CHAPTER 24 Congenital Chest Wall Deformities
PECTUS EXCAVATUM
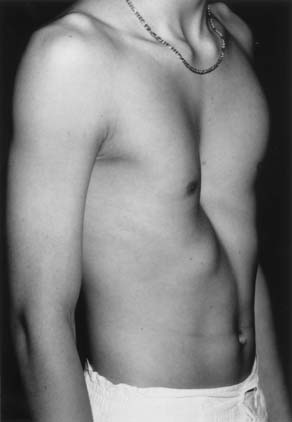
Pectus excavatum is the most frequent anterior chest wall deformity. The central depression of the chest is produced by posterior angulation of the sternum and the costal cartilages. The first and second costal cartilages and the manubrium are usually in a normal position (Fig. 24-1), but the lower costal cartilages, which insert into the sternum, and the body of the sternum are depressed. The most anterior segment of the ossified portion of the ribs may also be curved posteriorly in older adolescents and adults. The extent of sternal and cartilaginous deformity is quite variable. Numerous methods of grading these deformities have been proposed by Hümmer and Willital,1 von der Oelsnitz,2 Welch,3 Haller and associates,4 and others, but none has been universally accepted. Asymmetry of the depression is often present. The right side is frequently more depressed than the left, and the sternum may be rotated as well. A system to quantify the asymmetry based on computed tomography (CT) of the chest has been reported.5 The majority of affected children (86%) with pectus excavatum are identified at birth or within the first year of life (Fig. 24-2). They have a characteristic physique—a broad thin chest, dorsal lordosis, “hook shoulder” deformity, costal flaring, and poor posture. The deformity rarely resolves with increasing age, and it may worsen during the period of rapid growth in adolescence. Waters and associates identified scoliosis in 26% of 508 patients with pectus excavatum,6 so all patients with pectus deformities should be evaluated clinically for scoliosis. Asymmetric pectus excavatum with a deep right gutter and sternal rotation is often accompanied by scoliosis. Congenital heart disease was identified in 1.5% of infants and children undergoing chest wall correction at the Children’s Hospital in Boston (Table 24-1).7 The frequency of chest wall deformities among all patients with congenital heart disease evaluated at this institution was only 0.17%.
Table 24–1 Congenital Heart Disease Associated with Pectus Excavatum and Carinatum
| Congenital Heart Disease | Number of Cases |
|---|---|
| Aortic ring | 1 |
| Aortic regurgitation | 1 |
| Atrial septal defect primum | 2 |
| Atrial septal defect secundum | 3 |
| Complete atrioventricular canal | 3 |
| Dextrocardia | 3 |
| Ebstein’s malformation | 1 |
| Idiopathic hypertrophic subaortic stenosis | 2 |
| Patent ductus arteriosus | 1 |
| Pulmonic stenosis | 1 |
| Total anomalous pulmonary venous return | 1 |
| Transposition of great arteries | 6 |
| Tetralogy of Fallot | 3 |
| Tricuspid atresia | 1 |
| Truncus arteriosus | 1 |
| Ventricular septal defect | 6 |
From Shamberger RC, Welch KJ, Castaneda AR, Keane JF, Fyler DC. Anterior chest wall deformities and congenital heart disease. J Thorac Cardiovasc Surg 1988;96:427-32, with permission.
Asthma may be identified in association with pectus excavatum and carinatum. However, in a review of 694 consecutive cases, 35 patients with asthma were identified (5.2%), a frequency comparable to that of asthma in the general pediatric population.8
Etiology and Incidence
Ravitch reported that pectus excavatum may occur as frequently as 1 in 300 to 400 live births and that it is rare in blacks.9 It occurs more frequently in boys than girls, by an almost 4:1 ratio. Although the sternal depression appears to be caused by overgrowth of costal cartilages, the etiology of pectus deformities is unknown. Lester attributed its development to an abnormality of the diaphragm that tethered the sternum posteriorly.10 This theory was supported by the occurrence of pectus excavatum in children after repair of agenesis of the diaphragm and the frequent association of pectus excavatum and congenital diaphragmatic hernia.11,12 Histopathologic changes in the costal cartilages similar to those seen in scoliosis, aseptic osteonecrosis, and inflammatory processes are reported, but the etiology of these findings and their significance are unknown.13
A family history of chest wall deformity in 37% of 704 patients suggests a genetic predisposition to pectus excavatum.14 Three of four siblings were affected in one family. Analysis of 34 families with more than one family member with pectus excavatum has shown a variable pattern of inheritance that appears to be multifactorial.15
A high incidence of chest wall deformities occurs in children with Marfan syndrome, and these deformities are often severe and usually accompanied by scoliosis.16 Pectus excavatum is also commonly seen in individuals with the abdominal musculature deficiency syndrome (prune-belly syndrome).17 Pectus excavatum also occurs in association with other myopathies and chromosomal defects such as Turner’s syndrome. A summary of the associated musculoskeletal abnormalities is shown in Table 24-2.
Table 24–2 Musculoskeletal Abnormalities Identified in 130 of 704 Cases of Pectus Excavatum
| Musculoskeletal Abnormality | Number of Cases |
|---|---|
| Scoliosis | 107 |
| Kyphosis | 4 |
| Myopathy | 3 |
| Marfan syndrome | 2 |
| Pierre Robin syndrome | 2 |
| Prune-belly syndrome | 2 |
| Neurofibromatosis | 3 |
| Cerebral palsy | 4 |
| Tuberous sclerosis | 1 |
| Congenital diaphragmatic hernia | 2 |
From Shamberger RC, Welch KJ. J Pediatr Surg 1988;23:615, with permission.
Pathophysiology
The cardiopulmonary implications of the pectus excavatum deformity have been debated for many decades. Although some authors feel this deformity has a limited physiologic effect, many patients report increased stamina after surgical repair. These findings date back to the first surgical repair performed by Sauerbruch in 1913.18 The patient was an 18-year-old man who developed dyspnea and palpitations with very limited exercise. Three years after his operation, he could work 12 to 14 hours a day without tiring and without palpitations. Anecdotal reports during the next 3 decades repeated this observation. Investigators have sought to identify the physiologic abnormality or combination of abnormalities that could explain this symptomatic improvement after surgery. Early physiologic measurements of cardiac and pulmonary function were crude and did not yield convincing evidence of a cardiopulmonary deficit. In many early studies, the results fell within the broad range of normal values, if often at the lower limit.7 A recent analysis of a large multi-institutional cohort of 408 patients with pectus excavatum found that the median values for forced vital capacity (FVC) and forced expiratory volume in 1 second (FEV1) were 13% below predicted values, and the forced expiratory flow, midexpiratory phase (FEF2.5%-75%) median was 20% below the predicted value.19
Electrocardiographic abnormalities are common and result from the abnormal configuration of the chest wall, which produces displacement of the heart into the left thoracic cavity.20 Patients with a history of palpitations should have a 24-hour electrocardiogram to document the presence or absence of arrhythmias, as well as an echocardiogram to evaluate for mitral valve prolapse. Resolution of these supraventricular arrhythmias has been anecdotally reported after correction of a pectus excavatum deformity.
Pulmonary Function Studies
As early as 1951, Brown and Cook performed pulmonary evaluations on patients before and after surgical repair.21 They demonstrated that although vital capacity (VC) was normal, the maximum breathing capacity was diminished (50% or more) in 9 of 11 cases, and it increased an average of 31% after surgical repair. Weg and associates in 1967 evaluated 25 Air Force recruits with pectus excavatum and compared them with 50 unselected basic trainees.22 Although the lung compartments of both groups were equal, as were the vital capacities, the maximal voluntary ventilation was significantly lower in those with pectus excavatum than in the control population. Castile and coworkers in 1982 evaluated seven patients with pectus excavatum, five of whom were symptomatic with exercise.23 The mean total lung capacity of the group was 79% of predicted. Flow volume configurations were normal, excluding airway obstruction as a cause of the symptoms. Workload tests demonstrated normal response to exercise in the dead-space-to-tidal-volume ratio and in alveolar-to-arterial oxygen difference. The measured oxygen uptake, however, increasingly exceeded predicted values as workload approached maximum in the four “symptomatic” patients with pectus excavatum. This pattern of oxygen consumption was different from that in normal subjects and in the three asymptomatic patients with pectus excavatum, in whom a linear response was seen. The mean oxygen uptake in the symptomatic patients at maximal effort exceeded the predicted values by 25.4%. The three asymptomatic patients, on the other hand, demonstrated normal linear oxygen uptake during exercise. Increased oxygen uptake suggests increased work of breathing in these symptomatic individuals despite the normal or mildly reduced VCs. Increases in tidal volume with exercise were uniformly depressed in those with pectus excavatum.
Cahill and coworkers in 1984 performed preoperative and postoperative studies in five children and adolescents with pectus carinatum and in 14 with pectus excavatum.24 No abnormalities were demonstrated in the pectus carinatum group. The low normal vital capacities in excavatum patients were unchanged by operation, but a small improvement in the total lung capacity and a significant improvement in the maximal voluntary ventilation were seen. Exercise tolerance improved in those with pectus excavatum after operation, as determined both by total exercise time and maximal oxygen consumption. In addition, at any given workload, those with pectus excavatum demonstrated a lower heart rate, stable oxygen consumption, and higher minute ventilation after repair. Mead and associates in 1985 studied rib cage mobility by assessing intra-abdominal pressure.25 Normal abdominal pressure tracings in pectus excavatum suggested normal rib cage mobility.
Blickman and colleagues in 1985 assessed pulmonary function in 17 children with pectus excavatum by xenon perfusion and ventilation scintigraphy before and after surgery.26 Ventilation studies were abnormal in 12 children before surgery and improved in seven after repair. Perfusion scans were abnormal in 10 children before surgery and improved after operation in six children. The ventilation-perfusion ratios were abnormal in 10 of the 17 children preoperatively and normalized after repair in six children.
Derveaux and associates in 1989 evaluated 88 patients with pectus excavatum and carinatum by pulmonary function tests before and 1 to 20 years after repair (mean, 8 years).27 The surgical technique used a fairly extensive chest wall dissection. Preoperative studies were within the normal range (>80% of predicted) except in patients who had both scoliosis and pectus excavatum. The postoperative values for FEV1 and VC expressed as percent of expected were decreased in all groups, although the absolute values at follow-up may have been greater than at preoperative evaluation. Improved chest wall configuration was confirmed by radiologic evaluation, so the relative deterioration in pulmonary function was not the result of recurrence of the pectus deformity. An inverse relationship was found between preoperative and postoperative function. Those with less than 75% of predicted function had improved function after surgery, but function was worse after repair if the preoperative values were greater than 75% of predicted. Almost identical results were found in a study by Morshuis and coworkers in 1994.28 They evaluated 152 patients before and a mean of 8 years after surgery for pectus excavatum. These results of pulmonary evaluation were in contrast to the subjective symptomatic improvement reported by the patients and their improved chest wall configuration. The decline in pulmonary function in the postoperative studies was attributed to the operation, because the preoperative pulmonary defect appeared to be stable regardless of the age at initial repair. Both studies were marred by the obvious lack of an age- and severity-matched control group without surgery.
Derveaux and colleagues in 1988 evaluated transpulmonary and transdiaphragmatic pressures at total lung capacity in 17 individuals with pectus excavatum.29 Preoperative and long-term follow-up evaluations were performed a mean of 12 years apart. Reduced transpulmonary and transdiaphragmatic pressures demonstrated that the increased restrictive defect was produced by extrapulmonary rather than pulmonary factors, suggesting that surgery produced increased rigidity of the chest wall.
Wynn and others in 1990 assessed 12 children with pectus excavatum by pulmonary function tests and exercise testing.30 Eight children had repair and were evaluated preoperatively and postoperatively. Four children had two sets of evaluations but no operation. A decline in total lung capacity was identified in the repaired children compared with stable values in the control group. Cardiac output and stroke volume increased appropriately with exercise before and after operation in both groups, and the operation was believed to have produced no physiologically significant effect on the response to exercise.
Kaguraoka and associates in 1992 evaluated pulmonary function in 138 individuals before and after repair of pectus excavatum.31 A decrease in VC occurred during the first 2 months after surgery, with recovery to preoperative levels by 1 year after operation. At 42 months, the values were maintained at baseline, despite a significant improvement in the chest wall configuration. Tanaka and coworkers in 1993 found similar results in individuals who had the more extensive sternal turnover technique; in fact, they demonstrated a more significant and long-term decrease in VC.32 Morshuis and coworkers in 1994 evaluated 35 patients who had had pectus excavatum repaired as teenagers or young adults; ages were 17.9 ± 5.6 years.33 Preoperative evaluations were performed and repeated 1 year after surgery. Preoperative total lung capacity (86.0% ± 14.4% of predicted) and VC (79.7% ± 16.2%) were significantly decreased from predicted values and decreased further after surgery (−9.2% ± 9.2% and −6.6% ± 10.7%, respectively). The efficiency of breathing at maximal exercise improved significantly after operation. Exercise was limited by ventilation in 43% of the patients before repair. A tendency toward improvement occurred after operation. However, the group with no ventilatory limitation initially demonstrated one after operation, with a significant increase in oxygen consumption.
Quigley and colleagues in 1996 evaluated 36 adolescents with pectus excavatum and 10 age-matched healthy controls at baseline and then an average of 8 months after surgery for 15 patients and 9 months for controls.34 Adolescents with pectus excavatum had a decrease in VC compared with controls, although the mean values remained in the normal range. The mean total lung capacity was also normal. There was no difference in workload performance between patients with pectus excavatum and the controls, with both groups achieving a similar duration and level of exercise. No significant change in pulmonary function tests was seen at follow-up in either group. The duration of exercise and the level of work increased significantly in those who had surgery but not in the controls. The absence of adverse effects on pulmonary function after surgery was attributed to a less extensive surgical procedure than was used in the studies reported by Derveaux29 and Morshuis28,33 and their colleagues. Two series reported the effects on pulmonary function of the minimally invasive repair of pectus excavatum (MIRPE) described by Nuss.35,36 In these series, there were limited or no preoperative abnormalities and mild or no significant change after repair. Similar results were reported in a prospective study of 145 patients having a MIRPE.37 There was no improvement in static values 6 months after removal of the struts.
In composite, these studies of pulmonary function over the last 4 decades failed to document consistent improvement in pulmonary function resulting from surgical repair. In fact, some studies have demonstrated deterioration in pulmonary function at long-term evaluation, which was attributed to increased chest wall rigidity after repair. A meta-analysis of 12 of these studies revealed no statistically significant change in pulmonary function.38 Despite this finding, workload studies have shown improvement in exercise tolerance after repair, suggesting a cardiac basis for this enhanced performance.24,39
Cardiovascular Studies
Posterior displacement of the sternum can produce a deformity of the heart, particularly indentation of the right ventricle. Displacement of the heart to the left, often with a sternal “imprint” on the anterior wall of the right ventricle, was demonstrated angiographically by Garusi and D’Éttorre in 1964,40 and Howard41 demonstrated its resolution after surgical repair. Elevated right heart pressures have been reported by some authors, as have pressure curves similar to those seen in constrictive pericarditis.
In 1962, Bevegård studied 16 individuals with pectus excavatum by right heart catheterization and exercise testing.42 The physical work capacity in pectus excavatum at a given heart rate was significantly lower in the sitting than in the supine position. Those with 20% or greater decline in physical work capacity from the supine to the sitting position had shorter sternovertebral distances than those with less decrease in their physical work capacity. The measured stroke volume at rest decreased from supine to sitting positions by a mean of 40.3%, similar to that in normal subjects. In the supine position, stroke volume increased with exercise by 13.2%. In the sitting position, the increase in stroke volume from rest to exercise was 18.5% for the pectus excavatum group, significantly lower (P < .001) than the 51% increase seen in normal subjects. Thus, in the pectus excavatum group, an increased cardiac output could be achieved primarily by increased heart rate because only limited enhancement of the stroke volume could occur. Intracardiac pressures measured at rest and with exercise were normal in all patients despite this apparent limitation of ventricular volume.
Gattiker and Bühlmann in 1967 confirmed this limitation of the stroke volume in a study of 19 patients.43 In the upright position at a heart rate of 170 beats per minute, the physical work capacity was lower than in the supine position (mean 18% decrease) because of the decrease in stroke volume. Beiser and associates in 1972 performed cardiac catheterization in six adolescents and young adults with moderate degrees of pectus excavatum.44 Normal pressure and cardiac index were obtained at rest in the supine position. The cardiac index during moderate exercise was normal, but the response to upright exercise was below that predicted in two patients and at the lower limit of normal in three patients. The cardiac index was 6.8 ± 0.8 L/min/m2 compared with 8.9 ± 0.3 L/min/m2 in a group of 16 normal controls (P < .01). The difference in cardiac performance again appeared to be produced primarily by a smaller stroke volume in the group with pectus excavatum in an upright position. Stroke volume was 31% lower and cardiac output 28% lower during upright as compared with supine exercise. Postoperative studies were performed in three individuals, and two of them achieved a higher level of exercise tolerance after surgery. The cardiac index increased an average of 38%. Because heart rate at maximal exercise was not higher after repair, an enhanced stroke-volume response was responsible for this increase.
Peterson and associates in 1985 performed radionuclide angiography and exercise studies in 13 children with pectus excavatum.45 Ten of 13 were able to reach the target heart rate before surgical repair, four without symptoms. After operation, all but one child reached the target heart rate during the exercise protocol, and nine of 13 reached the target without becoming symptomatic. The left and right ventricular end-diastolic volumes were consistently increased after repair at rest, and the mean stroke volume was increased 19% after repair. These findings substantiated the ventricular volume changes previously demonstrated by cardiac catheterization, although an increase in the cardiac index was not demonstrated. Recent echocardiographic studies by Kowalewski and associates of 42 patients before and 6 months after surgery revealed statistically significant changes in the right ventricular volume indices after surgery.46 There was no correlation seen, however, between the pectus index and the changes in the right ventricular volume indices.
Malek and coauthors in 2003 performed maximal exercise testing and pulmonary function tests on 21 individuals with pectus excavatum, 18 of whom routinely did aerobic exercise.39 Their maximal oxygen uptake and oxygen pulse were both significantly lower than expected, and these limitations were attributed to cardiovascular and not ventilatory factors, and they resulted in an abnormally low threshold for lactate accumulation. Of note, patients with a Haller index of greater than 4.0 were eight times more likely to have reduced aerobic capacity than those with a lower severity index.
Results of cardiac function have more recently been assessed in patients having the MIRPE procedure.36 In 11 patients, an increase was demonstrated in the stroke volume measured echocardiographically 3 months after repair, to which subjective improvement in exercise tolerance was attributed.
A meta-analysis was performed of eight studies (representing 169 patients) that reported quantitative data on the cardiovascular function of patients before and after repair.47 The analysis suggested that surgical repair did not significantly improve cardiovascular function.
Echocardiographic Studies of Mitral Valve Prolapse
Bon Tempo, Salomon, and Schutte and their associates reported mitral valve prolapse in patients with narrow anterior-posterior chest diameters, anterior chest wall deformities, and scoliosis.48–50 Prospective echocardiographic studies of adults with pectus excavatum demonstrated mitral valve prolapse in six of 33 patients (18%) studied by Udoshi and associates51 and in 11 of 17 patients (65%) of Saint-Mezard and colleagues.52 Anterior compression of the heart by the depressed sternum may deform the mitral annulus or the ventricular chamber and produce mitral valve prolapse in these patients. We performed a preoperative echocardiographic evaluation of children and adolescents with pectus excavatum and identified 23 with mitral valve prolapse.53 Postoperative studies did not demonstrate mitral valve prolapse in 10 (43%) of these children, suggesting its resolution after correction of the chest wall deformity.
Coln and coauthors performed echocardiography on patients during exercise (123 preoperatively and 107 postoperatively).54 They demonstrated chamber compression in 117 (95%) before surgery and in none of the patients after repair. They also found in patients with chamber compression resolution of mitral valve prolapse (16 of 23) and mitral valve regurgitation (28 of 29) after repair, as I had previously demonstrated. Symptoms related to exertion, reported in 106 (86%) of the patients, resolved remarkably in all, which was attributed to the cardiac effects of repair.
Surgical Repair
The first surgical corrections of pectus excavatum were reported by Meyer55 in 1911 and Sauerbruch in 1920,18 and Ochsner and DeBakey56 summarized the early experience with various techniques. In 1949, Ravitch reported a technique that included excision of all deformed costal cartilages with the perichondrium, division of the xiphoid from the sternum, division of the intercostal bundles from the sternum, and a transverse sternal osteotomy securing the sternum anteriorly in an overcorrected position.57 Kirschner wire fixation was used in the first two patients, and silk suture fixation in later patients.
Baronofsky (1957) and Welch (1958) subsequently reported a technique for the correction of pectus excavatum that emphasized total preservation of the perichondrial sheaths as well as attachment of the upper sheaths and intercostal bundles to the sternum.58,59 Anterior fixation of the sternum was achieved with silk sutures. Haller and associates later developed a technique that they labeled as tripod fixation.60 Subperichondrial resection of the abnormal cartilages is performed, followed by a posterior sternal osteotomy. The most cephalad normal cartilages are then divided obliquely in a posterolateral direction. When the sternum is elevated, the sternal ends of the cartilage rest on the costal ends, providing further anterior support of the sternum. Support of the sternum by metallic struts after mobilization of the costal cartilages has been promoted by several authors. Rehbein and Wernicke developed struts that could be placed into the marrow cavity of the ribs at the costochondral junction.61 An arch was then formed by the struts anterior to the sternum, and the sternum was secured to this arch. Paltia and associates placed a transverse strut through the caudal end of the sternum, firmly fixing its location.62 The two ends of the strut are supported by the ribs laterally. Adkins and Blades,63 and Jensen and associates63a used retrosternal elevation by a metallic strut. Willital employed a similar retrosternal strut after creating multiple chondrotomies in the costal cartilages to provide flexibility.64 Recent innovations in these methods include bioabsorbable struts, or use of Marlex mesh or a Dacron vascular graft as a strut, but there is no evidence that these methods are preferable to traditional methods with metallic struts.65 Robicsek and Fokin described a large series of chest wall deformities.66 For patients with pectus excavatum, the sternum is mobilized more extensively with this technique than with others, except for the sternal turnover. The sternum is divided from the intercostal muscles and perichondrial sheaths from its tip to the upper extent of the deformity. It is then supported in an anterior position by a hammock of Marlex mesh, which is sewn to the ends of the ribs on each side. The advantages of this extensive mobilization and permanent implantation of the mesh are not clear. No randomized studies have compared the recurrence or complication rates between suture or strut fixation techniques. Von der Oelsnitz2 and Hecker and coworkers,13 using suture fixation, reported satisfactory repairs in 90% to 95% in their large series.
The sternal turnover was first proposed by Judet and Judet67 in 1954 and Jung68 in 1956 in the French literature. The sternum is mobilized and the costal cartilages are divided, allowing the sternum to be rotated 180 degrees. Wada and colleagues69 in 1970 reported a very large series from Japan using this technique, which is essentially a free graft of sternum. It is a radical approach and has been associated with major complications if infection occurs. Modifications of this technique by Taguchi and associates in 1975 have involved either preservation of the internal mammary vessels by wide dissection or reimplantation of the internal mammary artery.70 These modifications were developed because of the reported incidence of osteonecrosis and fistula formation, which occurred in up to 46% of patients older than 15 years.
Allen and Douglas implanted Silastic molds into the subcutaneous space to fill the depression in pectus excavatum.71 Although this approach may improve the external contour of the chest, extrusion of the molds has occurred, and this method does nothing to increase the volume of the thoracic cavity or relieve compression on the heart. Recently, other authors have reported favorable cosmetic results in adults, with no pulmonary restrictions but with a significant frequency of seroma and hematoma formation.72,73 Schier and coworkers73 described the use of a suction device placed over the chest in children and adults with pectus excavatum, and early results were encouraging, but the durability of the correction was not established. Haecker and Mayr74 described a similar device. Although improvement was seen, only 14.7% of the patients had the sternum lifted to a “normal” position after 12 months of therapy.
Minimally Invasive Repair of Pectus Excavatum
A method of elevation of the sternum with a retrosternal bar without resection or division of the costal cartilages was first reported by Nuss and associates in 1998.75 He repaired 42 patients younger than 15 years (median age, 5 years) by placing a convex steel bar under the sternum and anterior to the heart through small bilateral thoracic incisions. A long clamp was passed blindly behind the sternum and out an opening in the contralateral chest (Fig. 24-3A). A tape was then drawn across the chest in the clamp and used to pull the bar through the pleural cavity and anterior to the pericardium. The bar was initially placed with the concave side anteriorly and then it was rotated once it was in position (see Fig. 24-3B). The bar was left in position for 2 years before removal, when presumed permanent remodeling of the cartilages had occurred. Although in the initial report Nuss warned that the “upper limits of age for this procedure require further evaluation,” the technique has been widely used in older patients, and Hebra and colleagues reported the use of this technique in adults.76
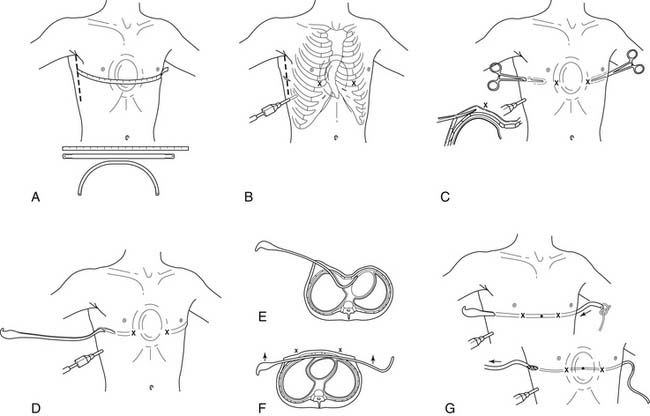
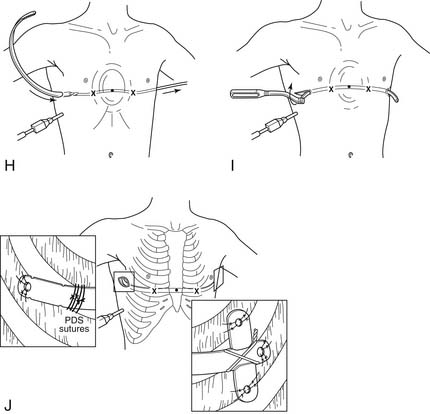
Figure 24–3 A, The length of the pectus bar is determined by measuring the distance from the right midaxillary line to the left midaxillary line and subtracting 2 cm or 1 inch, because the bar takes a shorter course than the tape measure. The measurement is done over the area of the deepest depression that is still part of the sternum. The bar is bent to the desired convex configuration, making note that the center of the bar should be flat for 2 to 4 cm to allow greater stability. B, A thoracoscope is inserted into the right chest two intercostal spaces below the planned bar placement to check that the internal anatomy corresponds with the external markings and to look for unexpected pathology. If all is well, the lateral thoracic incisions are made in the region of the midaxillary line, and subcutaneous tunnels are created to the greatest apex of the pectus deformity (X). The X’s represent the entrance and exit sites of the bar from the chest. They are in the intercostal space that is in the same horizontal place as the deepest depression and care should be taken that they are placed medial to the greatest apex of the chest. C, Skin tunnels are created above the muscle starting from each of the lateral thoracic incisions and going to the top of the pectus ridge on each side. The tunnels should be created so that the entry and exit sites of the bar from inside the chest are medial to the top of the pectus ridge on each side. With the thoracoscope in place, a tonsil clamp is inserted into the subcutaneous tunnel on the right, and a blunt thoracostomy is created at the X, taking care not to injure the intercostal vessels, lung, or pericardium. D, Under continued thoracoscopic visualization, a Lorenz (W. Lorenz Surgical, Jacksonville, FL) introducer is inserted into the chest through the right tunnel and thoracostomy site at the top of the pectus ridge. With great care and thoracoscopic guidance, the pleura and pericardium are dissected off the undersurface of the sternum, creating a substernal tunnel. The introducer is slowly advanced across the mediastinum and brought out through the corresponding intercostal space on the left and advanced out of the incision on the contralateral side. Again, this exit site is medial to the top of the pectus ridge. E, A 30-degree scope facilitates visualization during the substernal dissection, and care is taken to keep the point of the dissector underneath the sternum at all times to push the heart out of the way of the dissection plane. During the dissection, the ECG monitor should be turned to maximum volume to listen for any ectopy or arrhythmias. F, The introducer is pushed out of the thorax through the previously marked intercostal space (X) on the left and advanced out through the corresponding tunnel and incision. When the introducer is fully in place, the sternum is elevated by lifting the introducer on each side, thus correcting the pectus excavatum. The sternum is lifted out of its depressed position with the introducer, numerous times. This is facilitated by pressing down on the lower chest wall while lifting the introducer. G, Once the sternal depression has been corrected, umbilical tape is attached to the introducer, and the introducer is slowly withdrawn from the chest cavity with the umbilical tape attached. H, The pectus bar that was previously bent into a convex shape is then attached to the umbilical tape and slowly guided through the right subcutaneous tunnel under thoracoscopic visualization, and then through the substernal tunnel, with its convexity facing posteriorly until it emerges on the contralateral side. I, The pectus bar is positioned inside the chest with its convexity facing posteriorly and an equal amount of bar protruding on each side. Using the specially designed Lorenz bar flippers, the bar is rotated 180 degrees, giving instant correction to the pectus deformity. The sides of the bar should be resting comfortably against the musculature and should not be too tight or too loose. If the bar does not fit snugly on each side because of pressure on the middle, the bar can be re-flipped and molded as necessary while still in place in the chest. J, The bar is stabilized by attaching a stabilizer to the left end of the bar and wiring the bar and stabilizer together with #3 surgical steel wire. The stabilizer and bar are also secured by placing numerous interrupted absorbable sutures through the holes in the bar and adjacent fascia. An additional stabilizing technique involves a laparoscopic auto-suture needle to place multiple 0 PDS or Vicryl sutures around the bar and underlying ribs under thoracoscopic guidance.
(From Shamberger RC, Nuss D, Goretsky MJ. Surgical treatment of chest wall deformities. In: Spitz L, Coran AG, editors. Operative pediatric surgery, 6th edition. London: Hodder Arnold, 2006, with permission.)
In 2002, the results by Nuss and his associates using this technique in 303 patients were reported by Croitoru and coworkers.77 This included a group of children who were older than in the initial report (range, 21 months to 29 years; median age, 12.4 years). Two bars were required in 12.5% of the patients. Routine use of thoracoscopy to avoid cardiac injury was instituted in 1998 and routinely used thereafter. Lateral stabilizers were placed in 69.4% of the cases and were wired to the bar in 65.4% of cases. It is now recommended that the bars be left in the chest for 3 years. Epidural analgesic was administered for 2 to 4 days, and the median length of stay was 5 days, with a range of 3 to 10 days. The frequency of early complications was low. They included pneumothorax requiring aspiration, 1.0%; pericarditis, 2.3%, with only 0.3% requiring drainage; pneumonia, 0.7%; hemothorax, 0.3%; transient extremity paralysis, 0.3%; superficial wound infection, 2.3%; and bar infection requiring eventual removal of the bar, 0.7%.
A more recent summary by Shin and associates reported a series of 863 consecutive patients and showed a 1.5% incidence of infectious complications.78 This included six bar infections, four cases of cellulitis, and three stitch abscesses. Antibiotics and surgical drainage resolved the infection in three of the bar infections, but removal of the bar was required in three, one 3 months after surgery, and two 18 months after surgery. Staphylococcus aureus was involved in 83% of the cases. This degree of success in preserving the struts despite infectious complications was mirrored by the experience in two subsequent reports.79,80
Allergy to the retrosternal bars was also identified in 2.2% of the patients having MIRPE by Nuss and his group.81 The majority (63%) presented with rash and erythema, 32% had pleural effusions, and 15% were diagnosed on preoperative screening. It is critical to distinguish between allergic and infectious complications. The importance of placing the bars and stabilizers in a subcutaneous and not a submuscular position, to avoid extra-osseous bone formation around the strut and increased blood loss at the time of removal, has been demonstrated.82
Hebra and associates reported the results of a survey of members of the American Pediatric Surgery Association who had employed the minimally invasive (Nuss) technique.83 Thirty institutions contributed 251 cases, although it should be noted that 42% were performed by one surgeon. The complications reported were similar to those of Nuss and his associates, but the frequency was higher, presumably because the procedures were performed by more individuals less familiar with the operation. Displacement of the bar occurred in 9.2% of cases and pneumothorax requiring tube thoracostomy in 4.8%. Less frequently encountered complications included thoracic outlet syndrome, pericarditis, blood loss requiring a transfusion, cardiac injury, persistent cardiac arrhythmias, and erosion of the sternum by the bar. Many of the surgeons had adopted the use of thoracoscopy to improve the safety of passing the clamp anterior to the heart. Other surgeons elevate the sternum with a bone hook during passage of the clamp to open the retrosternal space anterior to the heart.
Engum and coauthors reported their series of 21 patients with a mean age of 8.2 years.84 Their patients had an average hospital stay of 4.9 days, which was comparable with the open repair. Complications encountered in their series were similar to the experience of others and included rotation of the bar, production of a marked pectus carinatum deformity, progressive chest wall asymmetry, and chronic persistent pain requiring removal of the bar in one case.
Molik and colleagues later enlarged this single-institution review and in a retrospective analysis compared 68 patients with standard surgical repair with 35 patients with a Nuss repair.85 The Nuss procedure required less time (3.3 hours) than the open technique (4.7 hours), but it had a higher complication rate (43%) than the open method (20%). Four patients with the standard operation (6%) and eight with the Nuss technique (29%) required reoperation. Length of stay was comparable between the open (4.8 days) and Nuss (4.0 days) techniques. The Nuss patients had a higher frequency of epidural analgesics postoperatively and an increased duration of patient-controlled analgesia after surgery.
Fonkalsrud and associates reported a similar retrospective comparison of the two techniques, each used at one institution.86 During a 5-year interval, 68 patients had the minimally invasive procedure and 139 had the open technique. There was a higher incidence of reoperations and hospitalizations in the Nuss group, but it was noted that 90% of the complications of this method occurred in the first 25 cases, again clearly demonstrating the role of experience in determining the frequency of surgical complications. It was difficult to determine in this study whether the differences noted in the use of epidural catheters and intravenous narcotics were attributable to truly different patient requirements or was a manifestation of institutional bias for analgesic techniques. There was a shorter mean hospitalization noted for the open procedure (2.9 days) than for the minimal-access procedure (6.5 days), and a similar difference between mean time before return to work or school (12 versus 18 days). These authors concluded that “long-term follow-up also will be required to assure both health professionals and the public that this is the procedure of choice for patients with pectus excavatum.”
The occurrence of “overcorrection” of the deformity or production of a true carinate deformity was first reported by Croitoru and associates and was associated with underlying connective tissue disorders (Marfan syndrome and Ehlers-Danlos syndrome).77 However, it was reported by Hebra to have occurred in an otherwise healthy 13-year-old boy 1 year after Nuss repair.87 What factors predispose some patients to this complication is not understood.
Other rare complications for the MIRPE include exsanguinating hemorrhage during removal of the strut, resulting from laceration of a pulmonary vessel, and bilateral sternoclavicular dislocation; cardiac tamponade and shock produced by erosion of the aorta by a strut that had rotated 90 degrees; and a near-fatal hemorrhage from erosion of the internal mammary vessel by a bar that had rotated 45 degrees.88–90
A prospective multi-institutional study of patients undergoing repair of pectus excavatum has been completed. It is hoped that this study will better define the physiologic effects of repair. Early reports from this study suggest that the pain and complications from the MIRPE and open procedures are similar and that repair can be accomplished with limited risks to the patients.91
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree