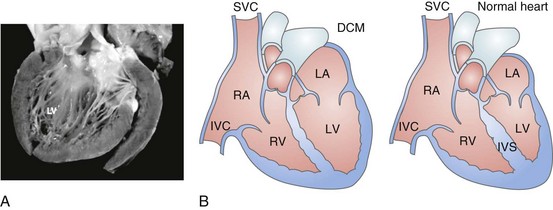
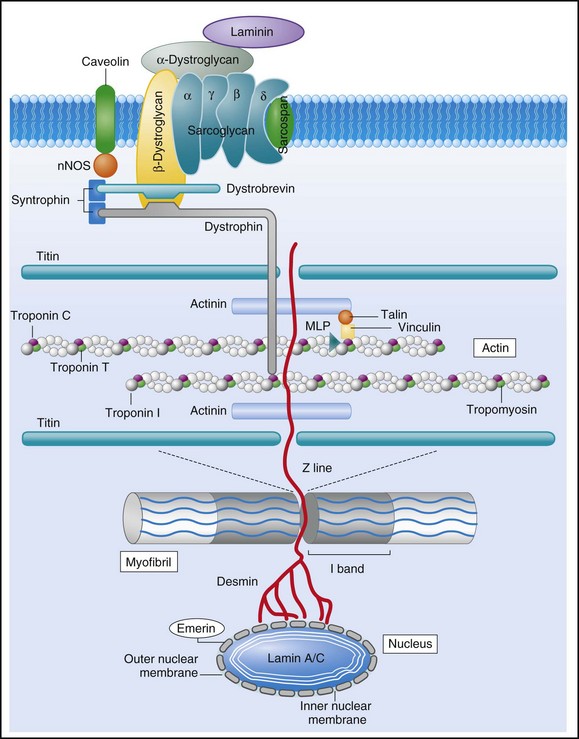
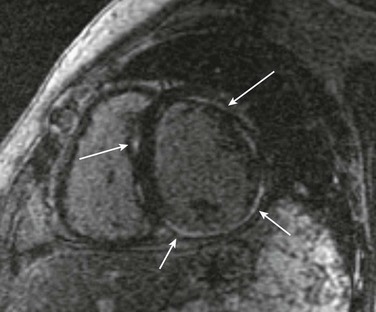
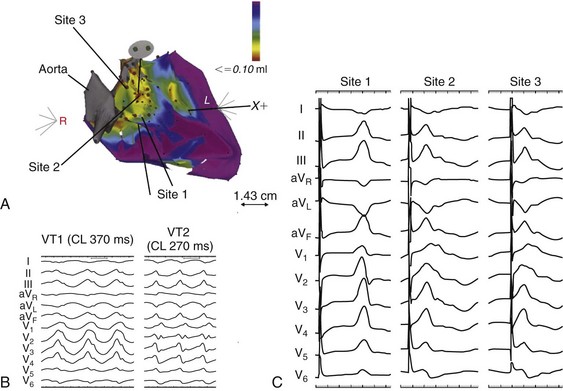
85 Dilated cardiomyopathy (DCM) is a disorder characterized by left or biventricular dilatation and impaired systolic function (Figure 85-1), frequently resulting in congestive heart failure (CHF). Valvular heart disease, excess ethanol ingestion, hypertension, pregnancy, and infections are common underlying etiologic factors. In idiopathic dilated cardiomyopathy, by definition an etiology has not been determined, although genetic, autoimmune, viral, and metabolic causes have been implicated. A common feature of DCM regardless of the underlying cause is a propensity to ventricular arrhythmias and sudden death. This chapter focuses on incidence and survival, mechanisms of ventricular arrhythmogenesis, predictors of sudden death, and management of ventricular arrhythmias in DCM. For the purpose of this chapter, DCM refers to idiopathic dilated cardiomyopathy, particularly excluding cardiomyopathies related to coronary artery disease, unless otherwise stated. Figure 85-1 Dilated cardiomyopathy. IVC, Inferior vena cava; IVS, intraventricular septum; LA, left atria; LV, left ventricle; RA, right atria; RV, right ventricle; SVC, superior vena cava. Congestive heart failure afflicts approximately 5.8 million individuals in the United States.1 The incidence rises steadily with age; there is a prevalence in men that increases from 8 per 1000 at 50 to 59 years old, to 66 per 1000 at 80 to 89 years old. CHF is anticipated to be an even larger societal burden as the population ages.2 Mortality rates after a diagnosis of CHF have steadily improved since the 1980s with the widespread use of neurohormonal medical therapy, such as angiotensin-converting enzyme (ACE) inhibitors and β-blockers, and with interventions such as implantable cardioverter defibrillators (ICDs) and biventricular (BiV) pacing. However, the overall mortality from CHF remains high in the face of the rising incidence of disease with an overall adjusted survival inclusive of both ischemic and nonischemic cardiomyopathy of 48% at 5 years.3 Survival rates in DCM are principally determined by the severity of symptoms and the degree of left ventricular (LV) dysfunction in the cohort being examined.4 It is difficult to estimate the survival in dilated cardiomyopathy alone, because establishing the etiology of heart failure from population-based studies is difficult and varies widely depending on diagnostic criteria, case ascertainment, and geographic location. However, DCM accounts for a substantial proportion of the population of patients with heart failure, with only 24% of women and 32% of men having any documented coronary disease among a cohort of patients with heart failure in Olmsted County, Minnesota.3 After accounting for other contributors such as valvular, ischemic, and hypertensive heart disease, another large cohort estimated DCM to account for 32% of cases.5 After accounting for comorbidities, the prognosis of DCM compared with ischemic cardiomyopathy appears to be slightly improved. Approximately 40% of patients with DCM have a familial contribution to their disease, most of which appears to be inherited in an autosomal dominant fashion; however, X-linked, autosomal recessive, and mitochondrial inheritance are also seen. Advances have been made in the last 20 years in the identification of candidate genes responsible for DCM (Figure 85-2). Mutations in more than 40 candidate genes have been reported in patients with DCM, and it is reported that up to 40% of patients with DCM may have inherited this disorder.6 The more common autosomal dominant form of familial DCM also has two main forms: “pure” DCM and DCM associated with cardiac conduction system disease. Mitochondrial inheritance is seen most often in childhood forms of familial DCM, whereas X-linked and autosomal recessive forms seem to be distributed evenly between childhood and adult forms of disease. In the case of X-linked forms of DCM, two disorders have been well characterized: X-linked cardiomyopathy, which appears in adolescents and young adults, and Barth syndrome, which is most frequently identified in infancy. Figure 85-2 Schematic of proteins implicated in the pathogenesis of dilated cardiomyopathy. Identified genes that result in dilated cardiomyopathy include the cytoskeletal protein-encoding genes b- and d-sarcoglycan in the sarcolemma, dystrophin, and the intermediate filament protein-encoding genes desmin and laminin A/C. Sarcomeric protein-encoding genes actin, b-myosin heavy chain, a-tropomyosin, and cardiac troponin T cause either dilated cardiomyopathy or hypertrophic cardiomyopathy, whereas cardiac troponin I, titin, and the myosin light chains cause hypertrophic cardiomyopathy. Mutations in dystrobrevin cause the intermediate phenotype, left ventricular noncompaction. MLP, Muscle LIM protein; nNOS, neuronal nitric oxide. In the case of pure DCM, linkage analysis has identified a number of candidate genes in which mutations have subsequently been found, including those that code for actin (chromosome 15q14),7 desmin (2q35),8 β-sarcoglycan (4q12),9 δ-sarcoglycan (5q33),10 cardiac troponin T (1q32), β-myosin heavy chain (chromosome 14q11),11 and α-tropomyosin (15q2).12 So far, DCM with conduction system disease candidate genes have included lamin A/C (1q21.2-3)13 and the sodium channel gene SCN5A (3p22–25).14 Recent data have shown increased susceptibility to arrhythmic events in patients with DCM and lamin A/C–related mutations.15 First described in 1987 as a form of DCM occurring in teenage boys and men in their early 20s, with rapid progression from CHF to death or transplantation, X-linked cardiomyopathy is distinguished by increased amounts of serum creatine kinase muscle isoforms, a sign of underlying skeletal muscle disease.16 Female carriers tend to develop mild to moderate DCM in their 50s, and the disease is slowly progressive. Towbin et al.17 identified a disease-causing gene that codes for dystrophin, a cytoskeletal protein. The relationship between individual genotypes and arrhythmogenicity is poorly understood. As more putative genes have been identified, genotyping has become possible; however, this has not become an important tool in determining the risk for ventricular arrhythmias or in the management of affected individuals. As with all proposals for genetic testing, certain caveats apply, such as relating to the possibility of uncertain results (either a negative screening result in someone at risk for ventricular arrhythmias or a positive result in the identification of a mutation of unclear significance for that particular individual). No single mechanism is responsible for ventricular arrhythmias in DCM; rather, multiple factors clearly contribute (Box 85-1). At autopsy, extensive subendocardial scarring in the LV has been described in 33% of patients, and multiple patchy areas of replacement fibrosis have been described in 57% of patients with DCM.18 These autopsy data correspond well with findings observed during voltage mapping of the LV and can be observed on a cardiac magnetic resonance imaging (MRI) examination (Figure 85-3). It is apparent that these areas can act as sites for reentry, one of the commonest mechanisms for ventricular tachycardia (VT) and sudden cardiac death (SCD) in patients with DCM. Ischemia, through smaller coronary artery thrombi or emboli and electrolyte imbalances (especially hypokalemia and hypomagnesemia), can also play a role in arrhythmogenesis in DCM and in other patients at risk for VT and SCD. Alterations in ventricular mechanics and geometry can predispose to reentrant arrhythmias through variable wall tension and stretch-dependent shortening of ventricular refractoriness, or by resulting in abnormal automaticity or triggered activity. Increased circulating catecholamines can cause arrhythmias indirectly by driving potassium intracellularly or directly through triggered activity. Finally, antiarrhythmic drugs themselves can exert a proarrhythmic effect in patients with DCM. Figure 85-3 A magnetic resonance image for a patient with dilated cardiomyopathy (DCM). This delayed gadolinium enhancement image illustrates patchy areas of fibrosis at the base of the LV in the mid myocardium, with no coronary distribution typical of a DCM. Arrows indicate areas of delayed enhancement corresponding to scar. Although other mechanisms of VT are common in patients with DCM, bundle branch reentry VT (BBR-VT) is perhaps the most characteristic (Figure 85-4). It has been observed that 45% of all patients who had BBR-VT induced had underlying DCM. Bundle branch reentry produces VT through a macroreentrant circuit involving the His-Purkinje system, usually with antegrade conduction over the right bundle branch and retrograde conduction over the left bundle branch. In one series, it was the mechanism responsible for VT in up to 6% of all patients and in up to 41% of patients with DCM.19 BBR-VT is usually rapid, with a mean cycle length of 280 ms. Not surprisingly, syncope occurs in the majority of such patients. Degeneration to ventricular fibrillation can also occur. Baseline electrocardiography typically shows a nonspecific intraventricular conduction delay or left bundle branch block (LBBB) morphology. H-V intervals recorded during sinus rhythm are characteristically prolonged. Tachycardias induced by right ventricular (RV) stimulation typically show a LBBB morphology. A short-to-long change in cycle length before the premature extrastimulus has been reported to be particularly successful at initiating BBR-VT. In patients with DCM coming for an electrophysiological study with a view to catheter ablation and who have an LBBB morphology VT induced, it is important to initially exclude bundle branch reentry as the underlying mechanism. Myocardial VT in this setting can be distinguished from bundle branch reentry by certain characteristics. First, a LBBB-QRS morphology with the presence of a His signal before the ventricular signal with an H-V interval that is equal to or longer than that in sinus rhythm due to rate-related reduction in conduction velocity in the RBB. Second, the finding that preceding H-H intervals predict the following V-V intervals. BBR-VT is an important arrhythmia to diagnose because catheter ablation of the right bundle branch is curative of this arrhythmia. Some patients will require pacing after right bundle branch ablation, particularly those with a significant left bundle branch conduction delay or complete LBBB at baseline. Because of the severity of LV dysfunction, a dual-chamber or BiV ICD is usually indicated in such patients. Electroanatomic mapping has continued to afford new insights into the basis of structural abnormalities in the genesis of ventricular arrhythmias in DCM through the creation of activation and voltage maps of the ventricles using endocardial and epicardial approaches. Mapping is typically performed during sinus rhythm or atrial fibrillation, such that ventricular activation is uniform during the mapping procedure. During mapping, attention is directed toward ensuring good catheter contact with the myocardium. Peak-to-peak electrogram voltages less than 0.5 mV are generally associated with the presence of myocardial “scar.” Voltages less than 1.5mV are generally associated with the presence of diseased myocardium and are typically found at regions near scarred myocardium. However, unlike in patients with cardiomyopathies resulting from prior myocardial infarcts, such as of low and intermediate voltages, are typically distributed in a nonregional manner. As a result, the areas of interest in patients with DCM may be found in a more diffuse and patchy distribution. Therefore, when creating voltage maps in patients with DCM, care must be taken to interrogate the entire ventricular surface. In addition, a high-density map leads to a better definition of regions, which can create a substrate for reentrant arrhythmias. While being generated, these voltage maps are displayed in a color-coded manner on the electroanatomic map (EAM), so that all areas of viable myocardium are given the same color code and the scar can be differentiated further into different zones according to the local voltage at multiple points within the scar. In patients with DCM, there is a propensity for regions of low voltage to be located at the basal LV, particularly at the level of the mitral valve. During mapping of the substrate, other electrograms of interest that might be important in the genesis of arrhythmias may be tagged on the EAM. These include fractionated electrograms that could represent regions of slow conduction, late (>10 ms after the terminal deflection of the surface QRS) and double potentials (electrograms with a wide isoelectric interval between them), which can represent regions of slow conduction or electrical block. This approach facilitates the definition of the myocardial substrate and putative regions of interest. Soejima et al.20 characterized VT in DCM using electroanatomic mapping and found macroreentrant VT to be the dominant mechanism. A critical isthmus was found in 12 patients during endocardial mapping and in 7 patients during epicardial mapping. All patients who had endocardial mapping had at least one area of scar, with an average of two scars per patient. More than half of the 32 scars extended to a valve annulus, most commonly the mitral annulus. The total area of endocardial scar per patient was approximately 16 cm2 (see Figure 85-5). Figure 85-5 A, Voltage map of the endocardial surface of the left ventricle (LV). An extensive low-voltage area is present in the LV outflow area. B, Two morphologies of ventricular tachycardia (VT). C, Pace mapping at sites 1, 2, and 3 labeled in (A) is shown. A good pace match for VT1 is observed at sites 2 and 3. A long S-QRS delay (200 ms), consistent with slow conduction away from the pacing site, is observed during pace mapping at site 1. A series of radiofrequency lesions (red circles) across this region from the aortic annulus to the dense scar above the mitral annulus abolished both VTs. (From Soejima K, Stevenson WG, Sapp JL, et al: Endocardial and epicardial radiofrequency ablation of ventricular tachycardia associated with dilated cardiomyopathy: the importance of low-voltage scars. J Am Coll Cardiol 43:1834–1842, 2004.) With the advent of telemetry and Holter monitoring, VT, previously thought to be a relatively rare condition, was noted in 50% to 60% of patients with DCM and was estimated to be responsible for 8% to 50% of deaths.4 Of patients with all-cause CHF, approximately 50% have been reported to die suddenly. An approximately equal number succumb to progressive ventricular dysfunction with subsequent mortality being related to progressive pump failure. Although the likelihood of death from progressive pump failure rather than sudden arrhythmic death increases with the severity of heart failure symptoms, the absolute likelihood of sudden death (presumed arrhythmic death) increases with New York Heart Association (NYHA) class. In the absence of an implanted device or telemetry, distinguishing sudden death from death caused by pump failure can be difficult. Despite their common occurrence, ventricular arrhythmias are not the only cause of sudden death in patients with DCM. Luu et al.21 reviewed 21 episodes of cardiac arrest in 20 hospitalized, monitored patients with end-stage heart failure caused by ischemic or nonischemic cardiomyopathy. Primary ventricular tachyarrhythmias occurred in 38% of arrests. Bradycardia or electromechanical dissociation was the initial event in 62% of the patients. First- and second-degree atrioventricular (AV) block have been identified as markers of poor prognosis in patients with DCM. Others have also noted that ischemia, secondary to acute coronary artery thrombi or emboli, can lead to sudden death in a small number of patients. Pulmonary emboli and electrolyte imbalances are other significant precipitants of sudden death in patients with heart failure.21,22 A more detailed examination of the presence, extent, and transmurality of myocardial scar is possible with cardiac magnetic resonance imaging (Figure 85-5). As previously observed on autopsy findings, it has been observed with MRI that the regions and depth of scar in patients with DCM differ from other forms of cardiomyopathies. As discussed previously, the location of the scar, as defined by late gadolinium enhancement (LGE), has a propensity for the basal regions. Transmurality is less commonly observed than in patients with ischemia as an underlying cause for their cardiomyopathy and subepicardial and midmyocardial scar is frequently observed in patients with DCM. Initially described by McCrohon et al.23 who found longitudinal striae of midwall enhancement consistent with fibrosis in 28% of patients with DCM, MRI can be used to quantitate the extent of scar and define its location. Nazarian et al.24 demonstrated a propensity for scar in the basal midmyocardial layers of the LV in patients with DCM and found that the amount of nontransmural scar (26% to 75% wall thickness), as defined by LGE, was an independent predictor of the presence of inducible VT during programmed ventricular stimulation. In several cases, the location of scar could be correlated with the origin of the clinical VT (see Figure 85-5). In a series of 141 patients studied by Hombach et al.25 predictors of poor clinical outcomes (cardiac death) included a longer QRS duration (>110 ms), comorbid diabetes mellitus, and the presence of LGE on MRI .
Ventricular Tachycardia in Patients With Dilated Cardiomyopathy
Incidence and Survival
Genetics
Pathophysiology
Arrhythmogenesis
Mechanisms of Ventricular Tachycardia
Insights from Electroanatomic Mapping
Mechanisms of Sudden Death
Imaging
Thoracic Key
Fastest Thoracic Insight Engine