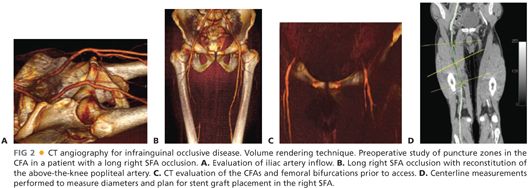
■ Computed tomographic arteriography (CTA): CTA has assumed an increasing role in guiding peripheral vascular intervention, particularly in regard to choosing appropriate devices and optimal interventional approach (e.g., ipsilateral antegrade vs. contralateral retrograde). This additional guidance, however, comes at the cost of substantially more iodinated contrast and radiation exposure than that provided by catheter-directed, intraarterial contrast arteriography, augmented by direct ultrasonic visualization and physiologic testing (FIG 2).
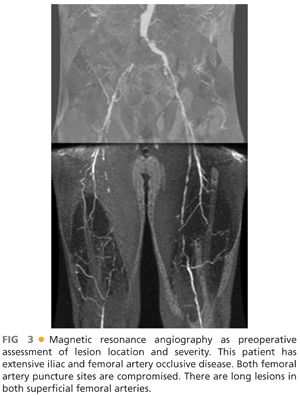
■ Magnetic resonance arteriography (MRA): MRA may also assist preoperative planning. Although MRA does not expose patients to ionizing radiation, artifactual overestimation of disease severity is common in low flow conditions. Also, gadolinium contrast administration is contraindicated in patients with a glomerular filtration rate of less than 30 mL per minute due to risk of contrast-associated glomerulosclerosis (FIG 3).
SURGICAL MANAGEMENT
■ Overview—Success in percutaneous management of femoral–popliteal occlusive disease has catalyzed a reciprocal decline in the use of open bypass for definitive revascularization. Key features of percutaneous management include detailed preoperative planning, choice of access site and closure techniques, and familiarity and facility with a wide range of complementary intraluminal wire-guided devices.4
Preoperative Planning
■ The operative plan includes access site selection, planned method of crossing, and options for arterial reconstruction.
■ Endovascular inventory: An essential element of endovascular success is a robust and redundant device inventory. In contrast to open reconstruction techniques, where similar instruments will suffice for all lower extremity bypass configurations, regardless of routing, a unique and task-specific repertoire is required for almost every endovascular approach. Procedural success requires that the necessary devices, including guidewires, sheaths, catheters, angioplasty balloons, stents, reentry devices, stent grafts, and atherectomy catheters are identified and available before intervention is attempted.
■ Appropriate radiation protection must be available for all individuals involved in interventional procedures. All team members must conscientiously wear a radiation dosimeter, submitted monthly for aggregate exposure documentation. Leadership ensures that all team members adhere to basic radiation safety tenants, including limiting the length and intensity of exposure to the minimum required for precision imaging and intervention (the “as low as reasonably achievable” [ALARA] principle). Safety principles, including distance from the radiation source, appropriate shielding and optimal table height, and source-image intensifier distance, must be understood and applied during every procedure.
■ Antibiotic prophylaxis is administered prior to the initiation of the procedure, whenever permanent implants are considered.
■ Percutaneous procedures are performed under local anesthesia with appropriate sedation. Care should be taken to avoid oversedation to ensure that patients can cooperate with instructions and imaging requirements during the procedure. When hybrid open endovascular procedures are contemplated, general anesthesia may facilitate more rapid and accurate device deployment, with reciprocally less radiation exposure for the patient, cath lab team, and operator.
■ An important initial consideration is the approach and optimal puncture site. The common femoral artery (CFA) is the most frequent access site. The approach is typically either up and over the aortic bifurcation from the contralateral femoral artery or ipsilateral antegrade femoral puncture. The transbrachial, transthoracic approach may also provide optimal antegrade access under certain circumstances.
Positioning
■ Surgeon position should provide forehand access, whenever possible (FIG 4).
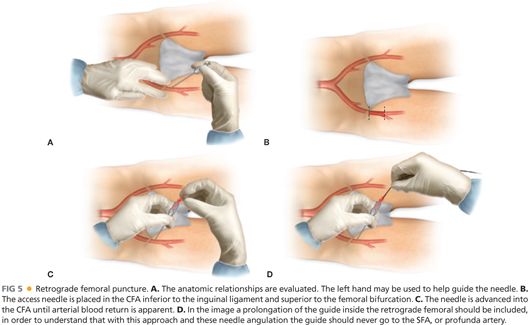
■ Retrograde femoral puncture: This is the most common type of access for all endovascular procedures, including femoral–popliteal revascularization (FIG 5). The needle is placed in the CFA and the guidewire is advanced retrograde into the iliac artery.
■ The femoral area is examined prior to puncture of the artery. The inguinal ligament extends from the anterior superior iliac spine to the pubic tubercle. The best puncture site is inferior to the inguinal ligament and at least a centimeter superior to the femoral bifurcation. Ultrasound provides useful guidance for arterial puncture (FIG 6). Following needle insertion, spot fluoroscopy from an ipsilateral oblique angle is obtained to confirm position. If arterial insertion is determined to be proximal to the femoral head, the access attempt is aborted before larger devices are inserted to minimize the risk of retroperitoneal hematoma formation due to inadequate compression or control following the procedure. Common femoral access also enables closure devices to be employed with confidence when necessary.
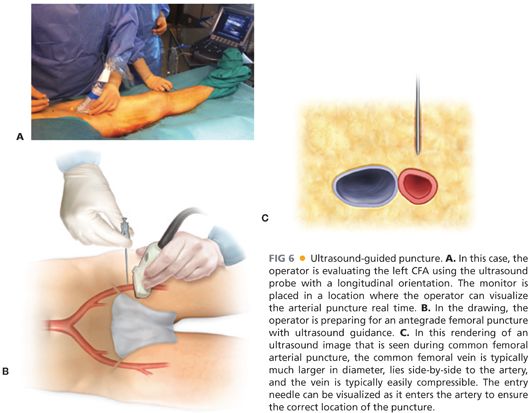
■ Closure devices: recommended for retrograde femoral access site management following insertion of greater than or equal to 6-French (Fr) sheaths. Sheath puncture less than 6 Fr is best managed by compression for 10 to 15 minutes, with or without adjuncts such as a thrombin-impregnated dressing (e.g., D-stat® patch).
■ When pulses are not palpable at the desired access site, ultrasound or fluoroscopic guidance (assisted by mural femoral artery calcification) may provide valuable assistance. Under these circumstances, bilateral femoral access and ipsilateral iliac intervention may be required for procedural success. Ideally, this eventuality is anticipated based on the results of preprocedural examination and physiologic testing. Fortunately, the pulseless femoral artery is often palpable based on mural calcification alone. Patience and spot fluoroscopic images to confirm needle and artery position following failed needle passes often ensures ultimate success.
■ Secondary puncture of the postoperative groin presents special challenges. Whenever possible, scar tissue and anastomoses should be avoided. Access in native artery is preferable to prosthetic or autogenous grafts. Considerable force may be required for needle and micropuncture set access; consideration should be given to using “stiff” 0.018-in wires and micropuncture sets specifically manufactured to facilitate difficult groin access.
■ Antegrade femoral puncture: The femoral pulse and inguinal ligament are carefully marked (FIG 7). Needle placement is directed proximal to the femoral artery bifurcation under real-time ultrasound guidance.
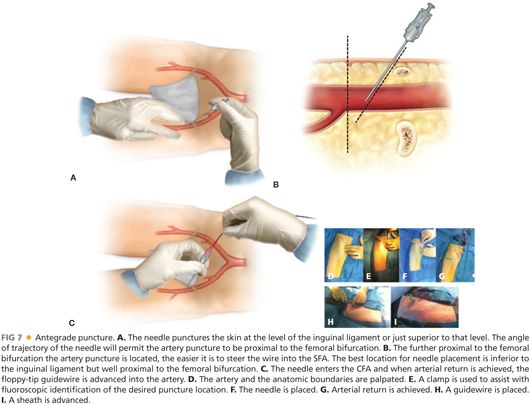
■ Guidewire placement into the superficial femoral artery (SFA) requires patience and practice. Ultrasound imaging in a longitudinal view may facilitate SFA wire intubation. When using a micropuncture set with a “steerable” 0.018-in wire (e.g., one with a slight curve placed at the tip), fluoroscopic control may also be employed. If repeated attempts result in deep femoral artery placement, the micropuncture set should be exchanged for an 11 cm 4- or 5-Fr sheath over a standard multipurpose (e.g., Bentsen) wire. Once safe antegrade deep femoral access is obtained, the 5-Fr sheath may be gradually withdrawn with sequential fluoroscopic contrast “puffs” of 1 mL or less performed until the femoral bifurcation is imaged (but while the sheath tip is still in the CFA). At this juncture, roadmapping or last-image-hold digital subtraction angiography from an ipsilateral oblique angle is performed to outline femoral bifurcation anatomy, after which a steerable hydrophilic guidewire and, ultimately, the 5-Fr sheath is directed under fluoroscopic imaging into the SFA.
■ Antegrade femoral access should be avoided in the obese, in patients with a short CFA, or in patients with extreme proximal or orificial SFA disease. Although antegrade access improves “pushability” across total occlusions and enables usage of a wider inventory of guidewire-catheter combinations, there is no option for inflow disease management using this approach. Also, the safety of current generation closure device placement is uncertain in antegrade approaches and should be avoided whenever possible.
■ Brachial puncture and transaortic sheath placement may provide an alternative option for “antegrade” femoral access. Upper extremity arteries are smaller, less forgiving, more prone to spasm, and less predictably managed with compression following access. Notoriously, small amounts of arterial extravasation may catalyze debilitating and permanent neurapraxia, even when brachial access is obtained well distal to the axillary fossa. Debilitating nerve injury from “axillary” sheath hematomas may occur at any location proximal to the antecubitum. In our practice, we minimize this risk by defaulting to surgical exposure and direct arterial puncture with suture closure for essentially all brachial artery access procedures. Exposure is easily obtained with local anesthetic in most patients.
■ The longer guidewires and catheters required to access the femoral and popliteal arteries are also less responsive to surgeon manipulation from a brachial approach and also limit the available inventory of appropriate devices for femoral or popliteal intervention.
■ When brachial access is required, the level of access is determined by the diameter of the largest sheath required to complete the procedure. For 6- or 7-Fr sheaths, the segment immediately proximal to the antecubital fossa is sufficient. For larger sheaths, access should be obtained in the distal axillary artery, proximal to the bifurcation of the deep brachial artery. The left arm should be used whenever possible to minimize risk for embolic iatrogenic stroke. During micropuncture access, even under direct vision, back-bleeding may not be pulsatile due to the smaller caliber of the brachial artery. Sheaths should be managed with frequent flushing with 100 units/mL heparin, as well as systemic anticoagulation once definitive interventional sheaths (6 to 7 Fr, 55 to 90 cm from the arm) are positioned in the target artery, or whenever sheaths appear to be occlusive. Intraarterial nitroglycerine injection may reduce arterial vasospasm to the distal extremity when necessary.
TECHNIQUES
■ Percutaneous femoral–popliteal revascularization techniques include balloon angioplasty alone, or self-expanding stent graft implantation as an adjunct to angioplasty. These techniques require placement of interventional-grade sheaths, braided when required to cross the aortic arch or bifurcation to prevent kinking as well as sufficient length to reach the treatment site without limiting device selection. Sheath access also permits serial angiographic imaging to guide device positioning, deployment, and confirm procedural success.
SHEATH PLACEMENT
■ Ensure that the appropriate sheath is selected and that alternatives are available should plans or needs change.
■ Decide on optimal sheath positioning. Usually, placement immediately adjacent to the target lesion maximizes the ability to cross the lesion, control the procedure, and minimize contrast usage. In many circumstances, it may not be possible or practical to advance the sheath past the femoral bifurcation when approaching from the contralateral femoral artery.
Up and Over Approach
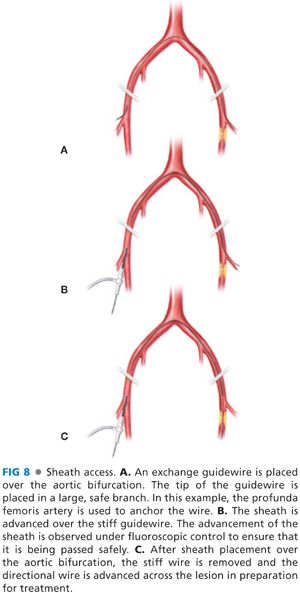
■ Crossing the aortic bifurcation (FIG 8): Through a contralateral retrograde puncture, an infrarenal aortogram is performed to evaluate aortic bifurcation anatomy and location. Usually located at the level of the iliac crest, vascular calcifications may outline the bifurcation and guide positioning. The aortic bifurcation must be free of occlusive or aneurysmal disease to ensure safe sheath passage. Occasionally, iliac artery lesions must be treated prior to femoral intervention to ensure optimal outcome.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


