CHAPTER 133 Image-Guided and Hybrid Surgery
Surgical Simulation
IMAGING TECHNOLOGY
Echocardiography
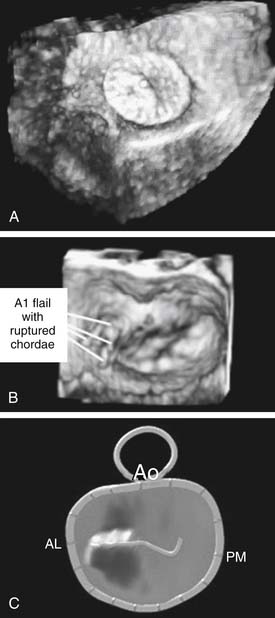
Echocardiography is a viable imaging tool for guiding beating-heart interventions. The nonionizing nature of ultrasound, the ease of data acquisition, the ability to focus on a specific anatomic structure, and a variety of additional quantification tools have enabled virtually routine application of 2D and 3D echocardiography in the operating room.1–3 Real-time 3D echocardiography (RT3DE) is a unique imaging and guiding tool, providing ample intraoperative assessment of intracardiac anatomy and enabling navigation of instruments and devices toward the target inside the beating heart (Fig. 133–1).4–9 Although intraoperative transesophageal RT3DE imaging was recently introduced, epicardial imaging remains a significant alternative for visualization of complex lesions in younger patients. Obstacles to application of 3D echocardiography as the sole guiding tool for beating-heart procedures, such as acoustic interference from metallic instruments causing shadowing and side-lobe artifacts in the field of view, have been reported. These can be minimized with instrument modifications (e.g., by applying a surface coating) and by varying the angle of instrument navigation with respect to the ultrasound probe.10,11
X-Ray Fluoroscopy
Fluoroscopy is still in most common use in the catheterization laboratory. It provides great resolution at fast frame rate (>30 Hz). However, it has significant limitations, such as absence of 3D information and lack of comprehensive representation of soft tissue. In addition, the potential harmfulness of x-rays has been widely discussed.12–14 Nevertheless, this imaging modality is still used with intraoperative angiography for additional diagnostic measurements of residual septal defects or aortic arch repairs, but it is crude and limited for other purposes (e.g., pulmonary artery plasty, unifocalization, aortopulmonary shunts).
Video-Assisted Cardioscopy
Alternative imaging techniques for visualization inside the beating heart in real time include video-assisted cardioscopy (VAC) using visible wavelength light. However, the real limitation to endoscopic intracardiac imaging has been inability to see through the blood. Although VAC offers detailed, high-magnification pictures of the target and provides greater confidence for fine manipulations of instruments, depth of field is extremely limited and the scope window must be pressed directly against the target structures for visualization.15–17 VAC is successfully used as an additional imaging tool in open-heart procedures to assess distal lesions and may assist with hybrid procedures such as pulmonary artery (PA) stent placement or apical VSD device closure.18–21
Other Imaging Modalities
Recently, fiberoptic infrared endoscopy was introduced to overcome the depth-of-field problem, because the wavelength used permits transmission through blood for a few millimeters.22 The depth of field, however, is still less than 1 to 2 cm, making navigation through the cardiac structures difficult and requiring the use of other imaging techniques such as fluoroscopy. An additional limitation of current infrared systems is that they have a relatively low frame rate, which requires significant computer processing for real-time imaging.
Multimodality Imaging and Surgical Displays
The purpose of multimodality imaging is to provide the surgeon with additional anatomic information that can be obtained with only one modality but when combined gives a more complete road map for navigation. Examples of such imaging include overlay of real-time x-ray or echocardiography information with preoperative CT or MRI patient data.23,24 Images are aligned based on anatomic features or on man-made objects, known as fiducials, that are introduced into the image. This approach is successfully implemented in arrhythmia ablation and other procedures.25–27 With advancements in computer graphics, novel surgical displays can simultaneously represent these complex 2D and 3D anatomic data from various imaging modalities, real-time instrument positioning, and patient functional information (Fig. 133–2). This combined visual information is extremely important for surgical decision making during intracardiac image-guided procedures, with the absence of direct visualization and haptic feedback.
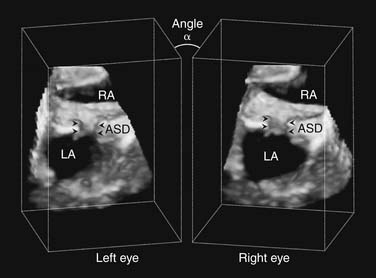
In addition to multimodality multitasking displays, tools for better comprehension of 3D visual information have been developed. Systems that mimic human-eye stereoscopic vision were first introduced for video-assisted endoscopy and implemented in minimally invasive robotically assisted surgery.28 Recently, stereoscopic vision display algorithms for 3D ultrasound imaging were introduced for beating-heart RT3DE-guided procedures (Fig. 133–3). While operating inside the beating heart, the surgeon may not always have an adequate display of intracardiac structures in 3D space and has to rely on indirect evidence for depth perception. Experimental studies showed that the use of stereoscopic displays provided significantly better spatial information and depth perception to the surgeon during beating-heart procedures, compared with conventional 2D displays.29,30
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree