Order
Family
Genus
Species
Mucorales
Syncephalastraceae
Syncephalastrum
Syncephalastrum racemosum
Saksenaeaceae
Apophysomyces
Apophysomyces elegans complex
Apophysomyces trapeziformis
Saksenaea
Saksenaea vasiformis complexa
Saksenaea erythrospora b
Cunninghamellaceae
Cunninghamella
Cunninghamella bertholletiae
Mucoraceae
Absidia
Absidia coerulea c
Actinomucor
Actinomucor elegans
Mucor
Mucor circinelloides (Rhizomucor variabilis)
Mucor hiemalis
Mucor indicus
Mucor racemosus c
Mucor ramosissimus
Rhizomucor
Rhizomucor pusillus
Rhizopus
Rhizopus azygosporus
Rhizopus microsporus
Rhizopus oryzae (Rhizopus arrhizus)
Rhizopus schipperae
Rhizopus stolonifer c
Thamnidiaceae
Cokeromyces
Cokeromyces recurvatus
Lichtheimiaceae
Lichtheimia (former Absidia)
Lichtheimia corymbifera (Mycocladus corymbifer, Absidia corymbifera)
Lichtheimia ramosa (Absidia ramosa)
Human pathogenic species principally belong to the genera Rhizopus, Mucor and Lichtheimia (Table 12.1). Rhizopus species predominate, accounting for around 50% of cases, with R. oryzae (syn: R. arrhizus) being the most common clinical isolate reported, followed by R. microsporus var. rhizopodiformis. Mucor and Lichtheimia account for a further 20–30% of reported cases of mucormycosis (Gomes et al. 2011; Reiss et al. 2012).
Other genera, such as Rhizomucor, Cunninghamella, Apophysomyces and Saksenaea cause human infection to a much lesser extent. Cokeromyces recurvatus and Syncephalastrum racemosum have been described as the causative agent in only a small percentage of cases. However, the possibility of incorrect speciation in reported cases cannot be ignored, especially because of the complexities of identification, together with the changing nomenclature of these fungi. Additionally, cases of the more unusual species may be underestimated due to incomplete or incorrect identification (Gomes et al. 2011). Regardless of causative species, the disease process and diagnostic strategies are similar.
III. Habitat and Sources of Mucorales
The species isolated from cases of human and animal infection are thermotolerant, and many are ubiquitous in the soil and on decomposing organic matter. These moulds are found in the indoor and outdoor air, on food items and in dust. The environmental microbiology literature provides a limited insight into how common the Mucorales are in the environment and provides few clues about which ecological niches these fungi are found in (Richardson 2009). In particular, examination of numerous air sampling surveys in indoor and outdoor environments might indicate the level of exposure or help explain the apparent seasonality of mucormycosis. Similar analyses of other environments might reveal specific point sources of fungal communities. These and other environmental issues have been reviewed previously (Richardson 2009) but are summarised here.
A. Indoor and Outdoor Environments
Sporangiospores released by the Mucorales range from 3 to 11 μm in diameter, are easily aerosolised and readily dispersed throughout the environment. This is the major mode of transmission. Spores can also be carried by insects, especially flies. However, there are very little data concerning the levels of mucormycete sporangiospores in outdoor and indoor air, especially in geographical areas where mucormycosis is particularly prevalent. The numbers of airborne mucormycete sporangiospores appear to depend on the climatic conditions that favour growth and dispersal.
A survey of indoor and outdoor air in and around 17 homes in Cincinnati, Ohio using mould-specific quantitative PCR failed to detect any pathogenic Mucorales (Meklin et al. 2007). During a 2-year air sampling survey in Barcelona, Spain, the following genera were found in decreasing order: Aureobasidum, Rhizopus, Mucor, Arthinium, Phoma, Fusarium, Trichoderma and Botrytis (Calvo et al. 1980). A 1-year aeromycological study was conducted in the area of Zagreb, in order to establish seasonal variations in the composition and concentration of aeromycota (Klaric and Pepeljnjak 2006). Sampling was carried out at three locations at regular intervals. Airborne fungi peaked during spring and summer (110–284 cfu/m3), whereas lower levels were detected in autumn and winter at each of the sampling sites (6–128 cfu/m3). In contrast to Cladosporium, Pencillium and Alternaria, very low levels of Mucor and Rhizopus were found.
In what appears to be the largest study of airborne indoor and outdoor fungal species and concentrations to date, Shelton and colleagues (2002) examined 12,026 fungal air samples (9,619 indoor samples and 2,407 outdoor samples) from 1,717 buildings located across the USA; these samples were collected during indoor air quality investigations performed from 1996 to 1998. For all buildings, both indoor and outdoor air samples were collected. The culturable airborne fungal concentrations in indoor air were lower than those in outdoor air. The fungal levels were highest in autumn and summer and lowest in the winter and spring. Geographically, the highest fungal levels were found in the Southwest, Far West and Southeast. The most common culturable airborne fungi, both indoors and outdoors and in all seasons and regions, were Cladosporium, Penicillium, non-sporulating fungi and Aspergillus. Stachybotrys chartarum was identified in the indoor air in 6% of the buildings studied and in the outdoor air of 1% of the buildings studied. Mucor (not specified), Rhizopus (not specified) and Cunninghamella were detected in indoor air more than outdoor air, although this varied from one geographical area of the USA to another. However, agents of mucormycosis were not included in the category of “common fungal types”.
A comparison of populations of mould species in homes in the UK and USA using mould-specific quantitative PCR did not detect any appreciable level of Mucorales in dust samples (Vesper et al. 2005). Mucormycetes do not appear to be common in buildings (Hyvärinen et al. 2002), suggesting that the various building materials used in house construction do not support the growth of these fungi, compared to the wide profile of deuteromycetes found on damp substrates. A significant proportion of private residences, offices and work places are known to be damp; estimates range from 20 % to 50 %. Numerous publications have reported the moisture levels required for growth of fungi on construction, finishing or furnishing materials. Rhizopus species have a high moisture requirement for growth and are classified as hydrophilic. Members of the Mucorales are non-cellulolytic microorganisms and do not have any enzymatic activity, even against the most susceptible forms of cellulose, and therefore are not very likely to be found on building materials. All of these observations suggest that house residents are not generally exposed to Mucormycetes in their home environment, apart from mould-contaminated food items such as bread and fruit. This notion is supported by the absence of Mucormycetes from indoor air sampling surveys (Shelton et al. 2002).
B. Soil and Composting Vegetation
The simplistic view is that the agents of mucormycosis are found in the soil, in composting vegetation, on rotting fruit, in dust, during heavy excavation and construction, and in air-conditioning filters. The elevated temperatures found in composting vegetation are selective for thermophilic species, such as some species of Lichtheimia, Mucor, Rhizopus and Rhizomucor. In general, these fungi are unable to utilise cellulose and lignin. They are characterised by rapid germination. Some species are found growing on dung.
At present, there are approximately 3,300 species of currently known soil fungi. Very few studies have identified Mucorales at particular sites in areas where cases of mucormycosis have been reported. For many years there has been the view that most soil fungi were cosmopolitan and that species at a particular site were only selected by various soil parameters. It has also been the view that most fungal species would potentially spend part of their life in the soil. This view has now been modified considerably as very many plant-parasitic species are never isolated from the soil. This is particularly pertinent when considering the Mucorales.
In general, composting is considered to be an aerobic process, suggesting high biological activity. It has been clearly demonstrated that the rise in temperature and the decomposition of composting plant materials is brought about by thermophilic microorganisms, including fungi (Ingold 1978). Temperature and changes in the available food supply probably exert the greatest influence in determining the species of organism comprising the population in a compost stack at any one time. Fungi, including Aspergillus fumigatus and Mucormycetes play an important role in the decomposition of cellulose, lignin and other more resistant materials, despite being confined primarily to the outer layers and becoming active only during the latter part of the composting period (Ingold 1978).
Many of the opportunistic Mucorales are typical inhabitants of natural composts, tropical soils and other heated materials. Indoor sites particularly associated with these fungi therefore may be those where humid organic material is exposed to heat, most notably within poorly maintained heating ducts and attached humidifier structures, in soils of potted plants, especially those placed in warm locations, and in indoor composts (James et al. 2006).
Rhizopus oryzae and Lichtheimia corymbifera (Absidia corymbifera) were found in potted plant soil in a haematology unit where a leukaemia patient developed invasive mucormycosis (Ribes et al. 2000). They were also abundantly represented in bird dung accumulating in an aviary (Ribes et al. 2000). In hospitals, wooden materials such as tongue depressors have repeatedly been a source of problematic Rhizopus inoculum (Ribes et al. 2000). Rhizopus oryzae has been isolated from grains, onions, various nuts and stored seed potatoes (Ribes et al. 2000).
A number of studies mention soil contamination of areas of soft-tissue damage followed by mucormycosis, apparently in immunocompetent patients (Vainrub et al. 1988). Traumatic inoculation of spores can lead to extensive necrotic cutaneous infections. This form of disease is most often seen in patients with burns and other forms of local trauma, such as traumatic implantation of sporangiospores in dirt, needle-stick injuries, illicit drug injection sites, tattooing, insect bites and stings. Disruption of the cutaneous barrier seems to be a prerequisite for acquiring cutaneous mucormycosis, with region-specific Mucormycetes being isolated from soil at the site of trauma as well as from the patient.
An illustrative case is where a patient who, whilst attempting to extinguish presumably burning clothes, rolled in moist soil that was found to be contaminated with Apophysomyces elegans (Cooter et al. 1990). Apophysomyces elegans was first isolated from soil samples collected from a mango orchard in northern India (Misra et al. 1979). In diabetic or immunosuppressed patients, cutaneous lesions may arise at an insulin injection site or a catheter insertion site. Cutaneous mucormycosis is an aggressive disease. It can lead to necrotising fasciitis or to widespread disseminated infection.
Biological soil crusts are the community of organisms living at the surface of desert soils. Major components are cyanobacteria, green algae, microfungi, mosses, liverworts and lichens. Mucormycetes do not appear to survive in this environment. Species of Mucormycetes have been cultured from geothermal soils in Yellowstone National Park, including species of Absidia and Cunninghamella, especially in close proximity to the perennial plant Dichanthelium lanuginosum, the hot springs panic grass (Redman et al. 1999).
C. Natural Disasters
Natural disasters have also revealed specific environmental niches of Mucormycetes. This was illustrated particularly by the Asian tsunami. Most of the tsunami survivors who experienced near-drowning events remained in unclean and traumatic conditions without receiving any immediate medical care for several hours. Patients lay for several hours or days in warm and stagnant water; normally poorly virulent environmental bacteria, fungi and amoebae found the ideal conditions to colonise open wounds and bone fractures and disseminate to other body sites. Many survivors presented with necrotising fasciitis, which is a soft-tissue infection associated with infarction of the dermis and subcutaneous tissue. The cause of this condition is usually polymicrobial but cutaneous mucormycosis is recognised as a cause of necrotising fasciitis since agents of mucormycosis exhibit vasculotropism, which is important in the pathophysiology of this condition, with the endpoint being ischaemic necrosis of the affected tissue (Snell and Tavakoli 2007).
The following case is illustrative (Snell and Tavakoli 2007). A survivor of the Asian tsunami with soft-tissue injuries and bilateral displaced fractures of her inferior and superior pubic rami was found to be infected with Apophysomyces elegans. Liposomal amphotericin was commenced and a total course of 42 days was maintained. After surgical débridement, topical negative pressure dressings, nursing care and antifungal therapy, split skin grafting was utilised to cover the resulting defect, with good result. It was assumed that the affected areas had become infected with the fungus in contaminated water or soil as a result of destruction of the urban infrastructure by the tsunami.
Tornados and hurricanes have been the cause of cutaneous infection by Mucorales species. Neblett Fanfair and colleagues (2012) describe a large cluster of cases of mucormycosis, with 13 Apophysomyces trapeziformis infections in persons injured during a tornado in Joplin, USA. Morbidity and mortality among the case patients were substantial; the primary risk factors for infection were penetrating trauma. These findings suggest that clinicians should consider environmental fungi as potential agents of soft-tissue infections in injured patients after natural disasters.
D. Other Sources of Mucormycetes
Even though many previous reviews on the epidemiology of mucormycosis highlight the growth and isolation of the Mucorales on intact and rotting fruit, bread and other food items (Ribes et al. 2000), there are very few systematic surveys. It is apparent from the recent literature at least that the Mucorales do not constitute a major part of the mycoflora of food-handling facilities.
IV. Exposure Pathways
A. Exposure Through Colonisation of Nasal Mucus
Because of their rapid growth and prolific spore production, inhalation of sporangiospores must be a daily occurrence (Sugar 2005). It is known that the sporangiospores of some Mucorales in indoor environments have a very low settling rate and remain airborne for a long time. Airborne fungal spores are almost ubiquitous and can be found on all human surfaces in contact with air, especially on upper and lower airway mucosa. Some 15,000–170,000 spores are inhaled over a 24 h period by an adult person, depending on environmental factors. Regional and climatic factors contribute to exposure. Fungal spores impacted on the mucus of airway mucosa are cleared by mucociliary transport. Therefore, it is surprising that members of the Mucorales are very rarely found in nasal mucus. Nasal mucus is produced by the nasal mucosa, and mucus lining the airways (trachea, bronchus, bronchioles) is produced by specialised airway epithelial cells (goblet cells) and submucosal glands. Small particles such as dust, particulate pollutants and allergens as well as infectious agents such as bacteria become caught in the viscous nasal or airway mucus.
Do mycological studies of the biodiversity of the nasal mucus help? The nose is a very efficient air sampler, and analysis of the nasal mucosa should reflect the air spora of the environment where a patient has been residing and working. In one study conducted in Austria, nasal mucus samples from patients suffering from chronic rhinosinusitis and from healthy persons were monitored over 28 months (Buzina et al. 2003). Mucus samples were obtained by flushing the noses of patients with saline or by endoscopic sinus surgery. Fungi from mucus were cultured and identification was performed microscopically and by polymerase chain reaction (PCR) with subsequent sequencing of the ribosomal internal transcribed spacer region. Altogether, 619 strains of fungi were cultivated from 233 subjects. Eighty-one species were identified, with a maximum of nine different species per person. The most prevalent isolates belonged to the genera Penicillium, Aspergillus, Cladosporium, Alternaria and Aureobasidium. Interestingly, Mucormycetes were found in only two subjects: in one, Rhizopus oryzae, and in another Cunninghamella bertholletiae.
Mucorales are found occasionally in water-damaged buildings, as demonstrated by air sampling and analysis of settled dust by quantitative PCR. However, inhalation of sporangiospores in dust has been linked to outbreaks of rhinocerebral or pulmonary mucormycosis linked to excavation, construction or contaminated air-conditioning filters. Some reports suggest that the lower airways are colonised with agents of mucormycosis (Connolly et al. 1999). Nevertheless, in patients with appropriate underlying conditions, the isolation of Mucorales species, even from non-sterile samples, is generally regarded as potentially significant (Torres-Narbona et al. 2008).
B. Exposure Through the Gastrointestinal Tract
Ingestion of contaminated food materials, such as fermented milk, fermented porridge, herbal/homeopathic remedies, and the use of contaminated tongue depressors are recognised exposure risks for gastrointestinal mucormycosis. Gastrointestinal mucormycosis is a rare condition that has usually been encountered in malnourished infants or children. Persistent ingestion of non-nutritional substances, gastric ulcers, severe systemic illness, age extremes and systemic immunosuppression are additional predisposing conditions for gastrointestinal tract mucormycosis.
C. Contaminated Medical Devices
Whereas most infections with Mucormycetes are community acquired, nosocomial acquisition, both sporadic cases and pseudo-outbreaks, have been linked with contaminated bandages, needles and tongue depressors used to construct splints for intravenous and arterial cannulation sites in preterm infants (Ribes et al. 2000). Percutaneous routes of exposure are very important in causing infection by Mucormycetes. Traumatic implantation of spores in dirt has been seen in a number of patients. Needle-stick exposures have been implicated in mucormycotic infections occurring at the site of medicine injection, catheter insertion sites, injection sites for illicit drug use and tattooing. Insect bites or stings have also been implicated in disease transmission in cases of cutaneous and subcutaneous mucormycosis. The development of wound mucormycosis has been seen with a variety of adhesive products used in the hospital setting.
D. Exposure in Hospital Environments
Hospital-acquired mucormycosis, both sporadic cases and pseudo-outbreaks, has been reported but is rare. Fifteen hospital outbreaks or clusters of infection are described in the English literature between 1966 and 2010, from the USA, Canada, China and Europe. These included cutaneous, pulmonary, rhinocerebral, gastrointestinal and disseminated infections. Rhizopus was identified as the cause in the majority of cases, although Rhizomucor, Lichtheimia, Cunninghamella and Mucor have also been implicated (Antoniadou 2009; Gomes et al. 2011).
The most common route of entry was via contact with contaminated objects, and sources included non-sterile bandages and adhesive dressings, and wooden tongue depressors used to prepare oral medications administered via nasogastric tubes, as well as to construct splints for intravenous and arterial cannulation sites in preterm infants. Less frequently, inhalation was the route of exposure, and cases have been linked to construction or renovation work, contaminated ventilation systems and water damage to a linen store and patient’s shower room. Corn-starch used in the manufacturing of allopurinol tablets and commercial ready-to-eat food was implicated in an outbreak of intestinal mucormycosis (Antoniadou 2009; Richardson 2009; Gomes et al. 2011).
V. Epidemiology
A. Modes of Transmission
Mucorales are thermotolerant fungi, generally reported in the literature to enjoy a global distribution, being common inhabitants of soil, found on plants and in decomposing organic matter. They are also found in the indoor environment, in dust and on foodstuffs. Such fungi are common plant pathogens and contaminants of grain, fruit and bread (Richardson 2009). Mucorales infections are also reported worldwide.
Mucorales fungi have efficient dispersion mechanisms, via sporangiospore release from sporangia. Each sporangium can contain between hundreds and hundreds of thousands of spores, depending on species, and when maturation is reached, the sporangium wall dissolves, discharging the spores into the environment (Fig. 12.1). Most species produce aerial growth only when the surrounding air is extremely damp. In such species, a splash dispersal mechanism is used, whereby water passes into the spore-mass through the columella as the sporangium wall dissolves, resulting in a “sporangial drop”. Other species can form sporangiospores in dry air, producing powdery spores that are readily aerosolised and dispersed by low-speed winds. The foraging of small animals also aids the dissemination of spores (Richardson 2009).
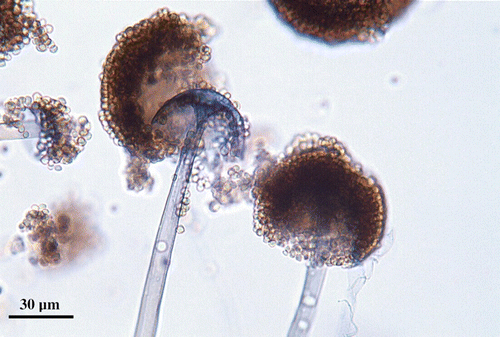

Fig. 12.1.
Microscopic morphology of Rhizopus oryzae
Such dispersal mechanisms suggest that humans must be exposed to these fungal spores on a regular basis, both via inhalation and topical exposure. Inhalation from environmental sources is regarded as the main portal of entry into the body, and certainly the nasal sinuses and lungs are the commonest initial sites of infection. However, despite such repeated contact, the frequency of human disease is relatively low and directly correlates with the immune status of the host. Studies suggest that those without properly functioning phagocytes are at increased risk of disease (Ibrahim et al. 2012; Roilides et al. 2012). Neutropenic individuals are therefore the predominant sufferers of mucormycosis. Less commonly, infection can also occur following cutaneous or percutaneous injuries. Intact skin is an effective anatomical barrier to infection; conversely, spores can be directly inoculated into abraded skin, often following trauma, or into occluded skin or mucosa as a result of contaminated dressings. Needle-stick exposures after medicinal or illicit drug injections, catheter insertion, or tattoos have been reported. Ingestion of fungal elements from contaminated foodstuffs is a rare cause of gastrointestinal disease (Ribes et al. 2000; Richardson and Warnock 2012; Liao et al. 2013).
B. Prevalence of Infection
Mucormycosis remains the second most common mould infection in immunocompromised individuals, after aspergillosis, although it is still regarded as relatively rare (Neofytos et al. 2013). Roden and colleagues reviewed the English language literature from 1885 to the 1990s, discovering 929 cases of confirmed mucormycosis (Roden et al. 2005). Estimates of prevalence in the USA, based on a population-based surveillance study during 1992 and 1993, indicated about 1.7 cases/million population, or approximately 500 cases per year (Rees et al. 1998). Some European and North American studies suggest an increase in such infections during the last decade, especially in cancer centres (Marr et al. 2002; Lass-Flörl 2009a; Waness et al. 2009), whereas other surveys do not support this (Torres-Narbona et al. 2008; Prasad et al. 2012).
Spanish laboratories report an isolation rate of 6 per 100,000 samples, with no significant increase over a decade (Torres-Narbona et al. 2008). Analysis of paediatric infections between 2003 and 2010 in the USA found no evidence of an increase in mucormycosis (Prasad et al. 2012). In contrast, in France, population-based surveillance, conducted between 1997 and 2006, showed an increase in incidence from 0.7 to 1.2 cases per million population per annum. Most of the increase was accounted for by infections among haematopoietic stem cell transplant (HSCT) recipients or persons with haematological malignancies (Richardson and Warnock 2012). In the USA, an increase in the frequency of mucormycosis has been reported since the mid-1990s, and is now identified in up to 6.8% of autopsies (Brown 2005).
A contrast in worldwide epidemiology appears to have arisen. While mucormycosis remains relatively rare in developed countries, primarily affecting haematology patients, in developing countries, especially India, the incidence of Mucorales disease is rapidly increasing and is predominantly related to an increase in individuals with uncontrolled diabetes (Meis and Chakrabarti 2009). This association has also been reported in Japan and Taiwan (Slavin and Chakrabarti 2012).
Reasons suggested to explain this observed global rise in cases of mucormycosis include changes in transplantation procedures, an increased use of immunosuppressive drugs, improved diagnostic ability, increased incidence of diabetes mellitus, and the introduction of antifungal agents, including voriconazole and caspofungin, with no Mucorales activity (Brown 2005; Cuenca-Estrella et al. 2009; Waness et al. 2009).
Five main clinical manifestations are encountered, varying in frequency: rhino-orbital-cerebral (craniofacial; 39%), pulmonary (24%), cutaneous (19%), gastrointestinal and disseminated disease (Roden et al. 2005). These different clinical forms are associated with particular underlying disorders (see Section V, D). Risk of multi-organ dissemination varies, depending on the underlying disease and immune status of the host; rates in the literature range from 3% to >50%.Dissemination has been reported in patients with cerebral (48% of cases), cutaneous (39%) and pulmonary (20%) disease (Garcia-Hermoso et al. 2011; Sun and Singh 2011; Richardson and Warnock 2012). Other presentations include renal, cardiac and bone disease (Gomes et al. 2011; Liao et al. 2013).
C. Is Mucormycosis a Seasonal Disease?
Mucormycosis has not previously been regarded to have a seasonal pattern. Seasonal variations in the atmospheric concentration of fungal spores for some moulds in several geographical locations have been reported. In the USA, the highest levels were found in the autumn and summer and lowest in the winter and spring but agents of mucormycosis were not commonly found (Shelton et al. 2002). Seasonal peaks of Aspergillus conidia and other moulds have been reported from Europe and the USA (Ribes et al. 2000) but little data exist on the seasonal frequency of the Mucorales or the seasonal prevalence of mucormycosis. Only one study by Talmi and colleagues (2002) reported dates of onset of symptoms in 19 patients in Israel with rhino-orbital and rhino-orbito-cerebral mucormycosis and those appeared to vary seasonally but did not establish statistical significance. A more recent study from the Lebanon suggested a clustering of onset of mucormycosis at the end of a dry, warm period that lasted from May to October (Al-Ajam et al. 2006).
D. Risk Factors
Mucormycosis predominantly affects immunocompromised patients. The main predisposing factors, pathogenic mechanisms and clinical presentations are summarised in Table 12.2. Ultimately, host factors play the most important role in the development of Mucorales disease. Patients most at risk are those with prolonged or profound neutropenia, poorly controlled diabetes with or without ketoacidosis, other metabolic acidosis, neonatal prematurity and extensive skin lesions following trauma or burns. Malnutrition, long-term corticosteroid use, illicit intravenous drug use, iron overload with or without desferrioxamine iron chelation therapy and use of broad-spectrum antifungal prophylactic agents are also described as risk factors (Lass-Flörl 2009a; Petrikkos and Drogari-Apiranthitou 2011; Richardson and Warnock 2012; Liao et al. 2013). Other possible predisposing factors include those with HIV infection, chronic infections such as tuberculosis, septicaemia with multi-organ failure, use of antibiotic therapy, chronic renal failure and sarcoidosis (Waness et al. 2009; Garcia-Hermoso et al. 2011).
Table 12.2
Predisposing conditions for mucormycosis, pathogenic mechanisms and clinical presentation
Predisposing conditions for mucormycosis | Pathogenic mechanism(s) | Clinical presentation forms (in order of frequency) |
|---|---|---|
Haematological malignancies | Prolonged neutropenia | 1. Pulmonary, rhinocerebral |
Haematopoietic stem cell transplantation | 2. Cutaneous | |
3. Sino-orbital | ||
Diabetic ketoacidosis | Impairment of neutrophil activation (functional neutropenia)/Fe usage by Mucormycetes for growth | 1. Rhinocerebral |
Uncontrolled diabetes mellitus | 2. Sino-orbital | |
3. Pulmonary | ||
4. Cutaneous | ||
Prolonged treatment with corticosteroids | Defects in macrophages and neutrophils, corticosteroid induced diabetes, hypocomplementaemia | 1. Disseminated |
2. Renal | ||
Autoimmune disease | 3. Cutaneous | |
4. Rhinocerebral | ||
5. Gastrointestinal | ||
Solid organ transplantation (SOT)/graft-versus-host disease | Cellular immune suppression, corticosteroid induced diabetes | 1. Pulmonary |
2. Sinus | ||
3. Cutaneous | ||
4. Rhinocerebral | ||
5. Disseminated | ||
HIV infection/intravenous illicit drug use | Injection of spores contained in drugs | 1. Cerebral |
2. Cutaneous | ||
3. Renal, heart | ||
4. Rhinocerebral | ||
5. Disseminated | ||
Iron overload | Fe usage by Mucormycetes for growth | 1. Disseminated |
2. Pulmonary | ||
Iron/aluminium chelation therapy with desferrioxamine (DFO) | Fe-DFO action as siderophore | 3. Rhinocerebral |
4. Cerebral | ||
5. Cutaneous
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|