Note: This chapter is adapted from Oh JK, Seward JB, Tajik AJ. The Echo Manual. 3rd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2006, with permission.
The evaluation of pericardial disorders is of increasing importance in the management of patients with congenital heart disease. Previous cardiac surgery now accounts for over 25% of cases of constrictive pericarditis, and diagnosis can be challenging, especially in patients with congenital heart disease and coexisting myocardial dysfunction. This chapter will outline the echocardiographic evaluation of pericardial disorders, including the distinction between constrictive pericarditis and restrictive myocardial disease.
Normal pericardium consists of an outer sac, the fibrous pericardium, and an inner double-layered sac, the serous pericardium. The visceral layer of the serous pericardium, or epicardium, covers the heart and proximal great vessels. It is reflected to form the parietal pericardium, which lines the fibrous pericardium (Fig. 24.1). The pericardium provides mechanical protection for the heart and lubrication to reduce friction between the heart and surrounding structures. The pericardium also has a significant hemodynamic impact on the atria and ventricles. The nondistensible pericardium limits acute distention of the heart. Ventricular volume is greater at any given ventricular filling pressure with the pericardium removed than with the pericardium intact. The pericardium also contributes to diastolic coupling between two ventricles: the distention of one ventricle alters the filling of the other, an effect that is important in the pathophysiology of cardiac tamponade and constrictive pericarditis. Ventricular interdependence becomes more marked at high ventricular filling pressures. Abnormalities of the pericardium can range from the pleuritic chest pain of pericarditis to marked heart failure and even death from tamponade or constriction.
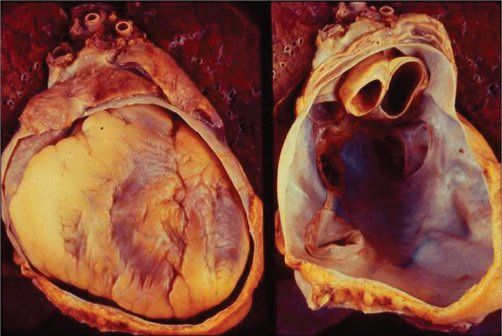
Figure 24.1. Pathology specimens demonstrating the double-layered pericardium with and without the heart in the fibrous pericardial cavity. (Courtesy of William Edwards, MD.)
Echocardiography is the most important clinical tool in the diagnosis and management of various pericardial diseases. Pericardial effusion, tamponade, pericardial cyst, and absent pericardium are readily recognized on two-dimensional (2D) echocardiography. The detection of pericardial effusion was clinically very difficult before the advent of echocardiography and was one of the most exciting initial clinical applications of cardiac ultrasonography 40 years ago. When a pericardial effusion needs to be drained, pericardiocentesis can be performed most safely under the guidance of 2D echocardiography. Although it usually is difficult to establish the diagnosis of constrictive pericarditis with 2D echocardiography alone, the characteristic respiratory variation in mitral inflow and hepatic vein Doppler velocities and tissue Doppler recording of mitral annulus velocity have added reliability and confidence to the noninvasive diagnosis of constrictive pericarditis. Transesophageal echocardiography (TEE) is helpful in measuring pericardial thickness, in evaluating diastolic function (for tamponade or constrictive physiology) from the pulmonary vein, and in detecting loculated pericardial effusion or other structural abnormalities of the pericardium. The various applications of echocardiography in the evaluation of pericardial diseases are illustrated in this chapter.
CONGENITALLY ABSENT PERICARDIUM
The congenital absence of the pericardium usually involves the left side of the pericardium. Complete absence of the pericardium on the right side is uncommon. The defect is more frequent in males, and it rarely creates symptoms such as chest pain, dyspnea, or syncope. Because of the pericardial defect, cardiac motion is exaggerated, especially the posterior wall of the left ventricle. The entire cardiac structure is shifted to the left; hence, the right ventricular (RV) cavity appears enlarged from the standard parasternal windows, mimicking the RV volume overload pattern on echocardiography. The absence of pericardium should be considered when the right ventricle appears enlarged from the parasternal window and is at the center of the usual apical image (Fig. 24.2A). This condition is also associated with a high incidence of atrial septal defect, bicuspid aortic valve, and bronchogenic cysts. It is readily recognized because of its typical 2D echocardiographic features, and the diagnosis can be confirmed with computed tomography (CT) or magnetic resonance imaging (MRI) (Fig. 24.2B).
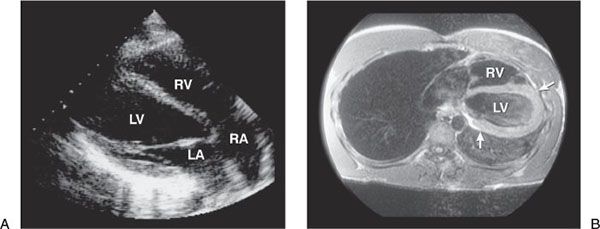
Figure 24.2. Patient with a congenitally absent pericardium. A: Two-dimensional still-frame obtained from the normal apical position. Because of the leftward shift of the heart, the right ventricle (RV) is at the center of the apical image rather than the left ventricular (LV) apex; this is often confused with RV volume overload. Cardiac catheterization was performed elsewhere to evaluate an atrial septal defect and showed no shunt before this evaluation. RA, right atrium; LA, left atrium. B: Magnetic resonance image of the chest showing a marked shift of the heart to the left side of the chest because of the partial absence of the pericardium on the left side. Arrows indicate area of absent pericardium.
Pericardial Cyst
A pericardial cyst typically is a benign structural abnormality of the pericardium that usually is detected as an incidental mass lesion on a chest radiograph or as a cystic mass on echocardiography (Fig. 24.3A). Most frequently, pericardial cysts are located in the right costophrenic angle, but they also are found in the left costophrenic angle, hilum, and superior mediastinum. Pericardial cysts need to be differentiated from malignant tumors, cardiac chamber enlargement, and diaphragmatic hernia. Two-dimensional echocardiography is useful in differentiating a pericardial cyst from other solid structures, because a cyst is filled with clear fluid and appears as an echo-free structure. It also has a characteristic appearance on CT or MRI (Fig. 24.3B).
Pericardial Effusion/Tamponade
When the potential pericardial space is filled with fluid or blood, it is detected as an echo-free space. When the amount of effusion is greater than 25 mL, an echo-free space persists throughout the cardiac cycle. A smaller amount of pericardial effusion may be detected as a posterior echo-free space that is present only during the systolic phase. As pericardial effusion increases, movement of the parietal pericardium decreases. When the amount of pericardial effusion is massive, the heart may have a “swinging” motion in the pericardial cavity (Fig. 24.4A), which is responsible for the electrocardiographic manifestation of cardiac tamponade, “electrical alternans” (Fig. 24.4B). The swinging motion is not always present in cardiac tamponade, however. Cardiac tamponade can occur with a small amount of pericardial effusion, if the accumulation of pericardial effusion happens rapidly. A clinical example is myocardial perforation after acute myocardial infarction or during implantation of a pacemaker. Various M-mode and 2D echocardiographic signs have been reported in this life-threatening condition: early diastolic collapse of the right ventricle, late diastolic right atrial (RA) inversion, abnormal ventricular septal motion, respiratory variation in ventricle chamber size (Fig. 24.5), and plethora of the inferior vena cava with blunted respiratory changes. These findings are caused by the characteristic hemodynamics of tamponade. Diastolic collapse of the right atrium and right ventricle is related to intrapericardial pressure rising above the intracardiac pressures, and abnormal ventricular septal motion is related to respiratory variation in ventricular filling. Diastolic collapse of the right heart may not occur if right heart pressure is elevated. In case of acute myocardial rupture or proximal aortic dissection, clotted blood may be seen in the pericardial sac; this finding is highly suggestive of hemopericardium (Fig. 24.6). When there is air in the pericardial sac (pneumopericardium) as a result of esophageal perforation, cardiac imaging (both transthoracic echocardiography (TTE) and TEE) is difficult because ultrasound does not penetrate air well.
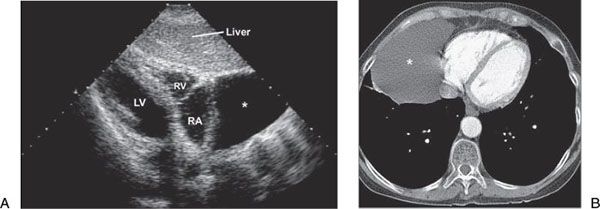
Figure 24.3. Pericardial cyst. A: Subcostal view showing a large pericardial cyst (asterisk) adjacent to the right atrium (RA). It has a typical echo-free appearance with a smooth boundary. LV, left ventricle. RV, right ventricle. B: MRI of the same patient as in A with a large pericardial cyst (asterisk).
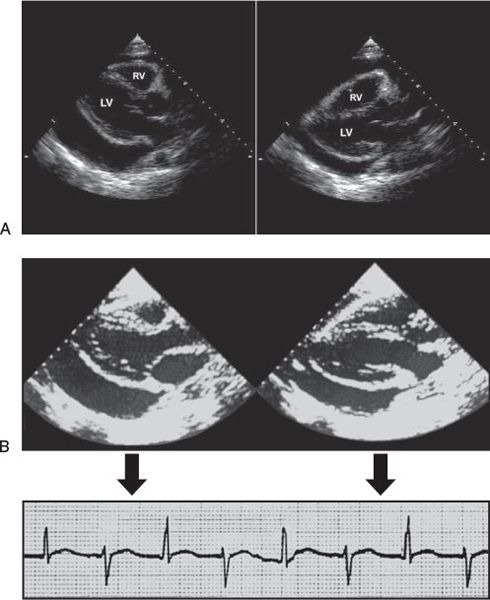
Figure 24.4. Pericardial effusion. A: Parasternal long-axis views showing a large pericardial effusion with a swinging motion of the heart. LV, left ventricle. B: With a large amount of pericardial effusion, the heart has a swinging motion, which is an ominous sign of cardiac tamponade. When the left ventricular cavity is close to the surface (left), the QRS voltage increases on the electrocardiogram, but it decreases when the LV swings away from the surface (right), producing electrical alternans.
Doppler echocardiographic features of pericardial effusion/tamponade are more sensitive than the 2D echocardiographic features mentioned earlier. The Doppler findings of cardiac tamponade are based on the following characteristic respiratory variations in intrathoracic and intracardiac hemodynamics (Fig. 24.7). Normally, intrapericardial pressure (hence, left atrial [LA] and left ventricular [LV] diastolic pressures) and intrathoracic pressure (hence, pulmonary capillary wedge pressure) fall the same degree during inspiration, but in cardiac tamponade intrapericardial (and intracardiac) pressure falls substantially less than intrathoracic pressure. Therefore, the LV filling pressure gradient (from pulmonary wedge pressure to LV diastolic pressure [the shaded area in Fig. 24.7]) decreases with inspiration. Consequently, mitral valve opening is delayed, which lengthens the isovolumic relaxation time (IVRT) and decreases mitral E velocity. In cardiac tamponade, the degree of ventricular filling depends on the other ventricle because of the relatively fixed combined cardiac volume (ventricular interdependence); thus, reciprocal changes occur in the right heart chambers (Fig. 24.8). Increased venous return to the right heart chambers with inspiration also contributes to the increased ventricular interdependence.
The respiratory flow velocity changes across the mitral and tricuspid valves are also reflected in the pulmonary and hepatic venous flow velocities, respectively: inspiratory decrease and expiratory increase in pulmonary vein diastolic forward flow, and expiratory decrease in hepatic vein forward flow and increase in expiratory reversal flow (Fig. 24.9).
Echocardiographically Guided Pericardiocentesis
The most effective treatment for cardiac tamponade is removal of the pericardial fluid. Although pericardiocentesis is lifesaving, a blind percutaneous attempt has a high rate of complications, including pneumothorax, puncture of the cardiac wall, or death. Two-dimensional echocardiography can guide pericardiocentesis by locating the optimal site of puncture (Fig. 24.10), by determining the depth of the pericardial effusion and the distance from the puncture site to the effusion, and by monitoring the results of the pericardiocentesis, usually from the subcostal view. The position of the pericardiocentesis needle can be checked by imaging with administration of agitated saline. Figure 24.11 demonstrates contrast (arrows) in the pericardial space, not in the right ventricle. Under most circumstances, a pigtail (6 or 7 French) catheter is introduced and left in the pericardial sac for several days with intermittent drainage (every 4 to 6 hours), which has markedly curtailed the rate of recurrent effusion and use of a sclerosing agent. At Mayo Clinic, most pericardiocentesis procedures are performed with the guidance of 2D echocardiography. The most common location of needle entry is in the parasternal area, but this depends on 2D echocardiographic findings. In our consecutive 1127 echo-guided pericardiocenteses, malignant effusion was most common (34%), followed by postoperative (25%), and complication of catheter-bases procedures (10%) (Fig. 24.12). The procedure was successful in 97%, and complications occurred in 4.4%, mostly minor. Major complications were death (1 patient), cardiac laceration (5), vessel laceration (1), pneumothorax (5), infection (1), and sustained ventricular tachycardia (1).
PERICARDIAL EFFUSION VERSUS PLEURAL EFFUSION
A pericardial effusion usually is located circumferentially. If there is an echo-free space anteriorly only, it more likely is an epicardial fat pad than pericardial effusion. Posteriorly, a pericardial effusion is located anterior to the descending thoracic aorta, whereas a pleural effusion is present posterior to the aorta (Fig. 24.13). Two-dimensional ultrasonographic imaging of pleural effusion is also helpful in planning for a thoracentesis to locate the optimal puncture site. Pleural effusion on the left side allows cardiac imaging from the back (Fig. 24.14).
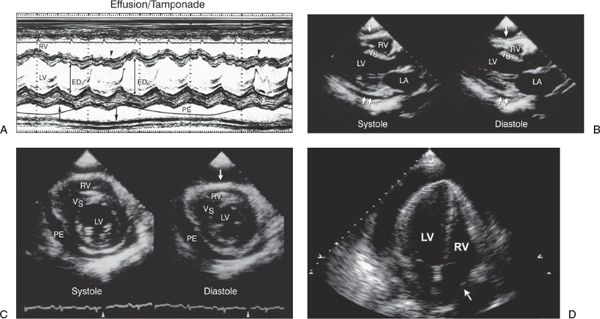
Figure 24.5. Pericardial effusion with tamponade. A: M-mode echocardiogram from the parasternal window in a patient with cardiac tamponade and large circumferential pericardial effusion (PE). The M-mode was recorded simultaneously with the respirometer tracing at the bottom (upward arrow indicates onset of inspiration and downward arrow indicates onset of expiration). The left ventricular (LV) dimension during inspiration (EDi) becomes smaller than with expiration (EDe). The opposite changes occur in the right ventricle (RV). The ventricular septum (arrowheads) moves toward the LV with inspiration and toward the RV with expiration, accounting for the abnormal ventricular septum in patients with cardiac tamponade. Parasternal long-axis (B) and short-axis (C) views of a patient with tamponade during systole and diastole. The pericardial effusion (double arrows in B; PE in C) appears small in the long axis and moderate in the short axis during systole, but during early diastole, the RV free wall collapses (arrow at top). VS, ventricular septum. D: Apical four-chamber view demonstrating late diastolic (arrowhead on electrocardiograph) collapse of right atrium (RA) wall (arrow). This sign is sensitive but not specific for tamponade. When RA inversion lasts longer than a third of the RR interval, it is specific for hemodynamically significant pericardial effusion.
Constrictive Pericarditis
Constrictive pericarditis is caused by thickened, inflamed, adherent, or calcific pericardium limiting the diastolic filling of the heart (Fig. 24.15). Constrictive pericarditis is not an uncommon condition but frequently escapes clinical detection because it is not clinically considered in many cases and no one diagnostic test alone can ensure the diagnosis of constrictive pericarditis with high confidence. Since this is a curable entity causing severe heart failure, constrictive pericarditis should be considered in all patients with heart failure, especially when systolic function is normal and/or there is a predisposing factor. Currently, previous cardiac surgery is the most common cause for constriction, followed by pericarditis, episodes of pericardial effusion, and radiation therapy. Figure 24.16 demonstrates the multiple underlying etiologies of constriction in more than 400 patients who underwent pericardiectomy since 1985 at the Mayo Clinic. Patients with constrictive pericarditis present with dyspnea, peripheral edema, ascites, pleural effusion, fatigue, or anasarca. Jugular venous pressure is almost always elevated with typical rapid “y” descent (Fig. 24.17). Kussmaul sign and pericardial knock are other typical physical findings. Because of their abdominal symptoms and elevation of liver enzymes because of hepatic venous congestion, many patients are labeled as having a liver or gastrointestinal disease and undergo noncardiac procedures such as liver biopsy, endoscopy, or even abdominal exploration before the diagnosis of constrictive pericarditis is made. The diagnosis of pericardial constriction can be particularly challenging in patients with operated or unoperated congenital heart disease as a result of coexisting lesions and myocardial dysfunction, making the correct diagnosis more elusive. In many patients with congenital heart disease and pericardial constriction, restrictive myocardial disease and residual hemodynamic lesions may contribute to the clinical presentation and findings.
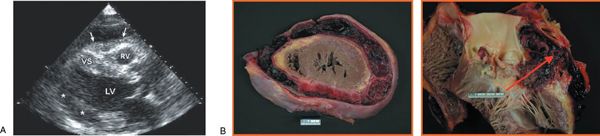
Figure 24.6. Hemopericardium. A: This echocardiogram was obtained from a 73-year-old man who is septic with Streptococcus mitis and is hypotensive. There was a moderate amount of circumferential pericardial effusion with soft-tissue density (arrows) over the right ventricle (RV), characteristic for coagulate tamponade or hemopericardium. B: Soon after this echocardiogram, the patient died. Pathology showed hemopericardium (left) because of a perforation of the proximal aorta (arrow) with aortic valve endocarditis (right).
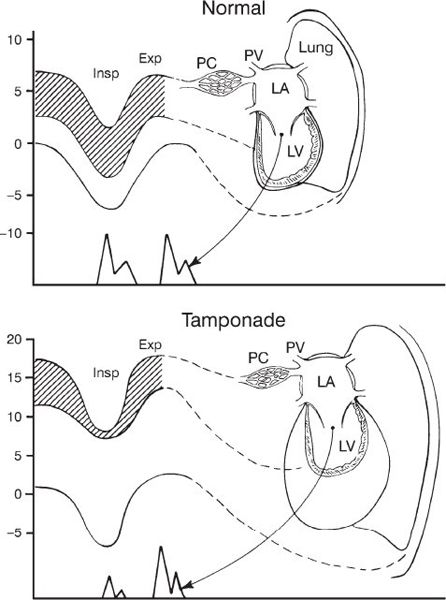
Figure 24.7. Diagram of intrathoracic and intracardiac pressure changes with respiration in normal and tamponade physiology. The shaded area indicates left ventricle (LV) filling pressure gradients (difference between pulmonary capillary wedge pressure and LV diastolic pressure). At the bottom of each drawing is a schematic mitral inflow Doppler velocity profile reflecting LV diastolic filling. In tamponade, there is a decrease in LV filling after inspiration (Insp) because the pressure decrease in the pericardium and LV cavity is smaller than the pressure fall in the pulmonary capillaries (PC). LV filling is restored after expiration (Exp). PV, pulmonary vein. (Modified from Sharp JT, Bunnell IL, Holland JF, et al. Hemodynamics during induced cardiac tamponade in man. Am J Med. 1960;29:640–646.)
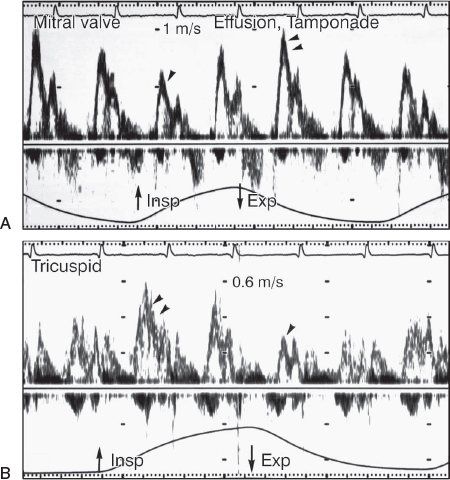
Figure 24.8. Typical pulsed-wave Doppler pattern of tamponade recorded with a nasal respirometer. A: Mitral inflow velocity decreases (single arrowhead) after inspiration (Insp) and increases (double arrowheads) after expiration (Exp). B: Tricuspid inflow velocity has the opposite changes. E velocity increases (double arrowheads) after inspiration and decreases (single arrowhead) after expiration. (From Oh JK, Hatle LK, Mulvagh SL, Tajik AJ. Transient constrictive pericarditis: diagnosis by two-dimensional Doppler echocardiography. Mayo Clin Proc. 1993;68:1158–1164.)
Pericardial calcification on chest radiography is helpful but is present in only 23% of cases (Fig. 24.18). Thickened pericardium is a usual finding in this condition, but pericardial thickness may be normal in up to 20% of cases. Pericardial thickness can be evaluated by echocardiography (most accurately by TEE) and by CT and MRI. In patients with congenital heart disease and univentricular physiology, the evaluation of pericardial thickness is of increased importance in suspected constrictive pericarditis, given the inability to evaluate interventricular hemodynamics. Traditional invasive hemodynamic features do have a large overlap with those found in restrictive cardiomyopathy or other myocardial diseases. New insights into the mechanism of constrictive pericarditis have allowed development of more reliable and specific diagnostic criteria for constriction using comprehensive echocardiography including 2D, Doppler, and tissue Doppler imaging. Subsequent to this observation, new diagnostic criteria of invasive hemodynamic features of constriction have been proposed.
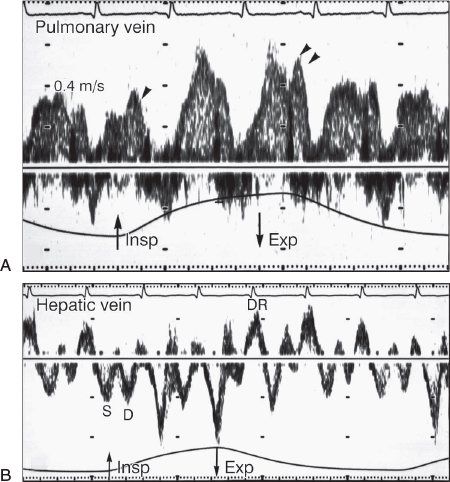
Figure 24.9. Pulmonary vein and hepatic vein Doppler patterns of tamponade. A: Diastolic forward pulmonary venous flow decreases (single arrowhead) after inspiration (Insp) and increases (double arrowheads) after expiration (Esp). B: The hepatic vein has a significant reduction in diastolic forward flow and an increase in diastolic reversals (DR) after expiration. D, diastolic flow; S, systolic flow. (From Oh JK, Hatle LK, Mulvagh SL, Tajik AJ. Transient constrictive pericarditis: diagnosis by two-dimensional Doppler echocardiography. Mayo Clin Proc. 1993;68:1158–1164.)
The M-mode (Fig. 24.19) and 2D echocardiographic features of constrictive pericarditis include thickened pericardium, abnormal ventricular septal motion, flattening of the LV posterior wall during diastole, respiratory variation in ventricular size, and a dilated inferior vena cava, but these findings are not sensitive or specific. Hatle et al. described the Doppler features typical of constriction, which are distinct from those of restrictive hemodynamics. Although the underlying pathologic mechanism is different from that of cardiac tamponade, the hemodynamic events of constriction in regard to respiratory variation in LV and RV filling are similar to those of tamponade.
To establish the diagnosis of constrictive pericarditis, the following two hemodynamic characteristics need to be demonstrated either by 2D/Doppler echocardiography or by cardiac catheterization:
1.Disassociation between intrathoracic and intracardiac pressures
2.Exaggerated ventricular interdependence
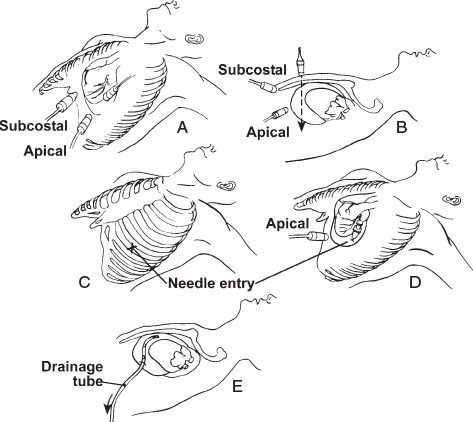
Figure 24.10. Echocardiographically guided pericardiocentesis.
STEP 1. Locate an area on the chest or subcostal region from which the largest amount of pericardial effusion can be visualized, and mark it (A–C).
STEP 2. Determine the depth of effusion from the marked position and the optimal angulation.
STEP 3. After sterile preparation and local anesthesia, perform pericardiocentesis (D).
STEP 4. When in doubt about the location of the needle, inject saline solution through the needle and image it from a remote site to locate the bubbles.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


