1 is soft and loud in intensity with alternate beats—is a rare finding of severe cardiac tamponade and is almost always associated with electrical alternans and pulsus paradoxus. Auscultatory alternans also has been observed in patients with pulsus alternans, in whom beat-to-beat alteration in the left ventricular dP/dT occurs (41).
Decreased conduction of sounds through the chest wall reduces the intensity of the S1 in patients with COPD, obesity, and pericardial effusion. In these circumstances, all heart sounds appear soft and distant. One of the practical difficulties in assessing the intensity of the S1 at the bedside is the lack of any objective method to standardize its intensity. The S1 normally is loudest at the apex and along the lower left sternal border.
Splitting
The splitting of the S1 (42) is best appreciated along the left parasternal areas and is most frequently observed in the presence of complete or incomplete right bundle branch block. In right bundle branch block, the S2 is also widely split, and the A2–P2 interval widens during inspiration. Delayed closure of T1 because of increased flow across the tricuspid valve (atrial septal defect) or increased transtricuspid valve pressure gradient (tricuspid stenosis) causes wide splitting of S1 without splitting of the S2. The widely split S1 is recognized in patients with the Ebstein anomaly, not only because of right ventricular conduction disturbances but also from the delayed closure of the tricuspid valve owing to atrialization of the right ventricle. In Ebstein anomaly, the S2 is also widely split, and, frequently, systolic and diastolic, scratchy, superficial sounds—so-called sail sounds—are present (43). The reversed splitting of the S1 is extremely rare and difficult to recognize at the bedside. Reversed splitting of the S1 can result from severe mitral stenosis and is rarely caused by left bundle branch block. In patients with severe mitral stenosis, delayed closure of the mitral valve contributes to the reversed splitting of the S1. However, earlier closure of the tricuspid valve resulting from secondary tricuspid regurgitation is also necessary for the reversed splitting of the S1 in mitral stenosis.
Sounds Mimicking the First Heart Sound
At the bedside, it is necessary to distinguish between splitting of the S1 and the presence of a loud atrial sound preceding the S1 (Fig. 16.6). The left ventricular atrial sound (S4) usually is localized over the cardiac apex and is heard best with the bell of the stethoscope. When auscultation is started with the use of the bell over the cardiac apex, S1 and S4 are heard easily. When the bell is converted to the diaphragm by applying firm pressure over the underlying skin, the S4 decreases in intensity or disappears, whereas the splitting of the S1 becomes more obvious.
 FIGURE 16.6. The causes and differential diagnosis of the first heart sound (S1). Abbreviations: A2, aortic component of the second heart sound; P2, pulmonary component of the second heart sound. |
The combination of a systolic ejection sound and S1 may also appear as split S1. The S1 and the ejection sound interval usually is greater than the normal M1–T1 interval. The aortic ejection sound is widely transmitted and, therefore, can be heard easily over the aortic area, along the left sternal border, and over the cardiac apex. On the other hand, the split S1 is best appreciated along the lower left sternal border, over the left third and fourth interspaces. Pulmonary valvular ejection sounds can be easily distinguished from T1. Pulmonary ejection sounds are usually localized and heard best over the left second interspace. Pulmonary valvular ejection sounds decrease in intensity during inspiration, whereas the intensity of T1 remains unchanged or increases after inspiration.
A combination of S1 and a midsystolic click owing to mitral valve prolapse is rarely confused with a split S1. The interval between S1 and a midsystolic click is much greater than the interval between M1 and T1. Furthermore, the S1 to midsystolic click interval can be changed by maneuvers such as standing and squatting. These maneuvers, however, usually do not alter the interval between M1 and T1 significantly enough to be appreciated at the bedside. The presence of a pacemaker sound preceding S1 may seem like a widely split S1. The pacemaker sound results from the stimulation of the intercostal muscles during pacing, precedes S1, and occurs well before the upstroke of the carotid pulse (44). Furthermore, the pacemaker sound disappears with discontinuation of pacing. The causes and the differential diagnosis of the abnormalities of the S1 are summarized in Table 16.4.
TABLE 16.4 Usual Causes of the Abnormalities of the First Heart Sound | |||||
|---|---|---|---|---|---|
|
Second Heart Sound
The genesis of the S2 appears to be related to closure of the aortic and pulmonary valves; thus, the S2 traditionally is regarded to consist of two components designated as A2 (associated with aortic valve closure) and P2 (associated with pulmonary valve closure). The first high-frequency component of A2 and P2 is
coincident with completion of closure of these semilunar valves (45). It should be appreciated that A2 and P2 are not produced by the clapping together of the valve leaflets but by the sudden deceleration of retrograde flow of the blood column in the aorta and pulmonary artery when the maximum tensing of these valve leaflets occurs. The abrupt deceleration of flow produces the vibration of the cardiohemic system, and the lower frequency vibrations are coincident with the incisura of the great vessels, whereas the higher frequency components cause A2 and P2.
coincident with completion of closure of these semilunar valves (45). It should be appreciated that A2 and P2 are not produced by the clapping together of the valve leaflets but by the sudden deceleration of retrograde flow of the blood column in the aorta and pulmonary artery when the maximum tensing of these valve leaflets occurs. The abrupt deceleration of flow produces the vibration of the cardiohemic system, and the lower frequency vibrations are coincident with the incisura of the great vessels, whereas the higher frequency components cause A2 and P2.
Intensity
Aortic Component
The amplitude and intensity of A2 and P2 are directly proportional to the rate of change of the diastolic pressure gradient that develops across the semilunar valves—that is, the driving force that accelerates the blood mass retrograde into the base of the great vessels (46). The rate of pressure decline in the ventricle and the level of the diastolic pressure in the great vessels determine the pressure gradient in the root of the great vessels. Normally, the diastolic pressure gradient in the aorta is significantly greater than that in the pulmonary artery, which explains the normal increased intensity of A2 compared with that of P2. The most common cause of the increased intensity of A2 is systemic hypertension. Occasionally, in addition to the increased intensity, a tambour quality of A2 is recognized in systemic hypertension. Such altered quality of A2 also is appreciated in some patients with aneurysm of the ascending aorta. The decreased intensity of A2 most frequently occurs from immobility of calcified, sclerosed aortic valves in calcific aortic stenosis. In aortic regurgitation resulting from fibrosed and retracted aortic valve leaflets, as in syphilitic aortic regurgitation, the aortic component of the S2 also is decreased in intensity. Normally, A2 is widely transmitted and well heard at the cardiac apex. However, in patients with significant primary mitral regurgitation, A2 may be drowned at the cardiac apex by the regurgitant murmur that frequently extends beyond the aortic valve closure.
Pulmonic Component
The pulmonic component of the S2—that is, P2—is softer than A2 and is rarely audible at the apex. Increase in the intensity of P2 indicates pulmonary hypertension, irrespective of its etiology. When there is a substantial increase in its intensity, P2 is also heard at the cardiac apex. Without pulmonary hypertension, it is uncommon for the P2 to be transmitted to the cardiac apex. In only approximately 5% of healthy subjects, and only when they are young (<20 years old), can P2 be recorded by phonocardiography over the cardiac apex (47). A palpable P2 over the left second interspace indicates severe pulmonary hypertension.
When the cardiac apex is occupied by the right ventricle—as in patients with large atrial septal defects—P2




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree