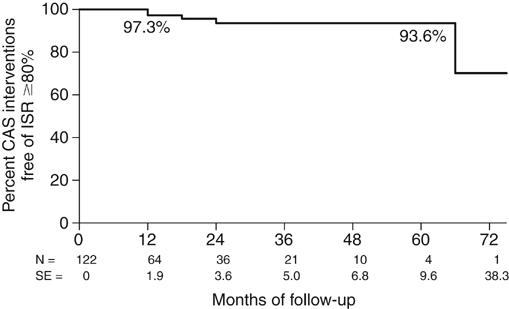
Life-table analysis provides specific information on ISR after CAS (Figure 1). Over a follow-up period of 1 to 74 months, only five patients demonstrated high-grade ISR (≥80%), and the projected 5-year recurrence rate for ISR of 80% or more was 6.4%. In the Carotid Revascularization, Endarterectomy versus Stent Trial (CREST), of the 1086 patients receiving CAS, only 58 patients developed 70% or greater ISR, for a 2-year recurrence rate of 6.0%. The Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy (SAPPHIRE) trial, Carotid and Vertebral Artery Transluminal Angioplasty Study (CAVATAS), Stent-Protected Angioplasty versus Carotid Endarterectomy (SPACE) study, and the Endarterectomy versus Angioplasty in Patients with Symptomatic Severe Carotid Stenosis (EVA-3S) trial, each reported similarly low recurrence rates ranging from 2.8% over 3 years to 10.5% over 5 years. Therefore, although ISR does not appear to occur at the high rates associated with bare-metal stenting of the coronary arteries, a substantial number of patients can be anticipated to progress to moderate and high-grade ISR. Duplex ultrasonography is the standard technique for following carotid stenosis patients treated with CEA or medical therapy alone. Ultrasound velocities correlate with angiographic percentage of stenosis in the native unstented carotid artery, and the appropriate threshold velocities signifying different degrees of stenoses have been intensively analyzed and identified. However, ultrasound velocity criteria have not been well established for patients undergoing CAS. In 2004, we reported that the placement of a stent altered the biomechanical properties of the carotid territory such that compliance was reduced. The enhanced stiffness of the stent–arterial wall complex rendered the flow–pressure relationship of the carotid artery closer to that observed in a rigid tube so that the energy normally applied to dilate the artery resulted in increased velocity. We therefore proposed revised velocity criteria to serve as guidelines for other vascular laboratories when studying stented carotid arteries (Table 1). These revised criteria have been confirmed by multiple single-institution studies and have also been used to measure ISR rates in randomized, controlled trials (EVA3S and CREST). TABLE 1 Suggested Duplex Ultrasound Threshold Velocity Criteria Defining Clinically Important Stenoses in the Stented Carotid Artery (University of Maryland Criteria)
Treatment of Recurrent Carotid Artery Stenosis After Percutaneous Angioplasty and Stenting
Incidence
Diagnosis
Percent Stenosis
Peak Systolic Velocity Threshold (cm/sec)
<20
<150
20-49
150-219
50-70
220-239
≥70
≥300
≥80
≥340 ![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Thoracic Key
Fastest Thoracic Insight Engine