Intra-aortic balloon console tracings. The screen displays the ECG trace at the top, the intra-aortic pressure waveform in the center, and balloon inflation/deflation at the bottom. (a) Full IABP augmentation with inflation 1:1 and inflation interval highlighted, showing correct balloon inflation at the dicrotic notch of the arterial pressure trace. (b & c) Full augmentation with inflation ratio of 1:2 and 1:3 respectively. (d) Balloon augmentation set at 50%. (e) IABP on full support but showing diminished effectiveness due to tachycardia. (f) Auto R-wave deflate mode for a patient in AF.
The effectiveness of IABP augmentation is diminished when there is excessive tachycardia (> 120 bpm) or when the cardiac rhythm becomes irregular, e.g. atrial fibrillation (see Figure 9.1e–f). Therefore, inotropic support should be moderated to minimize the occurrence of such rhythm disturbances. Invasive hemodynamic monitoring is indispensable and provides the best assessment of the adequacy of circulatory support. A Swan–Ganz pulmonary artery flotation catheter can provide important hemodynamic information including left ventricular preload (pulmonary capillary wedge pressure), left ventricular afterload (systemic vascular resistance), right ventricular afterload (pulmonary vascular resistance), and cardiac output, as well as providing information on the adequacy of systemic oxygen delivery (mixed venous oxygen saturation or SvO2). When cardiac function begins to recover, the inotrope dose should be reduced before IABP support is weaned. If the cardiac index is maintained above 2.2 l min−1 m−2 with acceptable preload (pulmonary capillary wedge pressure < 15 mmHg), attempts can be made to wean the IABP. Firstly, the IAB augmentation is reduced to 50% for 2–4 hours. The inflation ratio is then reduced from 1:1 to 1:2 for another 2–4 hours and then to 1:3 before the balloon catheter is removed. The IABP must be switched off and the catheter completely deflated just prior to removal. Heparin infusion should be discontinued at the start of the weaning process so that coagulation is normalized by the time the IAB catheter is ready for removal.
Complications of IABP use
Major vascular complications can occur in up to 15% of patients treated with an IABP. Femoral insertion of an IAB catheter may not be possible in 5% of patients, because of a tortuous or diseased ilio-femoral system. During insertion, vascular injury can lead to dissection, rupture or hemorrhage. Once in situ, distal limb ischemia can result from a combination of peripheral vasoconstriction and low cardiac output state or thromboembolism. Malpositioning of an IAB catheter may result in obstruction of visceral or renal arteries, making a visual check on catheter position via chest radiography essential.
In the operating room, placement is usually facilitated with the use of transesophageal echocardiography, which can also give an indication of the presence of atherosclerotic disease in the descending thoracic aorta.
Other complications can include infection, thrombocytopenia, and rupture of the IAB catheter. Helium is used as the driving gas for IAB inflation because of its high blood solubility, so reducing the risks from gaseous emboli in the event of balloon rupture. If the IAB ruptures, blood can be seen to track down the gas channel of the balloon catheter. Whenever this is observed, the IABP counterpulsation must be stopped immediately and the catheter removed.
Ventricular assist devices
Severe heart failure refractory to medical management and IABP support has an appalling prognosis. Inadequate systemic perfusion gives rise to end-organ dysfunction and metabolic acidosis, while excessive back-pressure from the failing left and right ventricles results in pulmonary edema and systemic venous congestion respectively. Ventricular assist devices (VADs) can be used to augment perfusion and relieve congestion, potentially reversing the damaging effects of severe heart failure.
VADs are mechanical blood pumps that can provide either left, right or biventricular support. A left ventricular assist device (LVAD) withdraws oxygenated blood from the left atrium or left ventricle, and returns it to the aorta; a right ventricular assist device (RVAD) withdraws venous blood from the right atrium or right ventricle, and returns it to the pulmonary artery. In general, it is preferable to cannulate the ventricle as VAD inflow as this configuration provides superior ventricular decompression, avoids ventricular stasis, and affords higher VAD flow rates.
The output of an LVAD is dependent on adequate right ventricular function to deliver sufficient blood flow across the lungs and into the left heart chambers for the LVAD to pump. Likewise, an RVAD can only provide benefit if the native left ventricle can generate enough stroke work to cope with the pulmonary blood flow produced by the RVAD. If both native ventricles are failing, two VADs are required in order to provide biventricular assistance to support the circulation.
Principles of assist devices
In its simplest form, emergency short-term VAD support can be provided by any blood pump (e.g. a Biomedicus centrifugal pump) and a couple of vascular cannulae: one for inflow from the heart to the pump; the other for outflow return from the pump to the aorta or pulmonary artery. In the absence of specialist VAD equipment, such a set-up can be lifesaving and used to maintain the circulation for hours or days.
There is, however, a growing number of temporary devices specifically intended for use as a VAD. These systems consist of blood pumps that are less traumatic to the blood components, and have cannulae that are designed to provide more secure attachment to the heart chambers with superior flow characteristics. Temporary VAD systems are intended for short-term circulatory support in the intensive care unit for days or weeks. Long-term VAD systems are designed to provide circulatory support for months or years. Continual improvements in implantable VADs are enabling patients to be discharged from hospital and treated as out-patients, often with a relatively normal quality of life.
The fate of patients receiving a VAD depends on the underlying cause of the cardiac dysfunction and its reversibility. In some cases of post-cardiotomy shock and fulminant myocarditis, cardiac function recovers after a period of circulatory support and the VAD can be weaned and removed, a process known as “bridge to recovery.” Unfortunately, in the majority of cases of chronic heart failure, e.g. ischemic or dilated cardiomyopathies, the myocardial dysfunction is unlikely to be reversible. Occasionally, a VAD is required for these patients who are usually already waiting for a heart transplant because of deteriorating cardiac status. Here the VAD is used to buy time for the patient until a suitable donor heart can be found, a process called “bridge to transplant” or BTT. For selected patients with advanced heart failure who are not transplant candidates, implantable VAD can be offered as a permanent support, a process sometimes called “destination therapy” or DT.
The VAD decision making process
The key decisions of which patient to support with a VAD, when to insert a VAD, whether the patient requires LVAD alone or biventricular assist device (BiVAD), and which VAD system to use are often difficult ones to make. They are influenced by a number of factors including the acuity of onset and the severity of heart failure, patient comorbidities, transplant waiting times, device and resource availability, and institutional experience.
Up until a few years ago, patients with heart failure were stratified according to the New York Heart Association (NYHA) functional classification, with Class I describing patients with cardiac disease but no symptoms or physical limitations, and Class IV describing patients in advanced heart failure with symtoms at rest who are often bedbound. However, among those patients with Class IV heart failure symptoms, there remained a very broad spectrum of pateints ranging from those who are stable on oral therapy to those who may be pre-terminal on inotropic support. The treatment strategy required varied according to the severity of heart failure. Therefore, it was necessary to further sub-classify patients with advanced heart failure.
The Interagency Registry of Mechanically Assisted Circulatory Support (INTERMACS) was established as a mandatory registry for all patients receiving an implantable mechanical circulatory support device (MCSD) in the USA. Based on patient characteristics at the time of device implantation and outcome analysis, seven profiles have been defined to further stratify patients in advanced heart failure, with Profile 1 being the most severely ill. NYHA Class IV patients are now subdivided into Profiles 1–6 while those in NYHA Class IIIb are described as Profile 7 (see Table 9.4).
| INTERMACS profile | Description | Time frame for definitive intervention |
|---|---|---|
| Profile 1 | Critical cardiogenic shock | Emergency intervention within hours |
| Profile 2 | Progressive decline | Urgent intervention within days |
| Profile 3 | Stable but inotrope dependent | Elective intervention within days/weeks |
| Profile 4 | Resting symptoms | Elective interventions within months |
| Profile 5 | Exertion intolerant | Variable urgency, maintain nutrition and organ function |
| Profile 6 | Exertion limited | Variable urgency, maintain nutrition and organ function |
| Profile 7 | Advanced NYHA III | Transplant or MCSD may not be currently indicated |
Profile 1 consists of decompensated patients with precarious hemodynamics and life-threatening hypoperfusion refractory to escalating inotropic support. There is worsening acidosis and rising lactate associated with oliguria and derangement of hepatic and renal functions, sometimes described as “crash and burn.” Unless circulatory support is provided within hours, irreversible shock will most likely follow. Due to the decompensated state of these patients, the use of implantantable devices in INTERMACS Profile 1 patients tend to result in a higher 30-day mortality (see Figure 9.2). For some of the sickest patients, despite restoring normal systemic perfusion, the function of some injured end organs may not recover and the patient does not survive. When there are significant uncertainties about the viability of a decompensated patient, a temporary MCSD may be deployed to see whether the shock state is reversible before more durable treatment options are considered. This approach is sometimes described as “bridge to decision.”
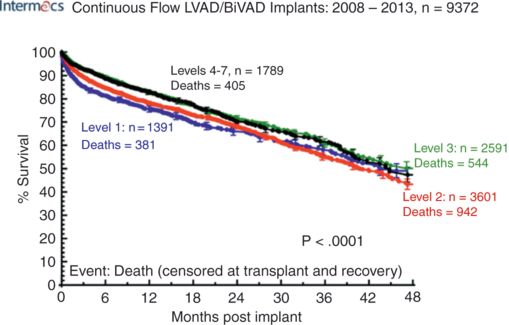
Actuarial survival after continuous-flow LVAD implant, stratified by INTERMACS level at implant.
Profile 2 describes patients with progressive decline in nutritional status, renal function, and/or fluid retention despite ongoing or escalating inotropes. Urgent intervention with MCSD is indicated to avoid further deterioration.
Profile 3 describes patients who are dependent on moderate levels of inotropic support but remain stable clinically and biochemically. In countries where waiting times for an urgent heart transplant is reasonable, this might be the more straightforward treatment option and a lower risk approach. However, close monitoring of hemodynamic and biochemical trends must be available to detect any subtle decline into INTERMACS Profile 2 as soon as possible in order to minimize potential increases in the risks associated with interventions using MCSDs.
Ventricular tachy-arrhythmia is an ominous sign and should prompt an earlier decision for VAD insertion. Since the “bridging” period to a transplant can range from months to sometimes over a year, these patients should be implanted with a long-term VAD so that they can be discharged home. Currently, there are several wearable or portable VAD systems that allow patients independent mobility during VAD support with reasonable quality of life.
Currently, there is insufficient evidence to support the use of MCSDs in INTERMACS Profile 4–7 patients. Earlier implantation of LVADs in these ambulatory patients does not confer superior post-implant survival when compared with inotrope-dependent INTERMACS Profile 3 patients. A number of prospective studies are currently underway to explore whether the use of implantable LVADs in Profile 4–7 patients could be justified.
There are certain patients who would otherwise be transplant candidates except for one or more serious, but potentially reversible, complications of advanced heart failure, e.g. pulmonary hypertension, renal dysfunction. Unless such complication(s) could be reversed, these patients are not transplantable and they will invariably die from their heart failure. Elevated pulmonary vascular resistance due to left ventricular failure and pulmonary venous congestion often revert to normal levels with mechanical unloading of the left ventricle using an implantable LVAD. When systemic perfusion is improved using MCS, renal dysfunction due to chronic low cardiac output state can sometimes be reversed. The use of LVAD implantation in these patients is sometimes described as “bridge to candidacy.”
Patients presenting with acute cardiogenic shock refractory to medical therapy have an appalling prognosis. The common causes of heart failure are listed in Table 9.5. Over the last decade, temporary MCSDs have been increasingly used to support such patients in order to maintain systemic perfusion while recovery of cardiac function is awaited. Those patients in whom cardiac function recovers sufficiently to be weaned from mechanical circulatory support tend to do so within 5–7 days. For those with insufficient cardiac recovery, a decision has to be made between escalating to longer-term devices, i.e. “bridge to bridge,” or treatment withdrawal and death. Cardiac arrested patients undergoing cardiopulmonary resuscitation can also be placed on peripheral extracorporeal life support to maintain systemic circulation while further cardiac and neurological assessment could be carried out. For patients with acute cardiogenic shock requiring treatment with temporary MCSDs, the overall survival to discharge is approximately 30–40%.
| Acute MI shock (5–8% of acute myocardial infarction) |
| Post-cardiotomy (1–4% after cardiac surgery) |
| Post-HTx. primary graft failure |
Miscellaneous Malignant ventricular arrhythmia Post-partum cardiomyopathy Fulminant myocarditis Poisoning, e.g. beta blocker overdose |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


