Fig. 1.
MRI standard protocol and findings according to tissue characteristics
MR Analysis
In the Breast Imaging-Reporting and Data System (BI-RADS) lexicon published in 2013, Chapter IV is devoted to describing breast implants, in the section on MRI. The MRI report should include implant type (silicone or saline content), location (pre or retropectoralis), contour (regular, focal budge, ruptured), and the location of the silicone. Intra-capsular rupture is observed in 80% and extra-capsular rupture in 20% of patients with silicone gel implants The combination of five different features needs to be analyzed to diagnose a rupture: (a) the fibrous capsule, (b) the implant envelope, (c) the signal of the periprosthetic fluid (d) the location of the detected silicone, and (e) the overall implant shape (Table 1).
Table 1.
Implant analysis using magnetic resonance imaging
Intact implant | Rupture | |||
|---|---|---|---|---|
Normal | Capsule contraction | Intracapsular | Extracapsular rupture | |
Fibrous capsule | Not seen | Focal bulge | Undisrupted | Seen Disrupted |
Implant envelope | Smooth, or radial fold (with dark ball end) | Smooth, or radial folds (with dark ball end) | Key-hole, tear drop, noose (with white ball end) Linguine sign | Linguine sign |
Periprosthetic fluid Signal | Thin | Thin or thick | Thick | Absent |
Water | Water | Silicone* | Silicone* | |
Silicone location* | Only inside the implant envelope | Only inside the implant envelope | Outside the implant envelope Inside fibrous capsule | Outside the implant envelope and fibrous capsule |
Overall implant shape | Normal | Abnormal | Normal | Abnormal (extruded silicon globe separated by a dark line) |
The radial folds are illustrated in Fig. 2. Water droplets inside breast implants are seen on water-specific images; they may be normal (due to the injection of silicone gel during surgery, for example) or associated with implant rupture. The presence of silicone within axillary lymph nodes does not necessarily imply implant rupture.
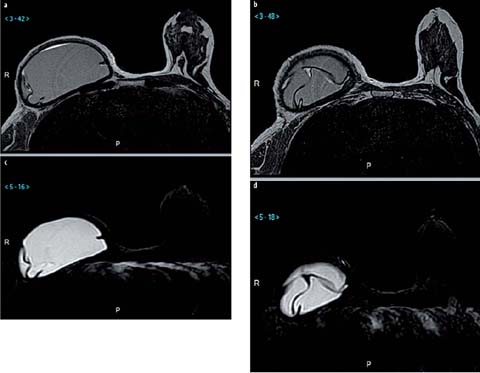
Fig. 2 a–d.
Radial folds. a, b T2-weighted sequence showing dark lines with different signal intensities inside and outside the implants. Periprosthetic fluid displays a high T2-weighted signal intensity. c, d Silicone-only sequence shows the absence of silicone outside the prosthesis
Autologous Reconstruction
During the last 20 years, oncological, reconstructive, and cosmetic surgery techniques have evolved considerably, with the development of novel surgical approaches and skin-and nipple-sparing mastectomies that can be completed with silicone-filled implants, an autologous flap (pedicled or free flaps), or by autologous fat grafting. In certain oncological or prophylactic cases, a skin-sparing or nipple-areola-complex-sparing mastectomy may allow enhanced cosmetic results by conserving most of the skin envelope. The need for systematic imaging follow-up of these patients is unclear, including for patients with oncological indications [11, 12]. Normal mammary gland is found in about 60% of patients after a skin-sparing mastectomy according to Tokin et al. [13]. In skin- and nipple-sparing mastectomy performed in oncology patients, it should be noted that the risk of local recurrence correlates with the margin status and the type of mastectomy. According to Kroll et al. [14], regardless of the reconstruction technique, this risk is 4–6% at 5 years, similar to that in the group of patients not undergoing reconstruction. Depending on the location of the recurrence, the patient may or may not be symptomatic. In 50–70% of cases, the recurrence develops in the anterolateral part of the reconstructed breast and is easily detected by the clinician, but in 30% of cases the recurrence may be in the axillary region or within the thoracic wall, which in either case is very difficult to diagnose clinically. When the patient is symptomatic, the radiologist must be able to distinguish normal imaging features from features of malignancy, which as a prerequisite requires an awareness of the different types of breast reconstruction options.
Autologous Flaps
General Considerations
The two most common types of conventional pedicled flaps are the latissimus dorsi myocutaneous (LDM) flap and the transverse rectus abdominis myocutaneous (TRAM) flap; the latter is harvested from excess abdominal tissue. These autologous breast reconstruction methods allow the insertion of a muscle with a skin island and are mainly indicated when there is a lack of skin after conservative mastectomy. The LDM flap is vascularized by the thoracodorsal pedicle whereas the epigastric superior vessels supply the TRAM flap. These flaps include the muscle to insure good vascularization of the skin and fat. By contrast, free flaps, such as the deep inferior epigastric perforator (DIEP) flap, are becoming increasingly popular because they are muscle-sparing. Autologous tissue transfer involves complete separation of the donor tissue. In the case of the abdominal excess transferred in the DIEP flap, a pedicle is dissected in the muscle, and there is absolutely no muscle within the flap (hence the name perforator flap). The harvested tissue is then connected to the recipient site by an anastomosis, with microsurgical sutures performed under the microscope: the deep inferior vessels are ligated to the internal mammary vessels lying under the cartilage of the third or fourth rib [15]. The techniques required for perforator flaps are more complex, with a significantly longer operating time, than those involved in the placement of a conventional pedicled flap [16, 17]. Other flaps that may be used are the superior inferior epigastric perforator (SIEP) flap, the superior gluteal artery perforator (SGAP) flap, and the inferior gluteal artery perforator (IGAP) flap.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


