Fig. 17.1
The International Society for the Study of Vascular Anomalies classification system. Vascular anomalies are divided on the basis of cellular kinetics and clinical behavior into two major categories: vascular tumors and vascular malformations. Vascular malformations are subdivided into high-flow and low-flow vascular malformations (capillary, venous, and lymphatic malformations)
The separation of vascular anomalies into tumors and malformations permits more effective communication between different medical specialists, since, as previously mentioned, the management of CVM has been characterized by confusing and contradictory nomenclature and classification that was present in the majority of the medical literature discussing these lesions. Unfortunately, despite the ISSVA classification, the use of confusing nomenclature persists in the literature, and archaic terms such as “cavernous hemangioma,” “port wine stains,” “salmon patch,” “angel’s kiss,” and “nevus simplex” are still frequently used by some specialists.
17.3 Pathophysiology
Congenital vascular malformations arise by vascular dysmorphogenesis, between the fourth and tenth weeks of intrauterine life, without increased endothelial proliferation affecting a limited number of vessels in a restricted area of the body [3]. Recent data suggest that the pathophysiologic mechanism underlying the formation of CVM is caused by dysfunctions in the signaling process responsible for regulation of proliferation, differentiation, maturation, adhesion, and apoptosis of vascular cells [25–27]. A genetic TIE2 mutation has been described in certain hereditary cutaneomucosal venous malformations [28]. A number of genes have also been identified in the process of lymphangiogenesis including VEGFR3, VEGFC, Ang2, Lyve1, Nrp2, and podoplanin [29]. It is postulated that genetic aberrations of these genes and their regulatory mechanisms underline developmental defects during embryonic lymphangiogenesis, resulting in lymphatic malformations. In contrast to CVM, vascular tumors are true neoplastic disorders, and pathohistologically they demonstrate increased endothelial cell turnover rate [26, 30]. Hemangiomas represent the most common type of vascular tumor. Less common vascular tumors include Kaposiform hemangioendothelioma, angiolipoma, angiosarcoma, and hemangiopericytoma. Although hemangiomas and CVM have distinct histopathologic characteristics and clinical courses, occasionally, they coexist in the same settings, suggesting a possible overlap in the pathogenesis of the vascular tumors and malformations in certain cases [31].
17.4 Clinical Presentation
The differential diagnosis of vascular tumors and vascular malformations can be made by clinical assessment in the majority of cases. Vascular malformations are present at birth and do not spontaneously regress. Often they grow proportionately with the child’s growth with many becoming more prominent later in life. Their rapid growth and appearance may be stimulated by trauma, infection, and the effects of hormones (during puberty or pregnancy), or they may occur spontaneously in the absence of any identified triggering factors [32]. By contrast, infantile hemangiomas are characterized by a rapid proliferative phase in the first several months of life, followed by an involutional phase of slow, spontaneous regression in the majority of cases, frequently leaving adipofibrotic overlying dermis and telangiectasias that can remain visible at the location of the initial hemangioma [33]. In challenging and complex cases, histopathologic evaluation, immunohistopathologic markers, and radiologic studies are needed to help distinguish vascular tumors from vascular malformations.
The morphology, clinical presentation, and course of vascular malformations are variable in their extent and severity, depending upon location, proximity to vital structures, size or organ involved, and the type of vessel affected. Congenital vascular malformations are rarely asymptomatic. Usually they cause discomfort, pain, hemorrhage, and negatively affect the patient’s appearance as well as emotional well-being. In addition, these patients often have a significant reduction in daily functional capacity and quality of life. As discussed previously, if the malformation has an arterial component, it is classified as HFVM. The absence of arterial blood flow is the characteristic used to differentiate HFVM from LFVM. Some authors use the term fast-flow or arteriovenous malformation to describe HFVM. Low-flow vascular malformations are subdivided into venous malformations, lymphatic malformations, capillary malformations, and lesions that combine two or more of these elements.
Capillary malformations usually appear as localized pink or red lesions. They are present at birth and grow in proportion to the growth of the child. They appear darker immediately after the birth and lighten slightly in the first several weeks of life. This is believed to be secondary to the higher hemoglobin concentration that characterize the immediate newborn period [34]. Capillary malformations can occur as isolated cutaneous lesions or in association with other vascular malformations or other structural abnormalities such as bony or soft tissue hyperplasia or atrophy and neurological defect. In the limbs, capillary malformations are usually associated with osteomuscular hypertrophy (Fig. 17.2). When located on the head, they may extend to the gingiva, lips, and oral mucosa. Midline occipital capillary malformations can herald the presence of an encephalocele or ectopic meninges. Capillary malformations over the spine can be associated with occult spinal dysraphism [35]. In a retrospective review, Guggisberg et al. suggested that the combination of two or more midline capillary malformations is highly suggestive of spinal dysraphism [36]. However, the significance of capillary malformations as a marker for underlying spinal abnormalities is still unclear, and currently there are no evidence-based guidelines regarding screening of patients with capillary malformations for spinal defects. The most common syndrome associated with capillary malformations is Sturge-Weber syndrome. This neuroectodermal syndrome is characterized by an overabundance of capillaries around the ophthalmic branch (V1) of the trigeminal nerve, ipsilateral leptomeningeal angiomatosis, glaucoma (in approximately 50 % of patients), and seizures. Seizures are frequently present within the first year of life and tend to worsen over time.
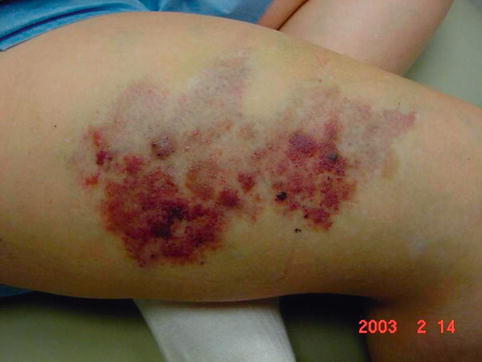

Fig. 17.2
Capillary malformation affecting the latero-medial aspect of the right thigh in the patient with Klippel-Trenaunay syndrome. Capillary malformations frequently occur in association with venous malformations and other structural abnormalities such as osteomuscular hypertrophy
Venous malformations are the most common peripheral LFVM encountered clinically. They are composed of anomalous dilated venous channels. They affect males and females equally with a reported prevalence of approximately 1 % [37]. On physical examination they appear as bluish, soft, and easily compressible, non-pulsatile masses that usually enlarge with activity, Valsalva maneuver (crying in children) or dependent posture, and empty with elevation. There is no increase in local skin temperature or thrill when the malformation is palpated, and there is no bruit present on auscultation (in contrast to HFVMs) (Fig. 17.3). Venous malformations are associated with swelling and episodes of pain as well as functional difficulties secondary to the involvement of muscles and joints. Although most venous malformations are solitary and occur in the skin and subcutaneous tissues, they can also occur as multiple, infiltrating lesions that can involve multiple soft tissue planes including muscles, abdominal viscera, and the central nervous system [38]. In terms of connection to the conducting veins, venous malformations are characterized as sequestered or communicating. In contrast to sequestered venous malformations, communicating malformations have direct connection to deep venous system. It is important to determine whether malformation is communicating since treatment of this type of malformation carries an increased risk of distal venous thromboembolic events. Venous malformations can also occur as part of syndromes. Klippel-Trenaunay syndrome (KTS) is a complex capillary-lymphatic-venous malformation which is associated with pathognomonic osteomuscular hypertrophy of the affected extremities in association with mixed venous and lymphatic and capillary lesions [39, 40]. In addition to obvious concerns about cosmesis, these patients often present with pain, swelling, orthostatic hypotension, or in severe cases pulmonary embolism secondary to insufficiency of the anomalous vein structures or repeated occult pulmonary emboli from intralesional thrombus. Blue rubber bleb nevus syndrome (BRBNS) is another rare syndrome associated with venous malformations. This sporadic disorder is characterized by multifocal venous malformations of the skin, soft tissues, and gastrointestinal tract [41, 42]. In addition to the risk of bleeding and subsequent anemia, gastrointestinal involvement makes these patients susceptible to intussusception and volvulus [43].
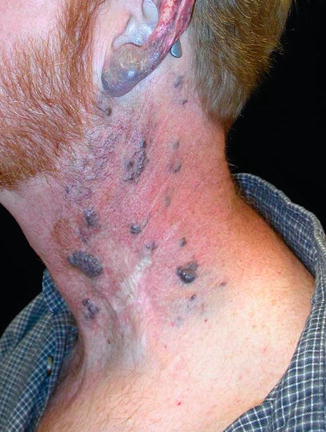

Fig. 17.3
Venous malformations appear as bluish, soft, and easily compressible, non-pulsatile masses that usually enlarge with activity. In contrast to high-flow vascular malformations, there is no increase in local skin temperature or thrill when the malformation is palpated, and there is no bruit present on auscultation
Lymphatic malformations are usually noted at birth or before the patient reaches the age of 2 years. Prenatal ultrasonography (US) can detect macrocystic lymphatic malformations during the late first trimester. The skin overlying these sponge-like lesions is usually normal or of bluish hue (Fig. 17.4). Lymphatic malformations can be characterized as microcystic, macrocystic, or combined. Macrocystic lymphatic malformations are generally defined as lesions that contain cyst spaces greater than 2 cm and microcystic as cysts smaller than 2 cm in diameter. Bleeding, bacterial infection, and swelling are the most common complications associated with lymphatic malformations. These complications can lead to the compression of surrounding structures, including the airway (if the malformation is located in the head or neck), or the optic nerve in the case of orbital lesions. In these patients, prompt management and urgent decompression are required to decompress the lesion and subsequently restore the normal function of the affected vital structures. Infection is a common complication of lymphatic malformations, and prompt recognition and treatment with systemic antibiotics that cover skin pathogens is imperative.
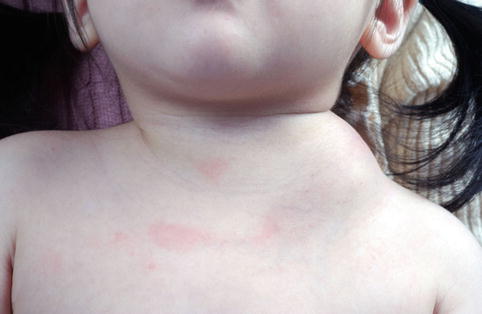

Fig. 17.4
A large lymphatic malformation extending from the left side of the neck. Lymphatic malformations are usually noted at birth (or before the patient reaches the age of 2 years). Macrocystic lymphatic malformations are defined as lesions that contain cyst spaces greater than 2 cm in diameter (*Courtesy of Dr. B.B. Lee)
17.5 Diagnosis
Despite distinct clinical, radiologic, and histological findings, LFVM are often confused with vascular tumors (most frequently with infantile hemangiomas) and many physicians do not understand the difference between these two vastly different lesions. Moreover, the complexity of many CVM contributes to the diagnostic challenge of differentiating LFVM from HFVM and differentiating LFVM from other congenital vascular anomalies. Since the prognosis, morbidity, and treatment significantly differ between vascular tumors, HFVM, and LFVM, proper diagnosis and accurate classification are critical for the successful management of these lesions [44]. A meticulous medical history and a detailed physical examination are essential initial steps in the management of CVM. However, clinical evaluation often underestimates the involvement of deep structures such as muscles, bones, joints, or abdominal viscera and is not sufficient to differentiate HFVM from LFVM and malformations from tumors in some of the more complicated lesions. Therefore, evaluation by advanced imaging modalities (color flow duplex ultrasound, magnetic resonance imaging) is of paramount importance for the correct diagnosis and management of CVM [45–47]. Ultrasonography of HFVMs is characterized by multidirectional blood flow and high-amplitude arterial waveform with spectral broadening. Ultrasonography of venous and lymphatic malformations reveals mixed venous waveform and complete absence of signal, respectively. On gray scale ultrasound, venous malformations appear as hypoechoic or heterogeneous lesions with anechoic structures visible in <50 % of cases. Duplex ultrasound of venous malformations generally demonstrates monophasic low-velocity flow. In some cases flow is only detectible with compression and release of the malformation. Macrocystic lymphatic malformations appear as anechoic cavities, often with internal septa and debris. Microcystic lymphatic malformations are hyperechoic, giving a more solid appearance.
In the evaluation of CVM, duplex ultrasound is useful to confirm the diagnosis, as it is rapid, readily available, and shows the flow velocity and vascularization [48]. It is also useful for initial assessment of superficial malformations; however it is frequently inadequate to demonstrate the extent of larger lesions. Therefore, magnetic resonance imaging (MRI) is the imaging modality of choice in the evaluation of CVM [49, 50]. It gives a bright hypersignal on T2-weighted spin-echo sequences that delineates the extent of the malformation throughout the involved tissues [51, 52]. In addition, MRI shows the lesion’s flow characteristics, relation to normal vascular and nonvascular structures, and provides good soft tissue definition (Fig. 17.5). In inconclusive cases, when suspicion of arterial flow is present based on MRI findings, an appropriate diagnostic workup includes an arteriogram, but only if treatment is deemed necessary. Every effort should be exerted to rule out a high-flow arterial component. It is worth emphasizing that this differentiation is of critical value in the management of CVM as treatment options for high-flow and low-flow lesions are different and the presence of an arterial component represents an absolute contraindication to transcutaneous sclerotherapy (due to the risk of arterial thrombosis and extensive tissue necrosis) which can be effectively used in the treatment of LFVM. In T2-weighted MRI, venous malformations demonstrate high-signal intensity, and this sequence is the best sequence to determine the full extent of the lesion and its relationship to adjacent tissues (Fig. 17.6a, b). Lymphatic malformations demonstrate predominantly fluid-type characteristics on all MRI sequences (low signal on T1 and high signal on T2 sequences). Macrocystic and microcystic components are easily differentiated since microcystic malformations demonstrate intermediate signal intensity on T1 and T2 spin-echo sequences.
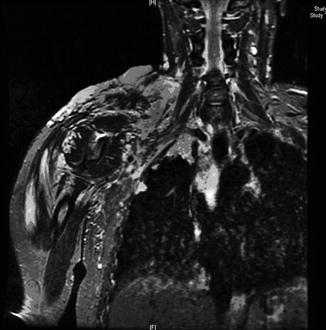

Fig. 17.5
Coronal T2-weighted image of the right upper extremity venous malformation. MRI gives a bright hypersignal on T2-weighted spin-echo sequences that delineates the extent of the malformation throughout the involved tissues
From a diagnostic standpoint, evaluation of the deep venous system deserves special consideration. In a study of 392 patients with CVM, Eifert et al. documented aplasia or hypoplasia of deep venous trunks in 8 % of patients (with venous predominance) [2]. In these patients venous blood flow from the affected limbs depends on superficial and abnormal vessels. Obliteration of these venous structures would compromise the venous circulation of the affected limb. Evaluation of patency and anatomic variations of the entire venous system (deep and superficial) is vital in these patients (Fig. 17.7). It has been reported that the prevalence of deep venous anomalies is even higher (18 %) in patients with KTS [53]. Based on the venous drainage channels and their response to treatment and rates of complications, Puig et al. divided venous malformations into four types: isolated malformations without discernible venous drainage (type I), lesions draining into normal veins (type II), lesions draining into dysplastic veins (type III), and lesions consisting primarily of venous ectasia (type IV). According to the same authors, types I and II respond best to sclerotherapy and higher rates of complications are attributed to types III and IV [17].
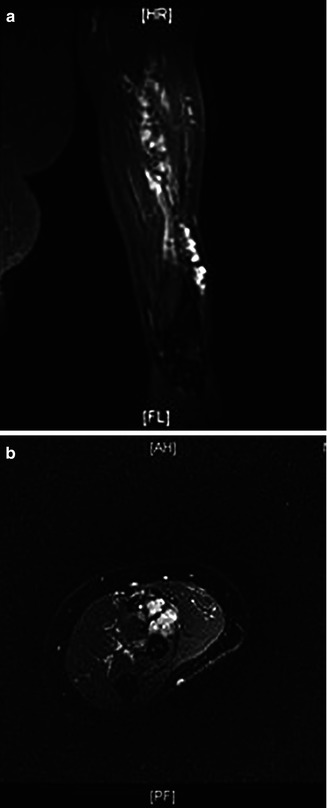
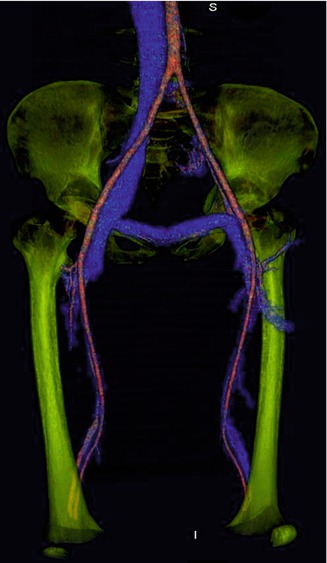

Fig. 17.6
MRI of the patient with venous malformation affecting the left upper extremity. A and B: coronal and axial (respectively) T2-weighted images of left upper extremity demonstrates multiple dilated venous channels in the subcutaneous and deeper soft tissues in the antero-lateral aspect of the left upper extremity

Fig. 17.7
MRI reconstruction of patient with Klippel-Trenaunay Syndrome (KTS) demonstrates absence of the left iliofemoral vein segment. The prevalence of deep venous anomalies is high (18 %) in patients with KTS. Evaluation of patency and anatomic variations of the deep venous system is important in patients with vascular malformations
In addition to being at increased risk of having deep venous anomalies, patients with extensive LFVMs may develop an intralesional consumptive coagulopathy [54, 55]. In a study of 118 patients, Mazoyer et al. demonstrated that localized intravascular coagulopathy (LIC) was present in 58 % of venous malformation patients [56]. In the literature, this coagulopathy is often erroneously labeled as Kasabach-Merritt syndrome (a distinct clinical entity characterized by disseminated intravascular coagulation and profound thrombocytopenia associated with vascular tumors) [57]. The platelet count in LIC is minimally diminished (in the 100–150 × 103/mL range). This distinction is important because, in contrast to patients with Kasabach-Merritt syndrome, LIC can be treated with heparin. Localized intravascular coagulopathy can be asymptomatic, but it may, rarely, be associated with painful intralesional thrombotic episodes and can progress to disseminated intravascular coagulation with life-threatening hemorrhage [58–60]. Although controversial, some authors advocate the use of aspirin or low molecular weight heparin (depending on the severity of symptoms) to be administered in patients with painful thrombotic episodes [58]. The increased risk of bleeding in some of these patients can be attributed to the increased consumption of coagulation factors. This is especially concerning in patients undergoing surgical resection of the lesion. A hypercoagulability profile should be considered in LFVM patients prior to undergoing imaging studies, surgical intervention, or sclerotherapy. An elevated D-dimer level and variable fibrinogen level is the hallmark of LIC. This finding is so common that some authors use D-dimer to differentiate venous malformations from lymphatic malformations (which do not show elevated D-dimer levels). Although LIC is usually latent and asymptomatic, it is worth emphasizing that these patients can become severely coagulopathic even during diagnostic procedures (US, MRI, angiography).
17.6 Treatment
Since the management of CVM falls in the range of several medical and surgical specialties, it is critical to establish a multidisciplinary approach for their diagnosis and treatment [61–64]. This was first discussed and introduced at the International Symposium for Congenital Vascular Malformations held in Seoul in 1996 [65]. In the past several years, several medical centers have developed multidisciplinary CVM teams [66]. Ideally, to achieve a consensus view of management, representatives from different medical specialties should be involved in the management of CVM patients and all treatments should be based on team assessments and decisions. This also affords the opportunity to streamline the evaluation process for patients with vascular anomalies, to coordinate the care of these patients, and to treat them comprehensively by reducing the need for multiple visits to different clinics. Given the heterogeneous nature and complexity of CVM, every patient and every lesion should be individually discussed. Multidisciplinary preoperative evaluation is of paramount importance, especially to rule out vascular tumors and to differentiate LFVM from HFVM and to identify lesions most amenable to resection. The decision for intervention has to take into account the size, location, proximity to vital structures and the natural history of the lesion, the risk of complications, and the relative risk of surgical or endovascular intervention. Low-flow vascular malformations can be treated with laser therapy, sclerotherapy, surgical excision, or combination therapy.
The flashlamp-pumped pulsed dye laser (FPDL) is the most frequently used as a treatment of choice for capillary malformations especially in areas in which there are concerns about ulcerations or hyperpigmentation from sclerotherapy. The most commonly used wavelengths are 577, 585, and 595 nm since they selectively target oxyhemoglobin. Pulse duration is used to concentrate and limit heat distribution (and subsequent collagen contraction and obliteration) to the capillary malformation while preserving surrounding structures in epidermis and dermis. Efficacy rates of the FPDL in the treatment of capillary malformations are variable. Reyes et al. reported good response in as many as 80 % of treated patients [67]. In a retrospective study of 259 adults and children, Renfro et al. demonstrated that responsiveness of the capillary malformations depends on the anatomical location of the lesion [68]. Lesions located on the central face or limbs were less responsive to FPDL than lesions located on the neck and trunk. It must be noted that multiple treatments are needed to achieve acceptable results. Treatments are usually scheduled every 6–8 weeks over the course of several months to a year depending upon the size, location, and responsiveness of the malformation to the therapy.
Despite the high rates of recurrence (25–52 %), surgical excision has been historically used as the treatment of choice for vascular malformations [69]. Traditionally, surgical resection was effectively used for encapsulated and small lesions. When a malformation is diffuse and multifocal, the surgical approach is relatively contraindicated as damage to major vital structures and massive hemorrhage may ensue. For larger lesions, complete surgical resection might not be possible and multiple partial surgical resections may be required. Partial surgical resections are associated with higher recurrence rates. Encapsulated, circumscribed, localized lesions and lesions composed of numerous small venous channels are the most amenable to surgical resection.
Since its introduction, sclerotherapy has been used as an effective alternative to surgery in the treatment of LFVM. Currently there are numerous agents available for sclerotherapy including ethanol, sodium tetradecyl sulfate (STS), polidocanol, ethibloc, and bleomycin. The most commonly used agents are ethanol and STS (in the United States) and polidocanol (in Europe), although the use of polidocanol in the United States has been increasing since its approval by the Food and Drug Administration (FDA) in March of 2010. Although proven to be effective [70, 71], ethanol sclerotherapy (ES) is associated with limitations and major side effects (local and systemic), including severe pain requiring general anesthesia, ethanol toxicity, and local tissue damage, and its use in pediatric patients remains controversial. In a study of 71 patients, Mason et al. demonstrated that patients who received up to 1 mL/kg of ethanol during ethanol embolization or sclerotherapy may have elevated serum ethanol levels that could be associated with increased risk of respiratory depression, cardiac arrhythmias, seizures, rhabdomyolysis, and hypoglycemia [72]. In a study of 98 sessions of ES in 30 CVM patients, Lee et al. documented complications in 26.7 % of patients which ranged from mild to severe and acute to delayed [73]. Authors reported nine cases with ischemic bullae, two with tissue fibrosis, two with tissue necrosis, one with deep venous thrombosis (DVT), one with pulmonary embolism, five with nerve palsy, and four cases of transient pulmonary pressure elevation. Of the five nerve palsies, one (affecting the peroneal nerve) was permanent. Other studies reported episodes of transient bradycardia and cardiac arrest during the treatment with ES [74]. Ethanol sclerotherapy can also result in transmural vessel necrosis, massive swelling (sometimes resulting in compartment syndrome), central nervous system (CNS) depression, hypertension, and pulmonary vasospasm [75, 76]. Some practitioners recommend continuous pulmonary pressure monitoring during ES and avoiding ethanol in regions adjacent to nerves such as the facial nerve or the sympathetic plexus in cervical lesions [76].
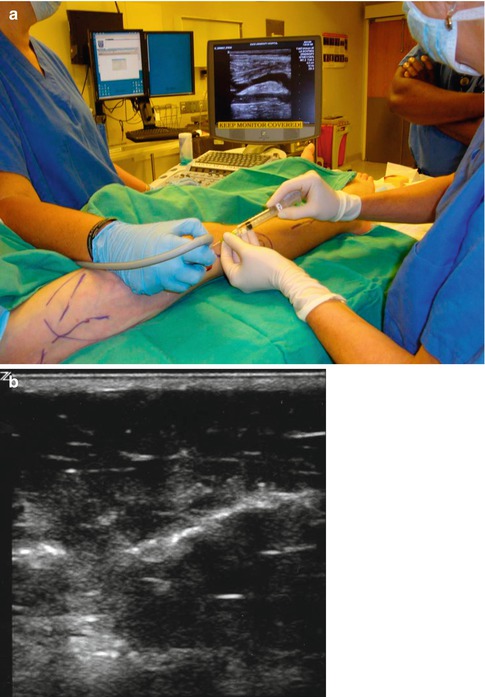
Since liquid sclerosants become diluted and inactivated by intralesional blood, the use of sclerosants in microfoam form significantly improves the procedure for LFVM [77–79]. The foam bubbles displace intralesional blood (preventing the sclerosant from becoming diluted) and achieve maximal effective exposure between the sclerosing agent and the endothelial lining. In addition, the echogenicity of the bubbles makes them visible on US surveillance making the procedure easier to perform (Figs. 17.8a, b). Foam treatments, in contrast to ES, can be given on a strictly ambulatory basis as they are minimally painful.