CHAPTER 18 Lung Cancer
Minimally Invasive Approaches
DEFINITION
Thoracoscopic lobectomy is defined as the anatomic resection of an entire lobe of the lung, using a videoscope and an access incision, without the use of a mechanical retractor and without rib spreading.1–4 The anatomic resection includes individual dissection and stapling of the involved pulmonary vein, pulmonary artery, and bronchus and appropriate management of the mediastinal lymph nodes, as would be performed with thoracotomy. In selected patients, thoracoscopic anatomic segmentectomy may be performed, adhering to the same oncologic principles that guide resection at thoracotomy.
Some surgeons have advocated simultaneous stapling of hilar structures with video assistance and the avoidance of rib spreading.5,6 Such an approach has been termed video-assisted simultaneously stapled lobectomy. Although this technique has been used successfully in selected patients, the reference to thoracoscopic lobectomy is limited to anatomic resection with individual vessel ligation. VATS wedge resection describes nonanatomic thoracoscopic resection of a lesion, which is considered useful for diagnostic procedures.
To be considered a viable alternative to conventional lobectomy, thoracoscopic lobectomy must be applied with the same oncologic principles: individual vessel ligation, complete anatomic resection with negative margins, complete hilar lymph node dissection, and appropriate management of the mediastinal lymph nodes. Theoretical advantages to minimally invasive resection include reduced surgical trauma, decreased postoperative pain, shorter chest tube duration, shorter length of stay, preserved pulmonary function, and superior cosmetic result when compared with lobectomy via open thoracotomy.7–9
HISTORY
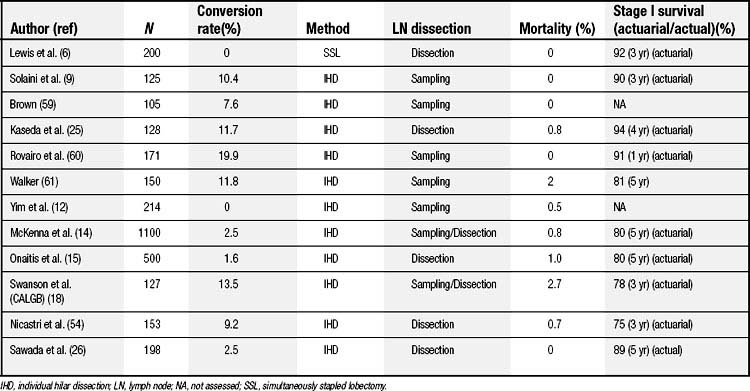
The history of minimally invasive thoracic surgery began in 1910 when Jacobeus used a cystoscope to lyse adhesions in order to collapse the lung to treat tuberculosis.10 This technique was widely applied in the early part of the century but was largely abandoned after streptomycin was introduced in 1945. However, with the emergence of laparoscopic cholecystectomy, minimally invasive approaches were applied more widely. The first descriptions of VATS to perform anatomic lobectomy were published in 1993 by Kirby and Rice1 and Walker and colleagues.2 The first randomized trial of VATS lobectomy versus the conventional open approach was presented in 1994 and demonstrated no significant benefits for VATS.11 With more widespread application of technology and refinements in technique, other groups have published series of VATS lobectomy (Table 18-1).
INDICATIONS
In general, the indications for thoracoscopic lobectomy are similar to those for lobectomy using the open approach.12–15 Thus, the procedure is applied to patients with known or suspected lung cancer (clinical stage I) if the disease appears amenable to complete resection by lobectomy. Preoperative staging and patient selection for thoracoscopic lobectomy should be conducted as for conventional thoracotomy.16
Tumor size may preclude the option of thoracoscopic lobectomy in some patients, as some large specimens may not be amenable to removal without rib spreading; however, no absolute size criteria are used. Although it is controversial, some have also argued that the thoracoscopic approach may allow recruitment and resection of some patients considered medically inoperable, who could not undergo conventional thoracotomy.17,18 A recent report demonstrates improved tolerance of thoracoscopic lobectomy as compared with thoracotomy lobectomy in patients older than 70.19 The minimal physiologic requirements for resection have not been agreed on; however, the selection of patients for thoracoscopic lobectomy must take into account that conversion to thoracotomy may be necessary.
CONTRAINDICATIONS
Absolute contraindications to thoracoscopic lobectomy include the inability to achieve complete resection with lobectomy, T3 (chest wall) or T4 tumors, active N2 or N3 disease, and inability to achieve single-lung ventilation.14,15,18 Relative contraindications include tumors visualized in the lobar orifice at bronchoscopy (although successful thoracoscopic sleeve resection has been reported20), the presence of complex, calcified benign hilar lymphadenopathy that would complicate vascular dissection, and prior thoracic irradiation. Prior thoracic surgery; T3 tumors that involve the pericardium, mediastinal pleura, or diaphragm; incomplete or absent fissures; and benign noncalcified mediastinal adenopathy should not be considered contraindications.15,17,18,21 Increasing experience has allowed successful thoracoscopic lobectomy after induction therapy, including for patients with stage IIIA (N2) disease.22 Finally, chest wall involvement would obviate thoracoscopic resection for most patients, but successful en bloc resection via VATS has been reported.23
STRATEGY FOR THORACOSCOPIC LOBECTOMY
Port placement is a matter of surgeon preference. Most surgeons use three or four incisions, although lobectomy can usually be accomplished using only two incisions.15 Using this strategy, the first incision, a 10-mm port access used predominantly for the thoracoscope, is placed in the seventh or eighth intercostal space in the midaxillary line. The location of this incision is chosen so that it does not compete with the anterior incision, yet it still provides anterior and superior visualization of the hilum. A port is used for placement of the telescope, but ports are not used for the other incisions. Before making the second incision, evidence that the patient is unresectable, such as parietal pleural involvement, should be sought.
The approach to the staging of mediastinal lymph nodes is controversial. Many advocate systematic sampling of mediastinal lymph nodes because of concerns about the adequacy and safety of formal dissection.24 Others accomplish mediastinal lymph node dissection by complete resection of the mediastinal nodes thoracoscopically, including levels 2, 4, 7, 8, and 9 on the right and levels 5, 6, 7, 8, and 9 on the left.15,25,26 A minimum of three mediastinal stations should be assessed.16
All lobectomy specimens are removed using a protective specimen bag, to prevent implantation of tumor cells in the incision. The lobectomy specimen and hilum are each inspected to ascertain that anatomic lobectomy has been performed. After retrieval, the hemithorax is irrigated with warm saline and the bronchial stump is inspected. If an air leak is encountered, repeat stapling or endoscopic suturing may be performed.27
TECHNICAL CONSIDERATIONS
Left Upper Lobectomy
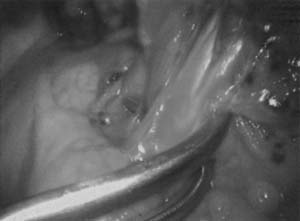
With the thoracoscope in the midaxillary incision, the horizontal and oblique fissures are inspected and the presence of the tumor in the left upper lobe is confirmed. The lung is retracted posteriorly, and the superior pulmonary vein is identified and mobilized. The left superior pulmonary vein is then encircled using a curved clamp; dissection behind the superior pulmonary vein allows identification of the pulmonary artery (Fig. 18-1). The stapling device is then applied (Fig. 18-2) and the superior pulmonary vein is divided, exposing the pulmonary artery. The pulmonary artery is mobilized, focusing on the apical and anterior branches (Fig. 18-3), which may then be stapled and divided. The left upper lobe bronchus is now visualized and may be stapled and divided (Fig. 18-4). Subsequently, the branches of the posterior and lingular arteries are stapled (Fig. 18-5). Finally, the fissures are completed and the specimen is retrieved.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree