PHYSICAL EXAMINATION OF THE CARDIOVASCULAR SYSTEM
The approach to a patient with known or suspected cardiovascular disease begins with the time-honored traditions of a directed history and a targeted physical examination. The scope of these activities depends on the clinical context at the time of presentation, ranging from an elective ambulatory follow-up visit to a more focused emergency department encounter. There has been a gradual decline in physical examination skills over the last two decades at every level, from student to faculty specialist, a development of great concern to both clinicians and medical educators. Classic cardiac findings are recognized by only a minority of internal medicine and family practice residents. Despite popular perceptions, clinical performance does not improve predictably as a function of experience; instead, the acquisition of new examination skills may become more difficult for a busy individual practitioner. Less time is now devoted to mentored cardiovascular examinations during the training of students and residents. One widely recognized outcome of these trends is the progressive overutilization of noninvasive imaging studies to establish the presence and severity of cardiovascular disease even when the examination findings imply a low pretest probability of significant pathology. Educational techniques to improve bedside skills include repetition, patient-centered teaching conferences, and visual display feedback of auscultatory events with Doppler echocardiographic imaging.
The evidence base that links the findings from the history and physical examination to the presence, severity, and prognosis of cardiovascular disease has been established most rigorously for coronary artery disease, heart failure, and valvular heart disease. For example, observations regarding heart rate, blood pressure, signs of pulmonary congestion, and the presence of mitral regurgitation (MR) contribute importantly to bedside risk assessment in patients with acute coronary syndromes. Observations from the physical examination in this setting can inform clinical decision making before the results of cardiac biomarkers testing are known. The prognosis of patients with systolic heart failure can be predicted on the basis of the jugular venous pressure (JVP) and the presence or absence of a third heart sound (S3). Accurate characterization of cardiac murmurs provides important insight into the natural history of many valvular and congenital heart lesions. Finally, the important role played by the physical examination in enhancing the clinician-patient relationship cannot be overestimated.
THE GENERAL PHYSICAL EXAMINATION
Any examination begins with an assessment of the general appearance of the patient, with notation of age, posture, demeanor, and overall health status. Is the patient in pain or resting quietly, dyspneic or diaphoretic? Does the patient choose to avoid certain body positions to reduce or eliminate pain, as might be the case with suspected acute pericarditis? Are there clues indicating that dyspnea may have a pulmonary cause, such as a barrel chest deformity with an increased anterior-posterior diameter, tachypnea, and pursed-lip breathing? Skin pallor, cyanosis, and jaundice can be appreciated readily and provide additional clues. A chronically ill-appearing emaciated patient may suggest the presence of long-standing heart failure or another systemic disorder, such as a malignancy. Various genetic syndromes, often with cardiovascular involvement, can also be recognized easily, such as trisomy 21, Marfan syndrome, and Holt-Oram syndrome. Height and weight should be measured routinely, and both body mass index and body surface area should be calculated. Knowledge of the waist circumference and the waist-to-hip ratio can be used to predict long-term cardiovascular risk. Mental status, level of alertness, and mood should be assessed continuously during the interview and examination.
Skin
Central cyanosis occurs with significant right-to-left shunting at the level of the heart or lungs, allowing deoxygenated blood to reach the systemic circulation. Peripheral cyanosis or acrocyanosis, in contrast, is usually related to reduced extremity blood flow due to small vessel constriction, as seen in patients with severe heart failure, shock, or peripheral vascular disease; it can be aggravated by the use of β-adrenergic blockers with unopposed α-mediated constriction. Differential cyanosis refers to isolated cyanosis affecting the lower but not the upper extremities in a patient with a large patent ductus arteriosus (PDA) and secondary pulmonary hypertension with right-to-left shunting at the great vessel level. Hereditary telangiectasias on the lips, tongue, and mucous membranes, as part of the Osler-Weber-Rendu syndrome (hereditary hemorrhagic telangiectasia), resemble spider nevi and can be a source of right-to-left shunting when also present in the lung. Malar telangiectasias also are seen in patients with advanced mitral stenosis and scleroderma. An unusually tan or bronze discoloration of the skin may suggest hemochromatosis as the cause of the associated systolic heart failure. Jaundice, which may be visible first in the sclerae, has a broad differential diagnosis but in the appropriate setting can be consistent with advanced right heart failure and congestive hepatomegaly or late-term “cardiac cirrhosis.” Cutaneous ecchymoses are seen frequently among patients taking vitamin K antagonists or antiplatelet agents such as aspirin and thienopyridines. Various lipid disorders sometimes are associated with subcutaneous xanthomas, particularly along the tendon sheaths or over the extensor surfaces of the extremities. Severe hypertriglyceridemia can be associated with eruptive xanthomatosis and lipemia retinalis. Palmar crease xanthomas are specific for type III hyperlipoproteinemia. Pseudoxanthoma elasticum, a disease associated with premature atherosclerosis, is manifested by a leathery, cobblestoned appearance of the skin in the axilla and neck creases and by angioid streaks on funduscopic examination. Extensive lentiginoses have been described in a variety of development delay–cardiovascular syndromes, including Carney syndrome, which includes multiple atrial myxomas. Cutaneous manifestations of sarcoidosis such as lupus pernio and erythema nodosum may suggest this disease as a cause of an associated dilated cardiomyopathy, especially with heart block, intraventricular conduction delay, or ventricular tachycardia.
Head and neck
Dentition and oral hygiene should be assessed in every patient both as a source of potential infection and as an index of general health. A high-arched palate is a feature of Marfan syndrome and other connective tissue disease syndromes. Bifid uvula has been described in patients with Loeys-Dietz syndrome, and orange tonsils are characteristic of Tangier disease. The ocular manifestations of hyperthyroidism have been well described. Many patients with congenital heart disease have associated hypertelorism, low-set ears, or micrognathia. Blue sclerae are a feature of osteogenesis imperfecta. An arcus senilis pattern lacks specificity as an index of coronary heart disease risk. The funduscopic examination is an often underutilized method by which to assess the microvasculature, especially among patients with established atherosclerosis, hypertension, or diabetes mellitus. A mydriatic agent may be necessary for optimal visualization. A funduscopic examination should be performed routinely in the assessment of patients with suspected endocarditis and those with a history of acute visual change. Branch retinal artery occlusion or visualization of a Hollenhorst plaque can narrow the differential diagnosis rapidly in the appropriate setting. Relapsing polychondritis may manifest as an inflamed pinna or, in its later stages, as a saddle-nose deformity because of destruction of nasal cartilage; Wegener’s granulomatosis can also lead to a saddle-nose deformity.
Chest
Midline sternotomy, left posterolateral thoracotomy, or infraclavicular scars at the site of pacemaker/defibrillator generator implantation should not be overlooked and may provide the first clue regarding an underlying cardiovascular disorder in patients unable to provide a relevant history. A prominent venous collateral pattern may suggest subclavian or vena caval obstruction. If the head and neck appear dusky and slightly cyanotic and the venous pressure is grossly elevated without visible pulsations, a diagnosis of superior vena cava syndrome should be entertained. Thoracic cage abnormalities have been well described among patients with connective tissue disease syndromes. They include pectus carinatum (“pigeon chest”) and pectus excavatum (“funnel chest”). Obstructive lung disease is suggested by a barrel chest deformity, especially with tachypnea, pursed-lip breathing, and use of accessory muscles. The characteristically severe kyphosis and compensatory lumbar, pelvic, and knee flexion of ankylosing spondylitis should prompt careful auscultation for a murmur of aortic regurgitation (AR). Straight back syndrome refers to the loss of the normal kyphosis of the thoracic spine and has been described in patients with mitral valve prolapse (MVP) and its variants. In some patients with cyanotic congenital heart disease, the chest wall appears to be asymmetric, with anterior displacement of the left hemithorax. The respiratory rate and pattern should be noted during spontaneous breathing, with additional attention to depth, audible wheezing, and stridor. Lung examination can reveal adventitious sounds indicative of pulmonary edema, pneumonia, or pleuritis.
Abdomen
In some patients with advanced obstructive lung disease, the point of maximal cardiac impulse may be in the epigastrium. The liver is frequently enlarged and tender in patients with chronic heart failure. Systolic pulsations over the liver signify severe tricuspid regurgitation (TR). Splenomegaly may be a feature of infective endocarditis, particularly when symptoms have persisted for weeks or months. Ascites is a nonspecific finding but may be present with advanced chronic right heart failure, constrictive pericarditis, hepatic cirrhosis, or an intraperitoneal malignancy. The finding of an elevated JVP implies a cardiovascular etiology. In nonobese patients, the aorta typically is palpated between the epigastrium and the umbilicus. The sensitivity of palpation for the detection of an abdominal aortic aneurysm (pulsatile and expansile mass) decreases as a function of body size. Because palpation alone is not sufficiently accurate to establish this diagnosis, a screening ultrasound examination is advised. The presence of an arterial bruit over the abdomen suggests high-grade atherosclerotic disease, though precise localization is difficult.
Extremities
The temperature and color of the extremities, the presence of clubbing, arachnodactyly, and pertinent nail findings can be surmised quickly during the examination. Clubbing implies the presence of central right-to-left shunting, although it has also been described in patients with endocarditis. Its appearance can range from cyanosis and softening of the root of the nail bed, to the classic loss of the normal angle between the base of the nail and the skin, to the skeletal and periosteal bony changes of hypertrophic osteoarthropathy, which is seen rarely in patients with advanced lung or liver disease. Patients with the Holt-Oram syndrome have an unopposable, “fingerized” thumb, whereas patients with Marfan syndrome may have arachnodactyly and a positive “wrist” (overlapping of the thumb and fifth finger around the wrist) or “thumb” (protrusion of the thumb beyond the ulnar aspect of the hand when the fingers are clenched over the thumb in a fist) sign. The Janeway lesions of endocarditis are nontender, slightly raised hemorrhages on the palms and soles, whereas Osler’s nodes are tender, raised nodules on the pads of the fingers or toes. Splinter hemorrhages are classically identified as linear petechiae in the midposition of the nail bed and should be distinguished from the more common traumatic petechiae, which are seen closer to the distal edge.
Lower extremity or presacral edema in the setting of an elevated JVP defines volume overload and may be a feature of chronic heart failure or constrictive pericarditis. Lower extremity edema in the absence of jugular venous hypertension may be due to lymphatic or venous obstruction or, more commonly, to venous insufficiency, as further suggested by the appearance of varicosities, venous ulcers (typically medial in location), and brownish cutaneous discoloration from hemosiderin deposition (eburnation). Pitting edema can also be seen in patients who use dihydropyridine calcium channel blockers. A Homan’s sign (posterior calf pain on active dorsiflexion of the foot against resistance) is neither specific nor sensitive for deep venous thrombosis. Muscular atrophy or the absence of hair along an extremity is consistent with severe arterial insufficiency or a primary neuromuscular disorder.
CARDIOVASCULAR EXAMINATION
Jugular venous pressure and waveform
Jugular venous pressure is the single most important bedside measurement from which to estimate the volume status. The internal jugular vein is preferred because the external jugular vein is valved and not directly in line with the superior vena cava and right atrium. Nevertheless, the external jugular vein has been used to discriminate between high and low central venous pressure (CVP) when tested among medical students, residents, and attending physicians. Precise estimation of the central venous or right atrial pressure from bedside assessment of the jugular venous waveform has proved difficult. Venous pressure traditionally has been measured as the vertical distance between the top of the jugular venous pulsation and the sternal inflection point (angle of Louis). A distance >4.5 cm at 30° elevation is considered abnormal. However, the actual distance between the mid-right atrium and the angle of Louis varies considerably as a function of both body size and the patient angle at which the assessment is made (30°, 45°, or 60°). The use of the sternal angle as a reference point leads to systematic underestimation of CVP, and this method should be used less for semiquantification than to distinguish a normal from an abnormally elevated CVP. The use of the clavicle may provide an easier reference for standardization. Venous pulsations above this level in the sitting position are clearly abnormal, as the distance between the clavicle and the right atrium is at least 10 cm. The patient should always be placed in the sitting position, with the legs dangling below the bedside, when an elevated pressure is suspected in the semisupine position. It should also be noted that bedside estimates of CVP are made in centimeters of water but must be converted to millimeters of mercury to provide correlation with accepted hemodynamic norms (1.36 cmH2O = 1.0 mmHg).
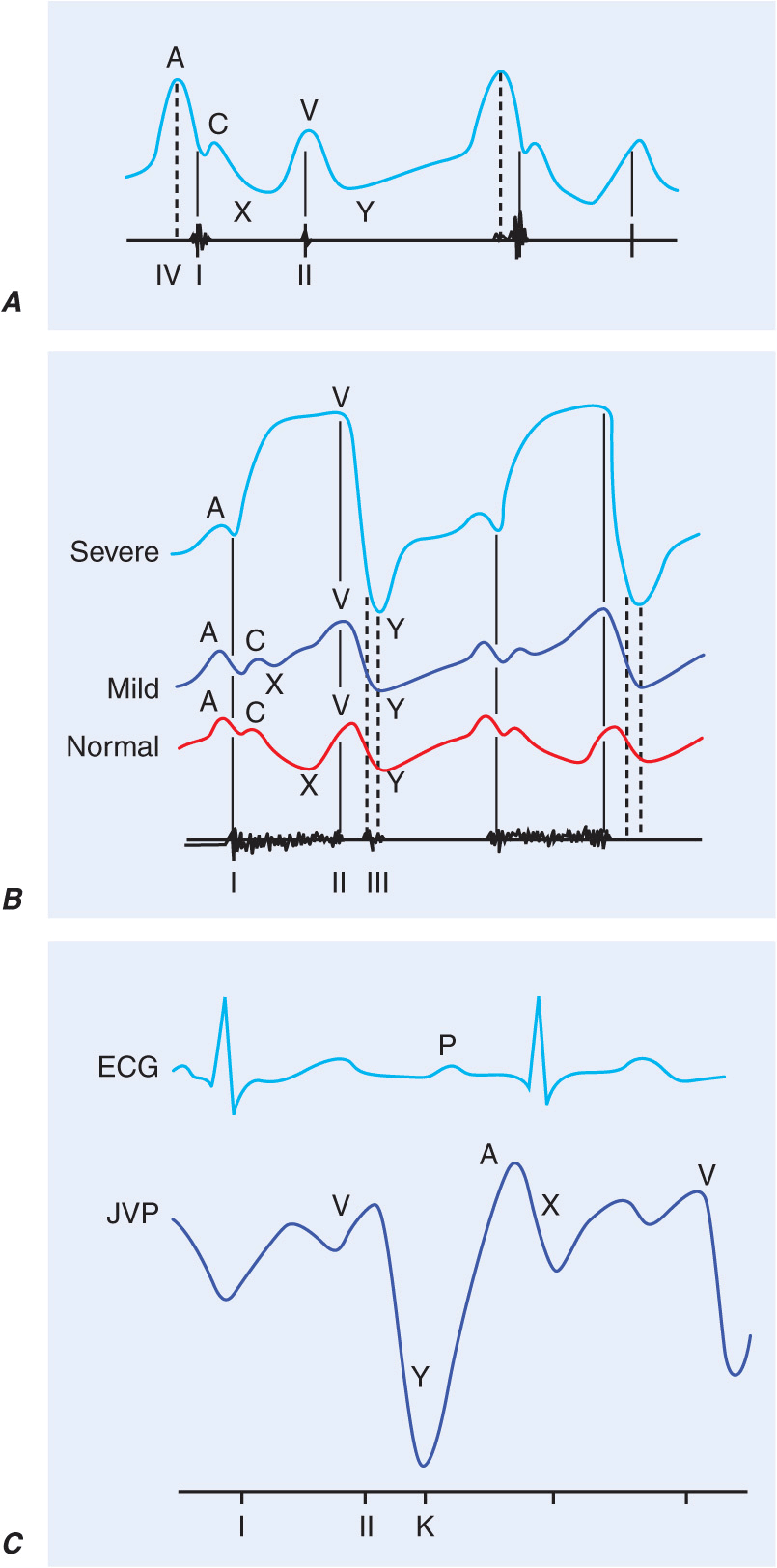
The venous waveform sometimes can be difficult to distinguish from the carotid pulse, especially during casual inspection. Nevertheless, the venous waveform has several characteristic features, and its individual components can be appreciated in most patients (Fig. 9-1). In patients in sinus rhythm, the venous waveform is typically biphasic, whereas the carotid upstroke is monophasic.
FIGURE 9-1
A. Jugular venous pulse wave tracing (top) with heart sounds (bottom). The A wave represents right atrial presystolic contraction and occurs just after the electrocardiographic P wave and just before the first heart sound (I). In this example, the A wave is accentuated and larger than normal due to decreased right ventricular compliance, as also suggested by the right-sided S4 (IV). The C wave may reflect the carotid pulsation in the neck and/or an early systolic increase in right atrial pressure as the right ventricle pushes the closed tricuspid valve into the right atrium. The x descent follows the A wave just as atrial pressure continues to fall. The V wave represents atrial filling during ventricular systole and peaks at the second heart sound (II). The y descent corresponds to the fall in right atrial pressure after tricuspid valve opening. B. Jugular venous waveforms in mild (middle) and severe (top) tricuspid regurgitation, compared with normal, with phonocardiographic representation of the corresponding heart sounds below. With increasing degrees of tricuspid regurgitation, the waveform becomes “ventricularized.” C. ECG (top), jugular venous waveform (middle), and heart sounds (bottom) in pericardial constriction. Note the prominent and rapid y descent, corresponding in timing to the pericardial knock (K). (From J Abrams: Synopsis of Cardiac Physical Diagnosis, 2nd ed. Boston, Butterworth Heinemann, 2001, pp 25–35.)
The venous waveform is divided into several distinct peaks. The a wave reflects right atrial presystolic contraction and occurs just after the electrocardiographic P wave, preceding the first heart sound (S1). A prominent a wave is seen in patients with reduced right ventricular compliance; a cannon a wave occurs with atrioventricular (AV) dissociation and right atrial contraction against a closed tricuspid valve. In a patient with a wide complex tachycardia, the appreciation of cannon a waves in the jugular venous waveform identifies the rhythm as ventricular in origin. The a wave is not present with atrial fibrillation. The x descent defines the fall in right atrial pressure after inscription of the a wave. The c wave interrupts this x descent and is followed by a further descent. The v wave represents atrial filling (atrial diastole) and occurs during ventricular systole. The height of the v wave is determined by right atrial compliance as well as the volume of blood returning to the right atrium either antegrade from the cavae or retrograde through an incompetent tricuspid valve. In patients with TR, the v wave is accentuated and the subsequent fall in pressure (y descent) is rapid. With progressive degrees of TR, the v wave merges with the c wave, and the right atrial and jugular vein waveforms become “ventricularized.” The y descent, which follows the peak of the v wave, can become prolonged or blunted with obstruction to right ventricular inflow, as may occur with tricuspid stenosis (TS) or pericardial tamponade. Normally, the venous pressure should fall by at least 3 mmHg with inspiration. Kussmaul’s sign is defined by either a rise or a lack of fall of the JVP with inspiration and is classically associated with constrictive pericarditis, although it has been reported in patients with restrictive cardiomyopathy, massive pulmonary embolism, right ventricular infarction, and advanced left ventricular systolic heart failure.
Venous hypertension sometimes can be elicited by performance of the abdominojugular reflex or with passive leg elevation. When these signs are positive, a volume-overloaded state with limited compliance of an overly distended or constricted venous system is present. The abdominojugular reflex is elicited with firm and consistent pressure over the upper portion of the abdomen, preferably over the right upper quadrant, for at least 10 s. A positive response is defined by a sustained rise of more than 3 cm in JVP for at least 15 s after release of the hand. Patients must be coached to refrain from breath holding or a Valsalva-like maneuver during the procedure. The abdominojugular reflex is useful in predicting a pulmonary artery wedge pressure in excess of 15 mmHg in patients with heart failure.
Although the JVP estimates right ventricular filling pressure, it has a predictable relationship with the pulmonary artery wedge pressure. In a large study of patients with advanced heart failure, the presence of a right atrial pressure >10 mmHg (as predicted on bedside examination) had a positive value of 88% for the prediction of a pulmonary artery wedge pressure of >22 mmHg. In addition, an elevated JVP has prognostic significance in patients with both symptomatic heart failure and asymptomatic left ventricular systolic dysfunction. The presence of an elevated JVP is associated with a higher risk of subsequent hospitalization for heart failure, death from heart failure, or both.
Assessment of blood pressure
Measurement of blood pressure usually is delegated to a medical assistant but should be repeated by the clinician. Accurate measurement depends on body position, arm size, time of measurement, place of measurement, device, device size, technique, and examiner. In general, physician-recorded blood pressures are higher than nurse-recorded pressures. Blood pressure is best measured in the seated position with the arm at the level of the heart, using an appropriately sized cuff, after 5–10 min of relaxation. When it is measured in the supine position, the arm should be raised to bring it to the level of the mid-right atrium. The length and width of the blood pressure cuff bladder should be 80% and 40% of the arm’s circumference, respectively. A common source of error in practice is to use an inappropriately small cuff, resulting in marked overestimation of true blood pressure, or an inappropriately large cuff, resulting in underestimation of true blood pressure. The cuff should be inflated to 30 mmHg above the expected systolic pressure and the pressure released at a rate of 2–3 mmHg/s. Systolic and diastolic pressures are defined by the first and fifth Korotkoff sounds, respectively. Very low (even 0 mmHg) diastolic blood pressures may be recorded in patients with chronic, severe AR or a large arteriovenous fistula because of enhanced diastolic “runoff.” In these instances, both the phase IV and phase V Korotkoff sounds should be recorded. Blood pressure is best assessed at the brachial artery level, though it can be measured at the radial, popliteal, or pedal pulse level. In general, systolic pressure increases and diastolic pressure decreases when measured in more distal arteries. Blood pressure should be measured in both arms, and the difference should be less than 10 mmHg. A blood pressure differential that exceeds this threshold may be associated with atherosclerotic or inflammatory subclavian artery disease, supravalvular aortic stenosis, aortic coarctation, or aortic dissection. Systolic leg pressures are usually as much as 20 mmHg higher than systolic arm pressures. Greater leg–arm pressure differences are seen in patients with chronic severe AR as well as patients with extensive and calcified lower extremity peripheral arterial disease. The ankle-brachial index (lower pressure in the dorsalis pedis or posterior tibial artery divided by the higher of the two brachial artery pressures) is a powerful predictor of long-term cardiovascular mortality.
The blood pressure measured in an office or hospital setting may not accurately reflect the pressure in other venues. “White coat hypertension” is defined by at least three separate clinic-based measurements >140/90 mmHg and at least two non-clinic-based measurements <140/90 mmHg in the absence of any evidence of target organ damage. Individuals with white coat hypertension may not benefit from drug therapy, although they may be more likely to develop sustained hypertension over time. Masked hypertension should be suspected when normal or even low blood pressures are recorded in patients with advanced atherosclerotic disease, especially when evidence of target organ damage is present or bruits are audible.
Orthostatic hypotension is defined by a fall in systolic pressure >20 mmHg or in diastolic pressure >10 mmHg in response to assumption of the upright posture from a supine position within 3 min. There may also be a lack of a compensatory tachycardia, an abnormal response that suggests autonomic insufficiency, as may be seen in patients with diabetes or Parkinson’s disease. Orthostatic hypotension is a common cause of postural lightheadedness/syncope and should be assessed routinely in patients for whom this diagnosis might pertain. It can be exacerbated by advanced age, dehydration, certain medications, food, deconditioning, and ambient temperature.
Arterial pulse
The carotid artery pulse occurs just after the ascending aortic pulse. The aortic pulse is best appreciated in the epigastrium, just above the level of the umbilicus. Peripheral arterial pulses that should be assessed routinely include the subclavian, brachial, radial, ulnar, femoral, popliteal, dorsalis pedis, and posterior tibial. In patients in whom the diagnosis of either temporal arteritis or polymyalgia rheumatica is suspected, the temporal arteries also should be examined. Although one of the two pedal pulses may not be palpable in up to 10% of normal subjects, the pair should be symmetric. The integrity of the arcuate system of the hand is assessed by Allen’s test, which is performed routinely before instrumentation of the radial artery. The pulses should be examined for their symmetry, volume, timing, contour, amplitude, and duration. If necessary, simultaneous auscultation of the heart can help identify a delay in the arrival of an arterial pulse. Simultaneous palpation of the radial and femoral pulses may reveal a femoral delay in a patient with hypertension and suspected aortic coarctation. The carotid upstrokes should never be examined simultaneously or before listening for a bruit. Light pressure should always be used to avoid precipitation of carotid hypersensitivity syndrome and syncope in a susceptible elderly individual. The arterial pulse usually becomes more rapid and spiking as a function of its distance from the heart, a phenomenon that reflects the muscular status of the more peripheral arteries and the summation of the incident and reflected waves. In general, the character and contour of the arterial pulse depend on the stroke volume, ejection velocity, vascular compliance, and systemic vascular resistance. The pulse examination can be misleading in patients with reduced cardiac output and in those with stiffened arteries from aging, chronic hypertension, or peripheral arterial disease.
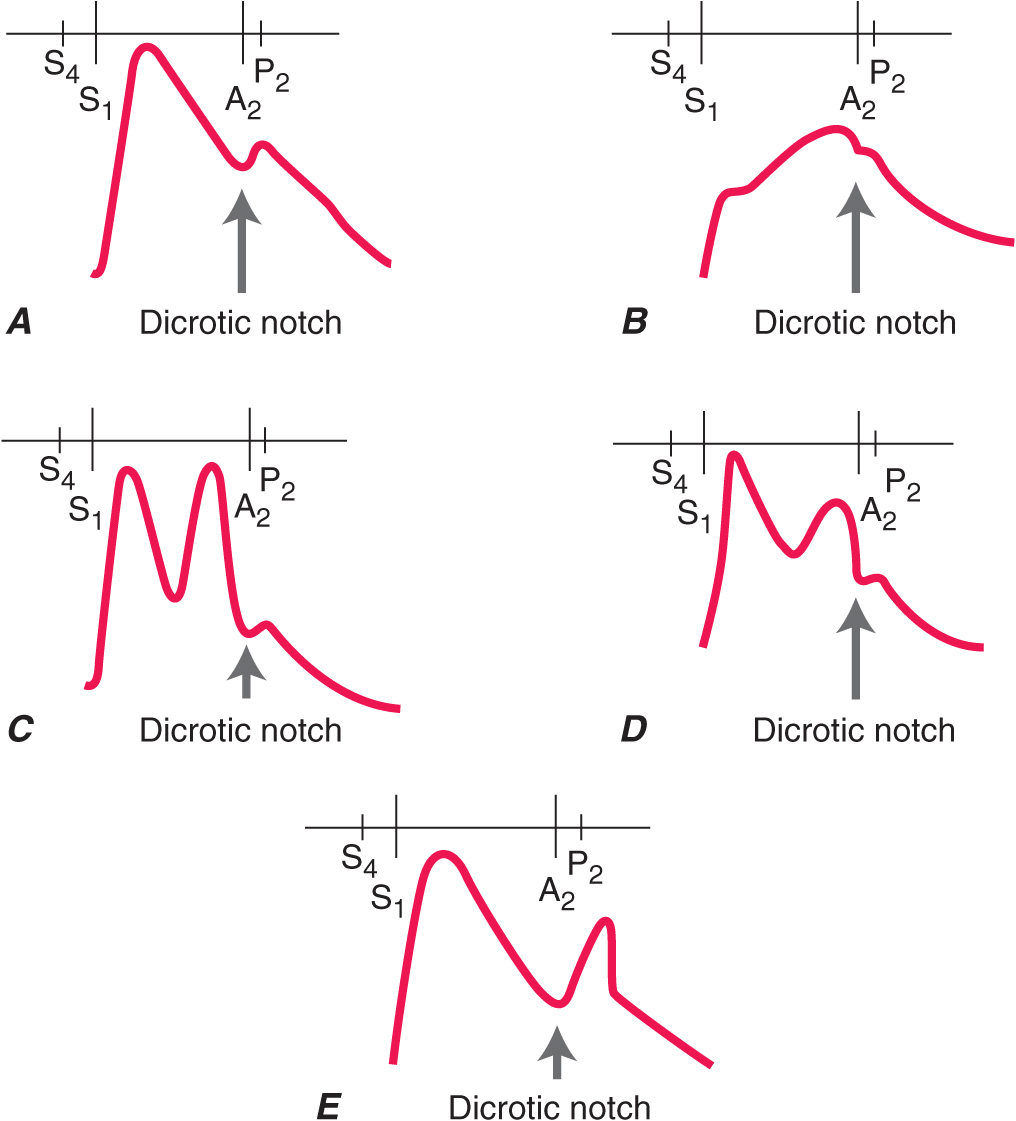
The character of the pulse is best appreciated at the carotid level (Fig. 9-2). A weak and delayed pulse (pulsus parvus et tardus) defines severe aortic stenosis (AS). Some patients with AS may also have a slow, notched, or interrupted upstroke (anacrotic pulse) with a thrill or shudder. With chronic severe AR, by contrast, the carotid upstroke has a sharp rise and rapid fall-off (Corrigan’s or water-hammer pulse). Some patients with advanced AR may have a bifid or bisferiens pulse, in which two systolic peaks can be appreciated. A bifid pulse is also described in patients with hypertrophic obstructive cardiomyopathy (HOCM), with inscription of percussion and tidal waves. A bifid pulse is easily appreciated in patients on intraaortic balloon counter-pulsation (IABP), in whom the second pulse is diastolic in timing.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree