INDICATIONS
Indications for LVRS in the modern era are derived from the results of the National Emphysema Treatment Trial (NETT). The largest randomized clinical trial (RCT) dealing with LVRS, the goals of NETT were to identify the effects of treatment as well as the optimal candidates for intervention. NETT involved 17 centers and enrolled 1,218 patients with end-stage emphysema. The first publication from the study occurred after noting a specific high-risk subset of patients—those with FEV1 <20% with homogenous disease or a diffusing capacity or transfer factor of the lung for carbon monoxide (DLCO) <20%—at the 6-month follow-up had high mortality with surgery, with minimal improvement. After this high-risk group was excluded, the major findings were derived from the remaining 1,078 patients. Mid-term and long-term results demonstrated clinically significant improvements in spirometry, lung volumes, diffusing parameters, oxygenation, quality of life, dyspnea, exercise capacity, and long-term survival. Two key predictive factors were identified in NETT: Distribution of emphysema and the level of baseline exercise capacity. Bilateral LVRS provides clinically significant improvements in quality of life for patients with heterogenous upper lobe predominant emphysema, with additional survival benefit in the low exercise capacity subset of these patients.
The Centers for Medicare & Medicaid Services (CMS) has based its reimbursement policies wholly on the results of NETT and thus will pay for bilateral LVRS in patients with advanced upper lobe predominant emphysema that meet NETT criteria. They will not pay for the procedure in those with lower lobe disease, alpha-1 antitrypsin deficiency, homogenous disease, extremely advanced disease (both FEV1 <20% and DLCO <20% predicted) and those with a contraindication for bilateral procedures (asymmetric unilateral disease or those with bilateral disease and a history of thoracotomy). CMS recommends that procedures be performed at facilities that fulfill one of the following criteria:
 Certified by the Joint Commission on Accreditation of Healthcare Organizations (JCAHO) under the LVRS disease-specific care certification program
Certified by the Joint Commission on Accreditation of Healthcare Organizations (JCAHO) under the LVRS disease-specific care certification program
 Or approved as Medicare lung or heart-lung transplantation hospitals
Or approved as Medicare lung or heart-lung transplantation hospitals
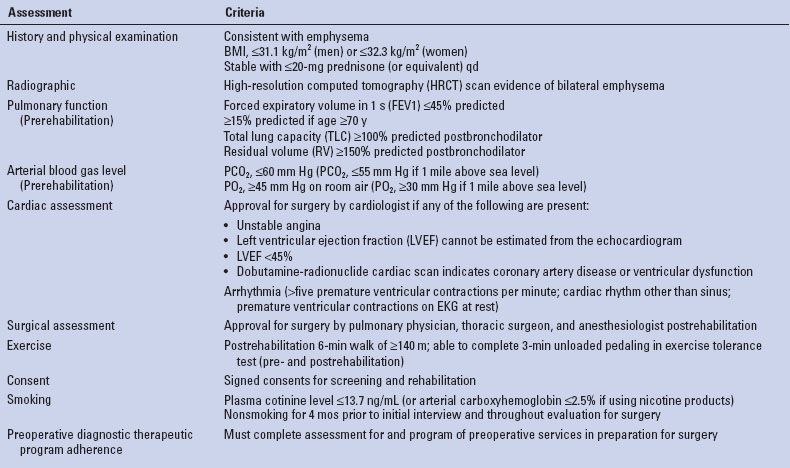
In addition, Medicare-covered LVRS approaches are limited to bilateral resections. The procedure is reimbursed if all of their criteria are met (Table 6.1) in the setting of severe upper lobe predominant emphysema (on CT scan), or severe nonupper lobe emphysema with low exercise capacity. Patients with low exercise capacity are those with maximal exercise capacity at or below 25 W for women and 40 W for men after completion of the preoperative therapeutic program in preparation for LVRS. Exercise capacity is measured by incremental, maximal, symptom-limited exercise with a cycle ergometer utilizing 5 or 10 watts/minute ramp on 30% oxygen after 3 minutes of unloaded pedaling.
TABLE 6.1 Indications for LVRS (CMS Criteria for Coverage)
 CONTRAINDICATIONS
CONTRAINDICATIONS
LVRS is not indicated in any of the following circumstances:
 Patient characteristics carry a high risk for perioperative morbidity and/or mortality.
Patient characteristics carry a high risk for perioperative morbidity and/or mortality.
 Disease is unsuitable for LVRS (i.e., homogenous disease).
Disease is unsuitable for LVRS (i.e., homogenous disease).
 Patient is unable to complete the preoperative and postoperative pulmonary diagnostic and therapeutic program required for surgery.
Patient is unable to complete the preoperative and postoperative pulmonary diagnostic and therapeutic program required for surgery.
 FEV1 ≤20% predicted + homogenous distribution of emphysema on CT scan, or DLCO ≤20% predicted (high-risk group identified in NETT).
FEV1 ≤20% predicted + homogenous distribution of emphysema on CT scan, or DLCO ≤20% predicted (high-risk group identified in NETT).
 Severe, nonupper lobe emphysema with high exercise capacity. High exercise capacity is defined as maximal workload at the completion of the preoperative diagnostic and therapeutic program that is above 25 W for women and 40 W for men.
Severe, nonupper lobe emphysema with high exercise capacity. High exercise capacity is defined as maximal workload at the completion of the preoperative diagnostic and therapeutic program that is above 25 W for women and 40 W for men.
 PREOPERATIVE PLANNING
PREOPERATIVE PLANNING
The surgery must be preceded and followed by a program of diagnostic and therapeutic services consistent with those outlined in NETT. These programs are designed to optimize patient’s health before surgery and improve the chance for successful recovery. Elements of the preoperative program include:
 Six- to ten-week series of at least 16, and not more than 20, preoperative sessions each lasting a minimum of 2 hours. The focus of the rehabilitation program is to optimize exercise capacity, achieve physical fitness to affect early postoperative mobilization and provide a baseline of optimized preoperative exercise capacity or comparison with postoperative exercise capacity.
Six- to ten-week series of at least 16, and not more than 20, preoperative sessions each lasting a minimum of 2 hours. The focus of the rehabilitation program is to optimize exercise capacity, achieve physical fitness to affect early postoperative mobilization and provide a baseline of optimized preoperative exercise capacity or comparison with postoperative exercise capacity.
 Components of the pulmonary rehabilitation program include:
Components of the pulmonary rehabilitation program include:
 Comprehensive evaluation of medical, psychosocial, and nutritional needs
Comprehensive evaluation of medical, psychosocial, and nutritional needs
 Setting of goals for education and exercise training
Setting of goals for education and exercise training
 Exercise training (lower extremity, flexibility, strengthening, and upper extremity)
Exercise training (lower extremity, flexibility, strengthening, and upper extremity)
 Education about emphysema and medical treatments
Education about emphysema and medical treatments
 Psychosocial counseling
Psychosocial counseling
 Nutritional counseling
Nutritional counseling
 The program should be arranged, monitored, and performed under the coordination of the facility where the surgery will take place.
The program should be arranged, monitored, and performed under the coordination of the facility where the surgery will take place.
 Common elements of preoperative pulmonary rehabilitation programs include controlled breathing and cough techniques, instructions in incentive spirometry exercises, and peripheral muscle exercise training consisting of aerobic work. Bronchodilator therapy may be used without steroids.
Common elements of preoperative pulmonary rehabilitation programs include controlled breathing and cough techniques, instructions in incentive spirometry exercises, and peripheral muscle exercise training consisting of aerobic work. Bronchodilator therapy may be used without steroids.
 SURGERY
SURGERY
Those who advocate for median sternotomy cite the shorter operative times (as repositioning during the case is not needed) and avoiding injury to chest wall muscles and intercostal nerves both from the operative approach and from chest tubes brought out below the costal arch.
Working IV lines, an arterial line, and a thoracic epidural are often placed at the beginning of the procedure. A working thoracic epidural is used to ensure adequate pain control thus allowing for adequate pulmonary toilet in the immediate postoperative period.
After induction of general anesthesia, the patient is first intubated with a single-lumen endotracheal tube to allow for bronchoscopy. Flexible bronchoscopy is used to suction secretions and obtain a specimen for STAT gram stain and cultures. In addition to pulmonary toilet, bronchoscopy allows for assessment of airway anatomy and to rule out any endoluminal abnormalities. The single-lumen endotracheal tube is then exchanged for a left-sided double-lumen endotracheal tube, with bronchoscopic confirmation of its position.
Figure 6.1 Positioning of patient for Transsternal LVRS.
Positioning
The patient is positioned with arms tucked, a shoulder roll to gently extend the neck, and pressure points appropriately padded (Fig. 6.1
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


