CHAPTER 52 Coronary Angiography: Valve and Hemodynamic Assessment
After the first intravascular catheter was placed by Forsmann in the 1920s,1 techniques to achieve vascular access developed rapidly. Sones and coworkers performed the first selective diagnostic coronary catheterization in 1956.2,3 In 1977, Grüntzig performed the first coronary angioplasty.4,5 Angioplasty and the placement of intravascular stents has now become the predominant form of catheter-based intervention in all major vascular beds.
Coronary artery disease (CAD) is the leading cause of death in the United States,6 and this has remained relatively unchanged over the past several decades despite improvements in primary and secondary prevention and the management of acute coronary syndromes, including rapid reperfusion for ST-segment myocardial infarction.7 In addition to having high mortality, CAD is a significant cause of morbidity in the United States, with increasing rates of congestive heart failure (CHF) noted over the past decade.8
Access for right or left heart catheterizations can be performed via the brachial, radial, or femoral artery.9–13 Other potential but less commonly used sites include direct cutdown (brachial or femoral) and, in rare instances, direct left ventricular (LV) puncture.14
DIAGNOSTIC CATHETERIZATION TECHNIQUES
Indications
The most common reason to perform coronary angiography is to assess for the presence of clinically suspected CAD.15–19 Diagnostic angiography is also performed when a percutaneous intervention or surgical revascularization might be planned, or when valve replacement or repair or percutaneous procedures are planned. The risk-to-benefit ratio should be determined prior to angiography to identify patients who will benefit most from imaging.
Contraindications
The principal contraindications to coronary angiography include bleeding diathesis, renal failure (true or impending), fever, ongoing infection, and severe anemia (Box 52-1). Also, uncontrolled or uncorrected hypokalemia, hyperkalemia, digoxin toxicity, severe allergy to contrast dyes, and over-anticoagulation with an international normalized ratio (INR) of greater than 1.8 (although for radial access, this may not be a contraindication) are considered relative contraindications to catheterization.
Complications
Complications of coronary angiography primarily involve vascular access site complications. Catheter manipulation in atherosclerotic vessels may lead to emboli (thrombus, atherosclerotic debris, calcium, or air) or clot formation, potentially leading to stroke, myocardial infarction (MI), worsening renal function, or CHF (Table 52-1). Pseudoaneurysm or other vascular access complications may be as high as 3%20 in patients with severe peripheral arterial occlusive disease.
Table 52–1 Possible Complications of Peripheral Catheterization
| Complication | Rate (%) |
|---|---|
| Vascular access dissection or perforation | 0.1-0.2 |
| Bleeding/hematoma | 1.5-2.0 |
| Allergic reaction | 0.5-2.0 |
| Vasovagal events | 1.0-2.0 |
| Death | 0.1-0.2 |
ANGIOGRAPHY
The most common access for coronary angiography is the femoral artery, although brachial, radial, and axillary approaches are also used. The most common method of cannulation of the vessel is the Seldinger technique.21 In this method, the vessel is punctured and a guide wire, usually J-tipped, is advanced into the vessel.22,23 This wire then serves as a rail over which the dilator and sheath enter the vessel.
Left Coronary Angiography
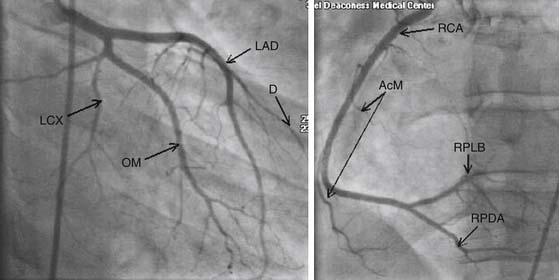
In cannulating the left main coronary ostium, a complete and safe study should be ensured by taking care to confirm that the pressure tracing is not dampened or ventricularized. Normally, a preshaped catheter such as a Judkins left 4 catheter (JL4) is used as a default catheter for left coronary angiography. It is successful in engaging the left main ostium approximately 80% of the time. If the aortic root is dilated or narrow, this can usually be accommodated with longer or shorter catheters (JL6 or JL3.5). In addition, if the anatomy is altered, with a left main origin that is posterior, an Amplatz catheter may be used to cannulate the ostium of the left main. The coronary anatomy is defined with contrast injections of 8 to 10 mL during cine runs. The angles taken during angiography allow three-dimensional reconstruction of the anatomy using orthogonal views to see the arteries in multiple planes. The left system begins with the left main, which then terminally bifurcates into the left anterior descending (LAD) and left circumflex (LCX) coronary arteries. In approximately one third of patients, the left main terminally trifurcates into the LAD, the LCX, and an intermediate branch (ramus intermedius) supplying much of the left ventricular free wall.24 The LAD gives off septal arteries as it courses down the interventricular groove, as well as various diagonal arteries supplying the anterolateral free wall of the left ventricle. The LCX gives off marginal arteries as it courses in the atrioventricular (AV) groove. The marginal arteries supply the lateral free wall of the left ventricle (Fig. 52-1).
Right Coronary Angiography
The right coronary artery (RCA) is usually engaged with a Judkins right 4-cm catheter (JR4). The RCA courses in the interventricular groove and gives off acute marginal and right ventricular branches that supply the right ventricular (RV) free wall. The RCA terminally bifurcates at the crux to form the right posterior descending and right posterolateral coronary arteries, which supply the inferior and inferolateral segments of the left ventricle, respectively. The right posterior descending artery courses in the posterior interventricular septum, supplying the septum as well (see Fig. 52-1).
The dominance of the coronary circulation depends on which artery supplies the posterior circulation—namely, the posterior descending artery or the posterolateral artery.25 About two thirds of the population is right-dominant (the RCA provides both of these branches), 25% is codominant (the RCA supplies the posterior descending artery and the LCX gives off the posterolateral artery), and 15% is left-dominant (the LCX provides both of these branches).25
Coronary Anomalies
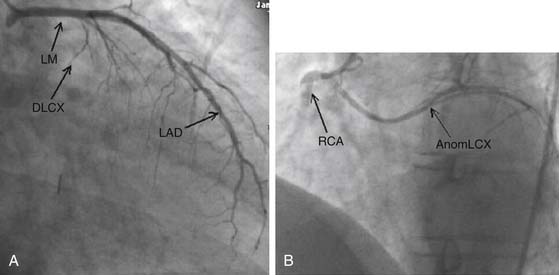
Several coronary anomalies exist. Most are anatomic variants, such as dual ostia for the LAD and LCX. Others may be congenital, such as those involving the origin of the LCX from the RCA (Fig. 52-2).26–32 Most congenital anomalies, such as these, have little impact on coronary circulation. However, in the case of the LAD originating from the RCA or right coronary cusp and coursing posteriorly, there is an associated increased mortality, usually secondary to arrhythmias and ischemia.29,31
Angiographic Projections
All major coronary arteries lie in one of two planes: the interventricular septum or the AV groove (Fig. 52-3). The image projections are designed to display the intended anatomy in profile. For example, the right posterior descending artery coursing along the interventricular septum and the inferior wall is best seen with the interventricular septum in its longest profile, the flat right anterior oblique (RAO) projection. On the other hand, the LCX, which courses along the AV groove, is best visualized in the anteroposterior (AP) or RAO caudal projection, looking at the AV groove in profile.
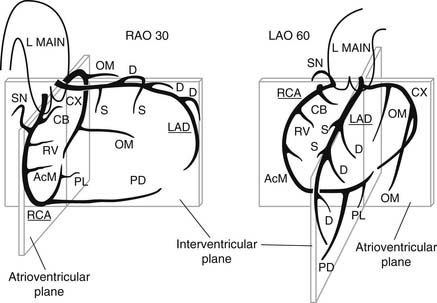
Figure 52–3 Representation of coronary anatomy in relationship to the interventricular and atrioventricular valve planes as seen in two views: right anterior oblique (RAO) and left anterior oblique (LAO). Coronary branches are as follows: AcM, acute marginal; CB, conus branch; CX, circumflex; D, diagonal; L Main, left main; LAD, left anterior descending; OM, obtuse marginal; PD, posterior descending; PL, posterolateral left ventricular; RCA, right coronary; RV, right ventricular; S, septal; SN, sinus node www.lww.com.
(Used with permission from Coronary angiography. In: Baim DS, Grossman W, editors. Grossman’s cardiac catheterization, angiography, and intervention. 6th edition. Philadelphia: Lippincott Williams and Wilkins; 2000.)
Graft Angiography
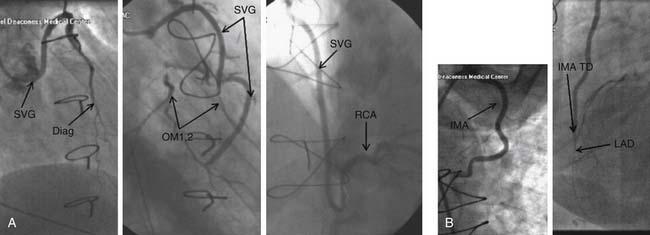
Commonly, saphenous vein grafts to the right and left coronary circulations arise from the anterior surface of the aorta several centimeters from the sinus of Valsalva. RCA grafts generally arise from the right anterior aorta and the left system grafts commonly arise from the left anterior side of the aorta, with the LAD grafts usually being lower than the LCX grafts. In many cases, the surgeon may place a ring at the origin of a graft that can greatly reduce the chance of missing a graft because one cannot cannulate or find it. The best views for the LAD/diagonal grafts are flat LAO and RAO projections to visualize the graft in its greatest profile. The distal (native) vessel is then imaged with some cranial or caudal projection to define all its segments after the distal anastomosis according to the vessel of interest (i.e., cranial for the LAD, and caudal for the LCX). These images are usually easier to obtain and evaluate because there is less overlap of other coronary anatomy to deal with. However, the ability to view well the origin or distal anastomosis may be challenging for some grafts. The RCA grafts are usually best seen with flat LAO and RAO projections. Again, after the graft has been imaged, the native vessel is imaged with some cranial or caudal angulation to fully define the anatomy after the distal anastomosis (Fig. 52-4).
Since the mid 1990s, the internal mammary artery (IMA) has increasingly been the conduit of choice for the LAD and in some cases for the RCA because of the high patency of these conduits.33 Generally, the IMA is cannulated after the subclavian artery is engaged with the preformed catheter and J wire. The catheter is advanced and cleared. Then, the catheter is withdrawn and a gentle counterclockwise torque is applied until the catheter engages the origin of the left IMA (LIMA). Once the vessel is engaged, the catheter is given a gentle clockwise torque to remove any excess tension on the catheter.34 The views for angiography for the LIMA are generally AP or slight RAO/cranial (0-20/40) for the proximal and mid segments of the graft, and steep flat LAO to lateral projection for the anastomosis of the LIMA with the LAD. The right IMA conduit is similarly engaged from the right subclavian artery. The views for the mid segment, origin, and anastomosis are, generally, flat LAO with some cranial and steep AP cranial, respectively (see Fig. 52-4).
HEMODYNAMICS
Principles
The hemodynamic assessment performed during coronary angiography is as integral a part of the procedure as is the imaging of the coronary vasculature. At any given moment, the hemodynamics reflect a culmination and interaction between various ongoing dynamic processes determining cardiac output, coronary artery disease, left ventricular function, systemic metabolic needs, and systemic and pulmonary pressures.35,36 Hemodynamic measurements (vessel or ventricular pressures), measurement of cardiac output, and the evaluation of shunts are an integral part of diagnostic coronary evaluation. All pressures should be measured with a transducer that will allow direct real-time measurements. An important element for this process is the establishment of a zero reference. The reference is usually accepted as the mid chest level in the anterior-posterior direction.
Right Heart Catheterization
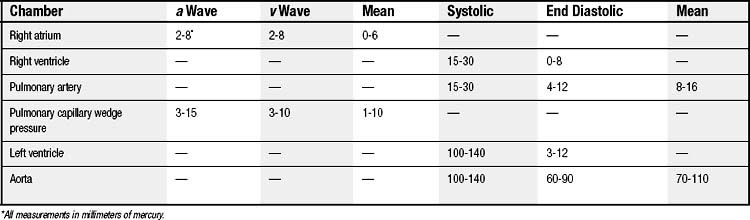
Pressure tracings are obtained at each level of the advancement of the RH catheter. Normal values for the RH and systemic arterial (left ventricular) pressures are shown in Table 52-2 and Figure 52-5. Since the introduction of the balloon-tipped RH catheter,37 RHC has become very common in the catheterization laboratory.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree