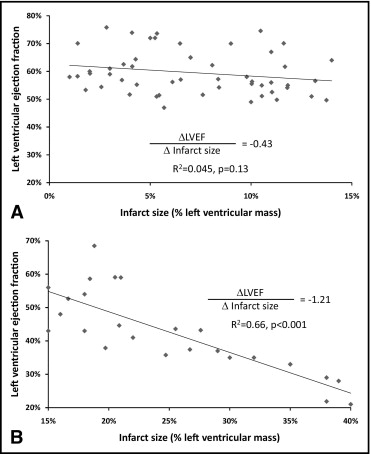
The goal of this analysis was to determine the relation between myocardial infarct size and left ventricular (LV) ejection fraction (EF) in patients with ST-segment elevation myocardial infarction (STEMI) after primary percutaneous coronary intervention (pPCI) using cardiovascular magnetic resonance imaging (CMR). After STEMI, LVEF and infarct size correlate with prognosis, but the relation between infarct size and LVEF is incompletely known. Consecutive subjects presenting to a single center with STEMI treated with pPCI were enrolled, and cine functional and late gadolinium enhancement CMR was performed 3 months after presentation. From cine images, LVEF was calculated using volumetric summation of disks method. Infarct size was measured as percent LV myocardial volume with late gadolinium enhancement. In the 78 patients enrolled (mean age 54.5 years, range 42 to 82), median LVEF was 56% (interquartile range 49 to 62) and median infarct size was 11% (interquartile range 5 to 18). Of the 53 patients with infarct size <15%, all had LVEF >40%, and there was no significant relation between infarct size and LVEF (slope −0.43, R 2 = 0.045, p = 0.13). In patients with infarct size ≥15%, there was a significant negative linear association between infarct size and LVEF (slope −1.21, R 2 = 0.66, p <0.001), such that for every 5% increase in infarct size, there was a 6.1% decrease in LVEF. In conclusion, there is a negative linear relation between infarct size and LVEF for moderate to large infarcts. For small infarcts there is no significant relation between infarct size and LVEF. Up to 15% of LV myocardial volume may be infarcted before there is any appreciable decrease in LVEF.
Following ST-segment elevation myocardial infarction (STEMI), decreased left ventricular (LV) systolic function has been associated with poor short- and long-term clinical outcomes, and LV ejection fraction (EF) has gained favor as a prognostic indicator of adverse outcomes. Since the introduction of rapid reperfusion therapy for STEMI, however, preserved LV function is more common, and there is likely a spectrum of risk of adverse events in patients with preserved LVEF. Cardiovascular magnetic resonance imaging (CMR) provides highly accurate, reproducible volumetric data regarding LVEF and infarct size. A larger infarct has been reported to be a better identifier of inducible ventricular tachycardia than LVEF in patients with STEMI and is associated with long-term adverse cardiovascular events. The association between infarct size and LVEF has not been specifically evaluated, although the presumption is that it is linear across the spectrum of infarct size. Our hypothesis was that in patients with relatively small infarcts, there would be no appreciable decrease in LVEF. We sought to determine the association between infarct size and LVEF using CMR in patients who underwent successful primary percutaneous coronary intervention (pPCI) for STEMI.
Methods
Patients who were ≥18 years of age were eligible for enrollment on presentation with their first STEMI to a single center and were subsequently treated with pPCI. Evidence of new STEMI required the presence of ≥1 of the following: typical symptoms of acute infarction, electrocardiographic evidence of new ST-segment elevation ≥1 mm in 2 contiguous leads, creatine phosphokinase levels >2 times the upper limit of normal with >5% creatine kinase-MB, or increased troponin I or T. Exclusion criteria were (1) previous MI, defined by pre-existing pathologic Q waves or previous admission for MI with elevated cardiac biomarkers or previous evidence of a regional wall motion abnormality; (2) nonsinus rhythm; (3) inability to obtain informed consent; or (4) fibrinolytic therapy. Patients’ demographics, Thrombolysis In Myocardial Infarction (TIMI) flow grade, TIMI myocardial perfusion grade, microvascular obstruction, infarct size, and LVEF were assessed. Those who were rehospitalized for MI or repeat revascularization before the 3-month CMR were excluded from the study to avoid possible confounding of multiple MIs causing hibernating myocardium, thus influencing CMR outcomes.
Participants provided written informed consent according to the hospital’s committee on clinical investigation. All participants were treated according to the standard of care for STEMI and underwent pPCI with serial sampling of serum cardiac biomarkers.
A blinded, experienced observer analyzed all 12-lead electrocardiograms obtained at baseline, 90 minutes after reperfusion, and at 3 months. Each was analyzed for ST-segment deviation and presence of pathologic Q waves. TIMI flow grade and TIMI myocardial perfusion grade were assessed at an angiographic core laboratory. All flow and perfusion data were over-read by a single experienced observer blinded to clinical outcomes and CMR data.
CMR was performed using a commercial 1.5-T Philips Gyroscan ACS/NT whole-body scanner (Philips Medical Systems, Best, The Netherlands) with a 60-cm diameter bore, Powertrak 6000 gradients (23 mT/m, 219-ms increase time), and a commercial 5-element cardiac synergy coil. All cine images were acquired with retrospective electrocardiographic gating during end-expiratory breath-holds of approximately 8 seconds. Each of the 9 to 12 contiguous short-axis slices (10 mm thick) was obtained using steady-state free precession methods. Gadolinium-diethylenetriamine penta-acetic acid was then given (total dose gadolinium-diethylenetriamine penta-acetic acid dose 0.20 mmol/kg) followed by late gadolinium enhancement-CMR 15 minutes later. Optimal inversion time was determined by a real-time interactive scan (Look-Locker) that demonstrates uniform suppression of noninfarcted myocardium.
CMR analyses were performed on a commercial workstation (EasyVision 5, Philips Medical Systems). LV mass and LVEF were measured from the cine LV short-axis dataset using standard volumetric techniques. Infarct size was measured from the LV short-axis stack of late gadolinium enhancement images as percent LV myocardial volume with late gadolinium enhancement (defined by pixels ≥1/2 maximal signal intensity of infarct area) and expressed in units of percent total LV myocardial volume.
Data are presented as mean ± SD. All continuous variables were tested for normality using the Shapiro-Wilke test. Continuous, normally distributed variables were expressed as mean ± SD. Continuous, non-normal distributions were expressed as median and interquartile range. Paired 2-tailed Student’s t test was used to assess for significance in continuous variables. Dichotomous variables were analyzed using Fisher’s exact test. The kappa statistic was employed to measure the agreement between CMR indexes and angiographic indexes. A p value <0.05 was considered statistically significant. All analyses were performed using STATA 8.2 (STATA Corp., College Station, Texas).
Results
Eighty-three consecutive patients presenting with their first STEMI were enrolled in the study, of whom 5 were excluded because they presented before the 3-month CMR follow-up for repeat MI or revascularization. Thus, 78 patients remained in our study cohort (67% men, mean age 54.5 years, range 42 to 82; Table 1 ).
| Median age (years) | 54.5 |
| Men | 67% |
| Medical history | |
| Hypertension | 52% |
| Diabetes mellitus | 13% |
| Hyperlipidemia | 63% |
| Current tobacco abuse | 50% |
| Family history of premature coronary artery disease | 31% |
| Medications before admission | |
| Aspirin | 10% |
| β blocker | 12 |
| Angiotensin-converting enzyme inhibitor/angiotensin receptor blocker | 6% |
| Diuretic | 15% |
| Statin | 33% |
| Transfer from outside hospital | 22% |
| Killip class | |
| I | 58% |
| II | 33% |
| III | 6% |
| IV | 3% |
| Mean Thrombolysis In Myocardial Infarction risk score | 1.5 |
| Door-to-balloon time (minutes), mean (range) | 82 (34–183) |
| Electrocardiographic changes | |
| Anterior location | 45% |
| Maximum ST-segment elevation (mm), mean (range) | 3.5 (2.1–7.8) |
| >70% ST-segment resolution at 90 minutes | 56% |
| Coronary angiography | |
| Culprit lesion location | |
| Left anterior descending coronary artery | 45% |
| Right coronary artery | 30% |
| Left circumflex coronary artery | 25% |
| Collateral flow to infarct-related artery | 12% |
| Disease extent | |
| 1 vessel | 78% |
| 2 vessels | 15% |
| 3 vessels | 7% |
| Following percutaneous coronary intervention | |
| Thrombolysis In Myocardial Infarction grade 2/3 flow | 97% |
| Median Thrombolysis In Myocardial Infarction myocardial perfusion grade | 2 |
| Discharge medications | |
| Aspirin | 100% |
| Clopidogrel | 100% |
| β blocker | 88% |
| Angiotensin-converting enzyme inhibitor/angiotensin receptor blocker | 83% |
| Diuretic | 35% |
| Statin | 96% |
Anatomic distribution of STEMIs is presented in Table 1 . Average maximum ST-segment elevation was 3.5 mm (range 2.1 to 7.8). Most patients (78%) had 1-vessel disease. The culprit lesion was in the left anterior descending coronary artery in 45% of cases, right coronary artery in 30%, and left circumflex artery in 25%. After PCI, TIMI grade 2 or 3 flow was present in 97% of patients. Complete ST-segment resolution (>70%) occurred in 56% of patients at 90 minutes after PCI.
Patients underwent CMR 3 months after initial presentation (median 99 days, range 87 to 110), at which time median LVEF was 56% (interquartile range 49 to 62). There were 25 patients with LVEF ≤40% and 4 with LVEF <30%. The remaining patients had relatively preserved LV function (LVEF >40%).
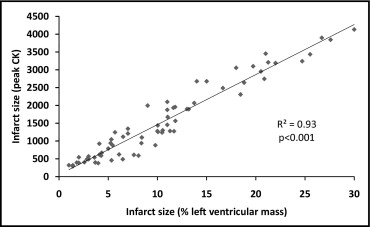
Median infarct size was 11% of LV mass (interquartile range 5 to 18). Most patients (n = 53, 68.0%) had infarct size <15%, and 12 had infarct size ≥25%. Infarct size, as measured by peak creatine kinase release, correlated very strongly with infarct size as assessed by percent LV mass ( Figure 1 ).
Of the 53 patients with infarct size <15%, all had LVEF >40%; of patients with infarct size ≥15%, 11 (44%) had LVEF ≤40% (p <0.001). In those patients with smaller infarcts (<15%), there was no significant relation between infarct size and LVEF (R 2 = 0.045, p = 0.13; Figure 2 ). For those patients with infarct size ≥15%, there was a significant negative linear association between infarct size and LVEF (R 2 = 0.66, p <0.001), such that for every 5% increase in infarct size, there was a 6.1% decrease in LVEF.
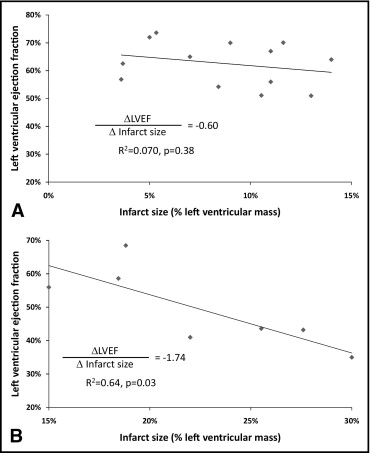
Patients were further stratified by infarct location. In patients with anterior infarcts, there was no significant relation between infarct size and LVEF in patients with infarct size <15% (R 2 = 0.070, p = 0.38); in those with large anterior infarcts (≥15%), there was a significant negative linear relation (R 2 = 0.64, p = 0.03; Figure 3 ). Similar results were observed in patients with small (<15%) inferior infarcts (R 2 = 0.081, p = 0.08) and large (≥15%) inferior infarcts (R 2 = 0.69, p <0.001; Figure 4 ).