Concerns exist regarding orthotropic heart transplantation in hepatitis C virus (HCV) seropositive recipients. Thus, a national registry was accessed to evaluate early and late outcome in HCV seropositive recipients undergoing heart transplant. Retrospective analysis of the United Network for Organ Sharing registry (1991 to 2014) was performed to evaluate recipient profile and clinical outcome of patients with HCV seropositive (HCV +ve) and seronegative (HCV −ve). Adjusted results of early mortality and late survival were compared between cohorts. From 23,507 patients (mean age 52 years; 75% men), 481 (2%) were HCV +ve (mean age 52 years; 77% men). Annual proportion of HCV +ve recipients was comparable over the study period (range 1.3% to 2.7%; p = 0.2). The HCV +ve cohort had more African-American (22% vs 17%; p = 0.01), previous left ventricular assist device utilization (21% vs 14%; p <0.01) and more hepatitis B core Ag+ve recipients (17% vs 5%; p <0.01). However, both cohorts were comparable in terms of extracorporeal membrane oxygenator usage (p = 0.7), inotropic support (p = 0.2), intraaortic balloon pump (p = 0.7) support, serum creatinine (p = 0.7), and serum bilirubin (p = 0.7). Proportion of status 1A patients was similar (24% HCV + vs 21% HCV −); however, wait time for HCV +ve recipients were longer (mean 23 vs 19 days; p <0.01). Among donor variables, age (p = 0.8), hepatitis B status (p = 0.4), and Center for Diseases Control high-risk status (p = 0.9) were comparable in both cohorts. At a median follow-up of 4 years, 67% patients were alive, 28% died, and 1.1% were retransplanted (3.4% missing). Overall survival was worse in the HCV+ cohort (64.3% vs 72.9% and 43.2% vs 55% at 5 and 10 years; p <0.01), respectively. Late renal (odds ratio [OR] 1.2 [1 to 1.6]; p = 0.02) and liver dysfunction (odds ratio 4.5 [1.2 to 15.7]; p = 0.01) occurs more frequently in HCV +ve recipients. On adjusted analysis, HCV seropositivity is associated with poorer survival (hazard ratio for mortality 1.4 [1.1 to 1.6]; p <0.001). In conclusion, a small proportion of patients receiving a heart transplant in the United States have hepatitis C. Despite comparable preoperative hepatic function, hepatitis C seropositive recipients demonstrate poorer long-term survival.
Hepatitis C remains one of the most common causes of chronic hepatitis in the Western world. Almost 1/3 of infected patients will progress to liver cirrhosis and eventually cancer. Till date, physicians are reluctant to list patients with hepatitis C for heart transplant. Heart failure is an important cause for stopping dual therapy in patients with hepatitis C, with isolated reports of cardiomyopathy secondary to interferon therapy. However, the introduction of direct active antivirals (DAA) has significantly improved therapy for hepatitis C virus (HCV). On this background, we analyzed the past 25-year national registry data to evaluate HCV +ve recipients undergoing heart transplant in the United States and their survival.
Methods
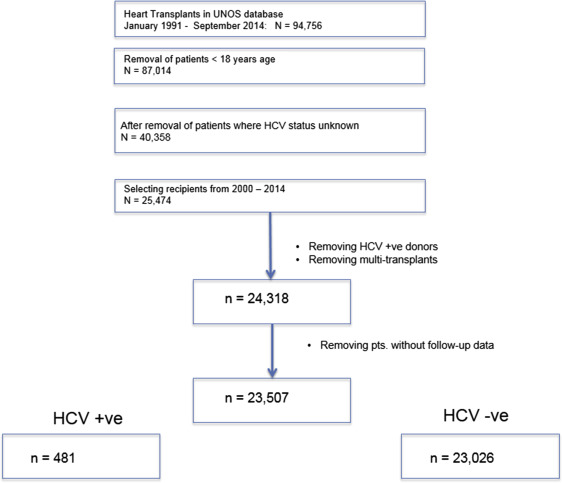
A retrospective analysis of the United Network for Organ Sharing (UNOS) database was conducted to identify adult (≥18 years) patients undergoing first-time heart transplant from January 1991 till September 2014. Recipients were excluded if (1) they did not have relevant information regarding HCV serostatus; (2) underwent multiorgan transplant; or (3) received HCV seropositive donors. The UNOS registry records recipient and donor HCV as seropositive or seronegative; information regarding HCV RNA status is not available. The UNOS Network is a self-reported registry obtaining information from every transplant center in the US at recipient listing, surgery, and at regular intervals thereafter. The UNOS database is updated every 3 months; the data implemented in our study were finalized in December 2014.
From the final study cohort, recipients were separated according to HCV serostatus at listing status into (1) HCV+ and (2) HCV− cohorts. The study protocol is outlined in Figure 1 .
Statistical analysis was conducted in the open-source software R.3.2.2. Continuous data are presented as mean (SD), whereas categorical data are presented as number (frequency). Recipient variables selected for analysis were: age, gender, race, blood group, mechanical circulatory support, height, weight and body mass index, status at listing, time on waiting list, diagnosis of heart failure, and year of transplant. Relevant serology viz. hepatitis B surface antigen, cytomegalovirus antibody status, and Epstein-Barr virus serostatus were also collected. Donor variables included were age, race, gender, blood group, blood group matching (identical or compatible), cocaine history, alcohol history, presence of diabetes, and need for cardiopulmonary resuscitation.
Preoperative variables and donor differences between HCV+ and HCV− cohorts were compared using the chi-square test or Fisher’s exact test for categorical variables; continuous data were compared with the Student t test or Wilcoxon rank-sum test. Adverse events were analyzed between cohorts and compared with multivariable logistic regression. Long-term survival rate was estimated using the Kaplan–Meier method; adjusted hazard ratio for mortality in HCV +ve was determined with the Cox proportional model. As presented in Table 1 , amount of missing data was minimal. Hence, rows with missing data were excluded while conducting multivariable regression analyses.
| Pre-operative Variable | HCV + cohort N = 481 | HCV- cohort N = 23,026 | p -value | Missing data (%) |
|---|---|---|---|---|
| Age | 52 ± 11 | 52 ± 12 | 0.64 | – |
| Weight | 81 ± 17 | 80 ± 17 | 0.42 | – |
| Male | 77% | 75% | 0.32 | – |
| Etiology | – | |||
| Ischemic Cardiomyopathy | 38% | 37% | 0.92 | |
| Idiopathic Dilated Cardiomyopathy | 35% | 35% | ||
| Diabetes mellitus | 2.2% | 2.7% | 0.59 | |
| Serum Bilirubin (total) (mg%) | 1.2 ± 1.6 | 1.1 ± 2.1 | 0.70 | |
| Serum Albumin (mg%) | 3.7 ± 0.7 | 3.6 ± 0.7 | 0.89 | 15% |
| Serum creatinine (at transplant) (mg%) | 1.2 ± 0.4 | 1.2 ± 0.5 | 0.63 | |
| Status at Listing | – | |||
| Status 1A | 24% | 21% | 0.39 | |
| Status 1B | 36% | 36% | ||
| Inotropic support | 33% | 36% | 0.24 | – |
| Intra-aortic balloon pump | 5.1% | 4.8% | 0.83 | – |
| ECMO support | 0.4% | 0.3% | 0.66 | – |
| Durable mechanical Support | < 0.001 | 11% | ||
| Left Ventricular Assist Device | 12% | 18% | ||
| Right Ventricular Assist Device | 0.06% | 0.6% | ||
| Bi-ventricular Assist Device | 1.6% | 1.4% | ||
| Total Artificial Heart | 0.3% | 0.6% | ||
| Total Wait-time | 99 (33,263) | 85 (25,243) | 0.92 | – |
| Status 1A time | 21 (9,43) | 19 (7,37) | 0.02 | – |
| Status 1B time | 49 (14,125) | 46(13,123) | 0.34 | – |
| Cytomegalovirus + sero-status | 68% | 59% | < 0.001 | 2% |
| Ebstein Barr virus + sero-status | 70% | 70% | 0.99 | 18% |
| Hepatitis B+ sero-status | 5% | 1% | <0.001 | 2% |
| Race | 0.01 | – | ||
| Caucasian | 65% | 71% | ||
| African American | 22% | 17% | ||
| Hispanic | 9% | 7% | ||
| Others | 2% | 1% | ||
| Human leucocyte antigen mismatch > 5 loci | 53% | 58% | 0.76 | 14% |
| Re-imbursement | 0.16 | – | ||
| Public payment | 43% | 40% | ||
| Private Insurance | 53% | 57% | ||
| Others | 3% | 2% | ||
| Donor Characteristics | ||||
| Age | 31 ± 11 | 31 ± 12 | 0.80 | – |
| Male | 72% | 71% | 0.82 | – |
| Cardiopulmonary Resuscitation performed | 5.8% | 5.2% | 0.67 | < 1% |
| Heavy alcohol use | 12% | 11% | 0.44 | 29% |
| Cocaine use | 14% | 14% | 0.99 | 2% |
| Extended Criteria Donor | 3.3% | 3.3% | 0.99 | – |
| Ischemic time | 3.2(2.5,3.9) | 3.1(2.4,3.8) | 0.35 | – |
Although the primary end point studied was long-term survival, we also compared 30-day mortality, renal failure, and incidence of rejection episodes between both cohorts. All results are presented at the 95% confidence level.
Results
During the 25-year period, 23,507 consecutive patients with nonmissing hepatitis C serology underwent isolated heart transplantation ( Figure 1 ). From this cohort, 481 (2.05%) were hepatitis C+ve (mean age 52.7 ± 11.2 years; men 77%). Ischemic and dilated cardiomyopathy was present in 38% and 35% respectively. Although 21% were listed as status 1A, 36% recipients were 1B at listing. The median wait time till transplant was 86 (26,244) days; recipients on status 1A spent 19 days, whereas 1B waited for a median period of 46 days before surgery.
Table 1 presents baseline characteristics of both cohorts. Mechanical circulatory support was implemented more in the HCV −ve cohort (p <0.001). Although 18% HCV +ve recipients were supported with a left ventricular assist device, 12% of patients with HCV −ve had a durable left ventricular assist device at listing. HCV +ve recipients waited longer as status 1A (median 21 vs 19 days) for a donor organ (p <0.001). HCV +ve recipients were more often Cytomegalovirus (68% vs 59%; p <0.0001) and hepatitis B (5% vs 1%; p <0.001) seropositive at listing. HCV +ve recipients were more often African-American (22% vs 17%; p <0.001); however, insurance sources were comparable between cohorts (p = 0.16).
The overall 30-day survival was 95.4%. Adjusted analyses demonstrated that HCV +ve cohort recipients (6% vs 4.4%) had higher postoperative mortality (odds ratio 1.54 [1.01 to 2.27]; p = 0.02). As reported in Table 2 , other adverse event rates were comparable in both cohorts. Postoperative stay was longer in the HCV +ve cohort (median 16 vs 14 days; p <0.01).



