Technical Aspects of Lobectomy
Stanley C. Fell
Malcolm M. DeCamp Jr.
Lobectomy by hilar dissection, first reported by Blades and Kent1 in 1940 for the surgical treatment of bronchiectasis, is now performed most commonly for the definitive treatment of lung cancer. The incision for lobectomy is usually a posterolateral thoracotomy because it allows greater exposure and maneuverability for the surgeon. Anterolateral thoracotomy, median sternotomy except for the left lower lobe, and muscle-sparing lateral or axillary thoracotomy are used also. Posterior thoracotomy, developed during the era of surgery for pulmonary tuberculosis and bronchiectasis—as described by Overholt and Langer,5 and generally performed on a special operating table with the patient prone—is now of historical interest. Video-assisted thoracoscopic (VATS) lobectomy is now a routine procedure in several institutions, especially for early-stage non-small-cell lung cancer (NSCLC) (Chapter 35).2,4 Oncologic outcomes are similar to those seen after open lobectomy. Key points in the performance of lobectomy are mobilization of the lobe, fissure dissection, and management of the vessels and bronchus.
Mobilization of the Lobe
The pleural cavity is entered through the fifth intercostal space for upper lobectomy or any central lesion. Entry through the sixth space is acceptable for peripheral lower lobe tumors. If adhesive pleuritis is anticipated, entrance through the bed of the resected fifth rib allows for more expeditious mobilization of the lung, either in the intrapleural or extrapleural plane. Web-like avascular adhesions are managed by finger dissection and a sponge stick; cautery is applied for vascular adhesions. Inflammatory and cavitary lesions adherent to the parietal pleura are mobilized in the extrapleural plane.
The need for chest wall resection for tumors adherent to the parietal pleura is controversial. Trastek and associates6 advocate en bloc resection of lung and chest wall in this situation. McCaughan and colleagues3 recommend attempting extrapleural dissection if extrapleural extension is not documented preoperatively. If an extrapleural plane is readily achieved, chest wall resection is not performed, because these tumors often involve only visceral pleura. If the tumor is determined to be fixed to the chest wall intraoperatively, the extrapleural approach is abandoned and en bloc resection is performed. This approach does not adversely influence the survival rate.
After mobilization of the lung, the mediastinal pleura is incised around the hilum, the pathology is evaluated, and node sampling is performed as indicated.
Fissure Dissection
Incomplete fissures result from congenital fusion of lung substance, inflammation, or extension of the pathologic process to the adjacent lobe. Combined sharp and blunt dissection along the interlobar plane is generally sufficient to separate the lobes. Application of a stapling device or dividing lung parenchyma by clamp and suture may be required. The interlobar arterial branches must be visualized during these efforts. Fused lobes also may be separated by retrograde dissection and traction on the divided bronchus using the intersegmental vein as a guide. Small blood vessels and air leaks require suture; if clamps or a stapler is used, diminution of lung volume and distortion of the remaining lobes should be kept to a minimum.
Single-lung anesthesia and stapling devices have facilitated lobectomy, especially when using a VATS technique, but total reliance on these methods is to be decried. The double-lumen endotracheal tube may be unavailable, impossible to place properly, or may become displaced. The surgeon must be able to dissect the lobar hilum and the fissures of a partially inflated lung, preferably manually ventilated.
Management of Lobar Vessels
The key to an orderly lobectomy is thorough knowledge of the anatomy of the pulmonary artery, variations in its branching, and its proper dissection (see Chapter 5). In contrast to the fragile pulmonary segmental arteries, located deep within the fissures and intimately related to the segmental bronchi, the pulmonary veins and their tributary trunks have relatively strong walls and are easily accessible at the anterosuperior and posteroinferior aspects of the hilum. Occasionally a common extrapericardial pulmonary vein is encountered.
The pulmonary arteries are best dissected from their fibrous sheath using scissors in the long axis of the vessel. Once a sufficient length of the vessel has been exposed, the sheath is grasped on either side and the artery is then rolled out of its sheath, allowing the passage of a right-angle or Semb clamp to encircle the vessel and draw a ligature beneath it. Dissection of the artery
with a clamp without prior sharp dissection invites hemorrhage. If segmental branches are short, additional length may be obtained by dissecting into lung parenchyma and dividing the lung substance with cautery.
with a clamp without prior sharp dissection invites hemorrhage. If segmental branches are short, additional length may be obtained by dissecting into lung parenchyma and dividing the lung substance with cautery.
Bronchial closure is generally performed with a 3.5- or 4.8-mm stapling device, depending on the compliance of the bronchus. Manual sewing of the bronchus may be indicated in some circumstances, an example being to ensure that the bronchial resection is proximal to an endobronchial tumor. Suture materials currently used include silk, polyglactin, and polypropylene. Manual closure proceeds as follows: a suitably sized toothed bronchus clamp is applied to the specimen side of the bronchus and stay sutures are inserted in the upper and lower borders of the bronchus. With the underlying vascular structures protected by a Semb clamp, the bronchus is transected. Bronchial sutures are placed about 3 mm apart and 3 mm from the cut edge. In the absence of single-lung ventilation, placing the first suture at the midpoint of the bronchus reduces air leak. Surgeons’ knots reduce tension on the cartilage, the knots being placed over the suture hole in the membranous portion to seal possible air leak from this area. The traction sutures may be tied in place over the cut end. Coverage of the bronchial stump is of value after any lobectomy performed in the face of active infection. We cover lower lobe and bilobectomy stumps routinely with parietal pleura or pericardial thymic fat. Upper lobe stumps are covered selectively in situations of purulence, immune suppression, or when pre- and/or postoperative radiotherapy will be used. After middle lobectomy, flap coverage is unnecessary, as the bronchial stump is readily covered by the remaining parenchymal tissue of the upper and lower lobes within the hemithorax.
After removal of the specimen, the integrity of the bronchial closure is tested by the application of positive pressure to the endotracheal tube with a saline-filled hemithorax. Parenchymal air leaks also are localized and repaired. Two thoracostomy tubes are placed through stab wounds in the anterior axillary line. The lower tube extends posteriorly on the diaphragm, and the upper tube lies anteriorly to reach the apex of the pleura. After closure of the chest, negative suction from 10 to 20 cm H2O may be applied to the drainage systems, although some surgeons advocate early if not immediate use of waterseal drainage alone. The tubes are removed serially once drainage is less than 250 mL in 24 hours and the air leak has ceased.
Right Upper Lobectomy
The anatomy of the hilar structures of the right upper lobe is more complex than that of any other lobe, and arterial anomalies are more common. In about 80% of individuals, the anterior segment of the right upper lobe is partially or completely fused to the middle lobe, producing an incomplete minor or horizontal fissure. This mandates a segmental dissection of this area to complete the lobectomy.
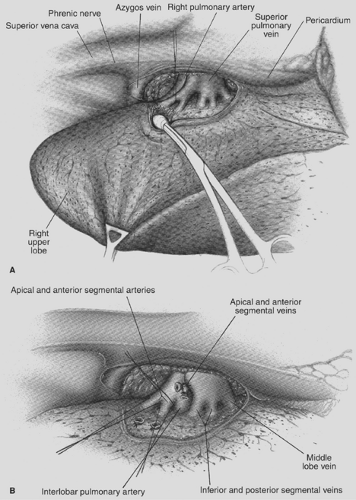
The mediastinal pleura is incised around the hilum of the right lung, lateral to the superior vena cava, inferior to the azygos vein, continuing posteriorly over the bronchus, anterior to the vagus nerve (which is visible subpleurally), to the level of the bronchus intermedius. Anteriorly, the incision is carried to the level of the superior pulmonary vein, posterior to the phrenic nerve (Fig. 28-1A). A pledget dissector is used to push the azygos vein superiorly, demonstrating the upper border of the right main bronchus and the upper lobe bronchus originating from it. Inferior to the azygocaval junction, a lymph node is found. Just below this lymph node is the upper border of the pulmonary artery. The areolar tissue overlying the pulmonary artery is dissected, and the superior arterial trunk is visualized. This artery and its apical and anterior segmental branches are dissected. The apical segmental vein crosses the anterior segmental artery; it is often convenient to ligate and divide this vein before dealing with the artery (see Fig. 28-1B). The superior arterial trunk is doubly tied with 0-0 silk; the apical and segmental branches are tied and then divided. If the segmental arteries are short, additional length may be obtained by dissecting with a right-angle clamp into the pulmonary parenchyma overlying the branches, dividing the parenchyma with cautery. Suture ligatures or clips are then applied, and the segmental arteries are divided. Alternatively a vascular stapler (2.0 mm staple height), which simultaneously ligates and divides the vessels, may be used.
After division of the superior trunk of the pulmonary artery, the common stem of the apical and anterior segmental veins is dissected and divided. The interlobar trunk of the pulmonary artery lies directly beneath the upper and middle stems of the superior pulmonary vein, and this dissection must be performed cautiously.
The remaining arterial supply to the right upper lobe is the posterior ascending artery, present in 90% of patients. Dissection of this artery can be the most formidable task in the procedure. Three approaches have been described: an anterior approach, an approach through the oblique fissure, and a retrograde approach.
The anterior approach requires prior division of the posterior and inferior venous tributaries of the middle stem of the superior vein, which is closely applied to the anterior surface of the inferior trunk of the pulmonary artery. Further dissection of the interlobar artery is required because the posterior segmental artery arises from the anterior aspect of the interlobar artery just above the superior segmental artery to the lower lobe. Proximal control of the right pulmonary artery may be required because laceration of the posterior ascending artery or the interlobar artery from which it arises may occur during this dissection.
An approach to the posterior segmental artery through the oblique fissure is acceptable provided that the oblique fissure is virtually complete. Otherwise, the artery is again at risk for injury. The retrograde method for completion of the dissection is both safe and expeditious.
Retrograde exposure of the posterior ascending artery proceeds as follows: attention is directed to the posterior aspect of the hilum. The vagus nerve is grasped with an Allis clamp and retracted, thus demonstrating its branches to the right upper lobe. The branches are divided (Fig. 28-2A). Deep to the vagal branches, the bronchial artery may be observed; it is clipped and divided. The lower border of the upper lobe bronchus is dissected. In the crotch between the upper lobe bronchus and the intermediate bronchus is a constant lymph node. This node is dissected toward the specimen, clearing the inferior border of the right-upper-lobe bronchus. It is not advisable to pass a clamp from the lower border of the right-upper-lobe bronchus medially to encircle the bronchus because the posterior ascending artery may be lacerated. Rather, scissor dissection of the medial surface of the bronchus is performed, sweeping areolar tissue
and nodes toward the specimen (see Fig. 28-2B). The bronchus is not denuded of its fascia, which supplies the vascularity required for healing. An index finger can then be inserted along the anterior aspect of the bronchus to reach its lower border (see Fig. 28-2C). A right-angle clamp may then be passed safely around the right-upper-lobe bronchus. A Semb clamp is used to widen the peribronchial space, allowing for the passage of a 4.8-mm stapling device. The bronchus is either stapled and divided or manually sutured. If stapled, the staple line generally includes the bronchial artery to the right upper lobe. The cut edge of the specimen side of the bronchus is grasped with an Allis clamp. Traction is placed on the Allis clamp, a toothed bronchus clamp is applied, and the Allis clamp is removed. By turning the handle of the bronchus clamp medially, thus elevating the cut
bronchus, the fissure dissection is facilitated. With gentle medial traction on the bronchus clamp, the areolar tissue and nodes are readily dissected off the interlobar pulmonary artery, and the posterior ascending artery is identified, ligated, and divided (Fig. 28-3). Occasionally, an additional arterial branch to the anterior segment originates from the interlobar artery. Rarely, the posterior segmental artery originates from the superior segmental artery.
and nodes toward the specimen (see Fig. 28-2B). The bronchus is not denuded of its fascia, which supplies the vascularity required for healing. An index finger can then be inserted along the anterior aspect of the bronchus to reach its lower border (see Fig. 28-2C). A right-angle clamp may then be passed safely around the right-upper-lobe bronchus. A Semb clamp is used to widen the peribronchial space, allowing for the passage of a 4.8-mm stapling device. The bronchus is either stapled and divided or manually sutured. If stapled, the staple line generally includes the bronchial artery to the right upper lobe. The cut edge of the specimen side of the bronchus is grasped with an Allis clamp. Traction is placed on the Allis clamp, a toothed bronchus clamp is applied, and the Allis clamp is removed. By turning the handle of the bronchus clamp medially, thus elevating the cut
bronchus, the fissure dissection is facilitated. With gentle medial traction on the bronchus clamp, the areolar tissue and nodes are readily dissected off the interlobar pulmonary artery, and the posterior ascending artery is identified, ligated, and divided (Fig. 28-3). Occasionally, an additional arterial branch to the anterior segment originates from the interlobar artery. Rarely, the posterior segmental artery originates from the superior segmental artery.
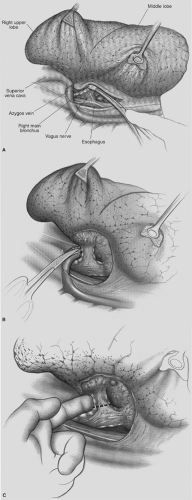 Figure 28-2. A: Posterior aspect of the right-upper-lobe hilum after division of the mediastinal pleura. Vagal branches posterior to the bronchus are not yet divided. B: The right-upper-lobe bronchus is dissected. C: Finger dissection separates the bronchus from the interlobar pulmonary artery.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|