Bronchoscopic Evaluation of the Lungs and Tracheobronchial Tree
William H. Warren
L. Penfield Faber
Visual inspection of the airways was once an art performed by a handful of highly skilled surgeons using rigid instrumentation. It was almost always performed in the operating theater under general anesthesia. In the early 1970s, fiberoptic bronchoscopy was introduced, revolutionizing examination of the airways. Over the ensuing years, flexible bronchoscopy has almost completely replaced the rigid bronchoscope as the diagnostic instrument of choice. Flexible bronchoscopy is now performed by pulmonologists, anesthesiologists, otolaryngologists, critical care specialists, and thoracic surgeons in a variety of clinical settings, including the hospital ward, intensive care unit, and operating room. The well-trained thoracic surgeon must have a clear understanding of all aspects of both flexible and rigid bronchoscopy, including indications, anesthetic techniques, instrument options, and the management of complications.
Rigid Bronchoscopy
The indications for rigid bronchoscopy include examination and management of obstructing airway lesions, retrieval of foreign bodies, suctioning of inspissated secretions, and obtaining tissue biopsy specimens of endobronchial lesions deeper and more generous than those provided by the flexible bronchoscope. A rigid bronchoscope should always be available when tracheal lesions are examined. Bleeding from biopsy sites or edema from tissue trauma can obstruct a compromised lumen, which can be forcibly dilated with a rigid instrument, and ventilation can be restored through the instrument.
The rigid bronchoscopes most commonly used in adults have an internal diameter of 6, 7, or 8 mm and are 40 cm in length (Fig. 17-1). Traditional models provide only a tunnel view, but modern rigid bronchoscopes also can be used with telescopic lenses. Illumination is supplied from a halogen light source. A fiberoptic cable is attached to a light carrier that passes down the side wall of the bronchoscope, through the telescope, or both. With the help of the telescope, generous biopsy specimens can be taken without compromising visualization. Both models have a ventilating side port permitting assisted ventilation (Fig. 17-2).
Rigid bronchoscopy is almost always performed under general anesthesia, with all of its attendant risks. Despite the clearer view provided with telescopes, sampling of the upper lobe or distal airway cannot easily be accomplished through the rigid bronchoscope. The advantages and disadvantages of rigid bronchoscopy are listed in Table 17-1.
Rigid bronchoscopy is generally performed under general anesthesia, using the ventilation port on the side of the bronchoscope and capping the end of the scope with an eyepiece to convert the procedure to a nearly closed system. If no telescope is being used, the glass eyepiece must be removed for biopsy and aspiration (Fig. 17-3). If a telescope is used, plastic caps seal the scope. Some loss of tidal volume around the bronchoscope is inevitable, but this can be minimized by packing the hypopharynx with saline-soaked gauze. The anesthesiologist must monitor the adequacy of ventilation continuously; increased minute ventilation and tidal volumes may be required.
To pass the bronchoscope, the patient is placed in a supine position with an assistant positioning the head so that the neck is slightly flexed and the chin extended. The endoscopist elevates the epiglottis with the tip of the bronchoscope and passes the lubricated instrument through the glottis and into the upper trachea with the scope initially rotated so that the tip is lateral. It is important that the patient’s upper teeth be protected with the operator’s thumb or long finger and that the vocal cords be viewed by an anterior displacement of the bronchoscope rather than a lever motion on the teeth. Telescopes provide a magnified field of vision. Examination of the trachea is performed as the bronchoscope passes through the glottis and advances down to the carina. The carina is assessed for sharpness and mobility during ventilation. Splaying or fixation suggests the presence of a subcarinal mass. Biopsy of the bronchial mucosa on the carina is performed if mucosal or submucosal changes are observed. Thirty- and 90-degree angle-viewing telescopes allow the endoscopist to view the upper lobe bronchi. Alternatively, a flexible bronchoscope can be passed through an appropriately sized rigid instrument to view upper lobe orifices and all segmental bronchi (Fig. 17-4).
The risks of rigid bronchoscopy include injury to the gums and tooth dislodgment, hypoventilation, airway bleeding, and direct injury to the larynx or rupture of the tracheobronchial tree. With experience, these complications can be minimized.
The rigid bronchoscope must be sterilized after each use. The scope and ancillary equipment must be mechanically cleaned of
all blood and mucous debris before they are sent for sterilization. All glass and metal components should be autoclaved, and the plastic caps should be wrapped separately and gas sterilized.
all blood and mucous debris before they are sent for sterilization. All glass and metal components should be autoclaved, and the plastic caps should be wrapped separately and gas sterilized.
Flexible Bronchoscopy
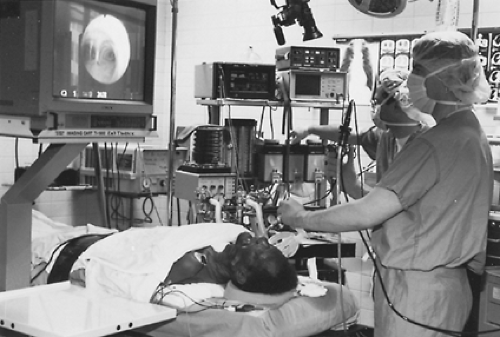
Ideally, flexible bronchoscopy should be performed in a dedicated area of the hospital or outpatient facility. Because intravenous sedation is routinely used, recovery areas are also needed. In the best clinical scenario, such suites would allow clinicians to perform procedures with fluoroscopic or computed tomography (CT) guidance and with the ability to administer general anesthesia. The examination room must be large enough to store all equipment, supplies, and accessories. It is ideal to have a storage area adjacent to the endoscopic suite. Special holding racks and storage cases should be routinely used to minimize the risk of scope breakage.
Table 17-1 Rigid Bronchoscopy | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
A portable cart supplied with a light source, bronchoscope, and accessories is a convenient way to perform bedside bronchoscopy. It should be returned to the storage area for proper cleaning and restocking.
Flexible bronchoscopy has many advantages over rigid bronchoscopy, as listed in Table 17-2. Diagnostic material from peripheral as well as central lesions can be obtained with minimal risk. The examination is usually performed under topical anesthesia and intravenous sedation, avoiding the risks of general anesthesia. Retained secretions can be aspirated at the bedside in the ward or in the intensive care unit. Patients receiving ventilator support can be examined without compromising the airway using a side-arm adaptor. The scope can be passed through narrowed and distorted airways or beyond an obstructing lesion. Flexible bronchoscopy is particularly valuable in
assessing lobar and segmental airways to the upper lobes. Still or videophotography is easily performed to document findings (Fig. 17-5).
assessing lobar and segmental airways to the upper lobes. Still or videophotography is easily performed to document findings (Fig. 17-5).
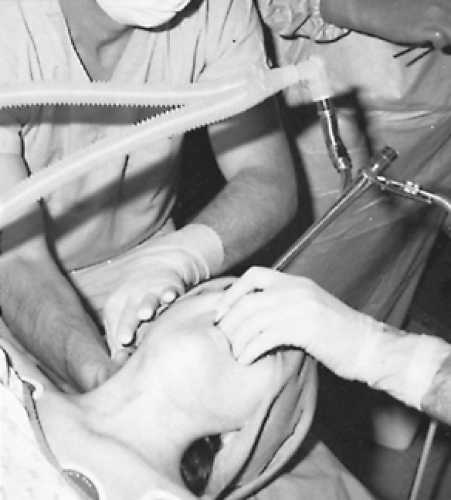 Figure 17-3. Rigid ventilating bronchoscopy with biopsy performed using general anesthesia and open-tube technique. |
A new and persistent cough or a change in the cough pattern of a smoker warrants a bronchoscopic examination. Furthermore, a wheeze, particularly one that is unilateral and fails to clear with coughing, should be investigated. In so doing, lesions (especially those in the trachea and major airways) can be diagnosed, even in the absence of radiologic findings.
Sputum can be obtained in a sterile fashion to assess for opportunistic infections, especially in the immunocompromised patient. However, if the patient is suspected of having tuberculosis, induced sputum specimens (using inhaled saline) should be examined before the scope and the health-care workers can become contaminated. Endoscopic lobar and segmental saline lavage has proven to be of benefit in postoperative patients unable to clear their secretions. Retained thick, tenacious mucous secretions are easily suctioned at the bedside with minimal patient discomfort.
 Figure 17-4. Flexible bronchoscope passed through a rigid bronchoscope to examine upper lobe orifices and airways beyond the tip of the rigid bronchoscope. |
Table 17-2 Flexible Fiberoptic Bronchoscopy | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Although the most common cause of hemoptysis is chronic bronchitis, carcinoma is common in patients with abnormal chest radiographs. Even if the chest radiograph is normal, according to Jackson16 and Poe42 and their associates, malignancy should be considered seriously if the patient is ≥40 years of age, has a significant smoking history, or has episodes of hemoptysis >1 week in duration. In addition to lung cancer, hemoptysis associated with a wheeze or atelectasis may be caused by a bronchial carcinoid or an inflammatory bronchial stricture.
An abnormal chest radiograph suggesting carcinoma warrants a careful evaluation of the entire tracheobronchial tree. Clinical judgment should always be used in deciding when to use bronchoscopy, but the physician should err on the side of performing an endoscopic evaluation to rule out a neoplasm. An obstructing carcinoma may be the underlying cause of an unresolving pneumonia; an upper lobar infiltrate should be viewed with particular suspicion. Bronchoscopy may also be used to assess for metastatic tumors.
Bronchoscopy is indicated in the diagnosis and management of lung abscess. The passage of brushes and biopsy forceps into the abscess cavity can promote bronchial drainage.
Furthermore, occasionally an obstructing neoplasm or foreign body is discovered.
Furthermore, occasionally an obstructing neoplasm or foreign body is discovered.
The therapeutic value of bronchoscopy to remove aspirated gastric contents remains in question. However, a rapid and efficient bronchoscopy examination can support the diagnosis and may have therapeutic benefit, particularly if particulate matter is retrieved.
After inhalation of smoke or caustic fumes, bronchoscopy is a safe and expeditious way of assessing damage to the tracheobronchial mucosa. In these compromised patients, the airways frequently need repeated bronchoscopy to debride necrotic mucosa and inspissated secretions. The inflammatory response to this injury may progress for days, leading to delayed airway obstruction.
Patients with cystic fibrosis may also, on occasion, require aspiration of persistently thick and tenacious secretions. However, routine bronchoscopy, once a mainstay of the care in such patients, has been largely supplanted by the routine use of vigorous physiotherapy and mucolytic agents.
The role of bronchoscopic examination of a patient with a fever of unknown origin or a pulmonary infiltrate after lung transplantation is widely accepted, as summarized by Higenbottam and colleagues.16 However, the need for routine surveillance biopsies in lung transplant recipients is still controversial. According to Trulock,56 routine surveillance bronchoscopies with random biopsies should be performed 2 to 3 weeks, 6 to 8 weeks, 9 to 12 weeks, 6 months, and 1 year after transplantation and annually thereafter. The most frequent unsuspected findings have been acute rejection and silent cytomegalovirus pneumonia.
Early detection and treatment of these entities is thought to decrease the risk for chronic rejection, according to Steinhoff and associates.52 Others, such as Stillwell and colleagues,53 maintain that in asymptomatic patients, early stages of acute rejection or viral infection pose little risk of leading to more serious conditions.
Another indication for bronchoscopic assessment of the tracheobronchial tree is the suspicion of airway trauma. Wheezing, hemoptysis, and the presence of subcutaneous or mediastinal emphysema are the classic findings, but injury of the airway is often overshadowed by concomitant injuries such as aortic rupture or myocardial contusion. Even when suspected, the endoscopic findings of a torn bronchus can be remarkably subtle. Early diagnosis and repair of these injuries are of the utmost importance.
Transbronchoscopic bronchial biopsies, cytologic brushings, transbronchial needle aspiration (TBNA), and bronchoalveolar lavage (BAL) are readily performed; the endoscopist should be familiar with indications, techniques, and complications of each. Airway bleeding can be the most devastating complication. Even severely ill and debilitated patients can undergo bronchoscopy safely if it is performed in an appropriate facility by an experienced endoscopist.
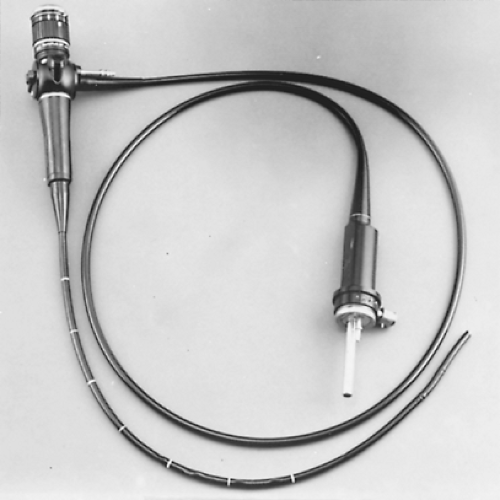
Currently available standard flexible bronchoscopes range from 6.2 mm in outer diameter (3.2-mm working channel to aspirate inspissated secretions and blood clots) to 2.7 mm in outer diameter (1.2-mm working channel) for pediatric patients (Table 17-3). One of the most often used adult bronchoscopes has an external diameter of 5.9 mm and a 2.2-mm working channel, allowing clear visualization and specimen sampling of the entire tracheobronchial tree down to the fourth- or fifth-order bronchi (Fig. 17-6). A forward field of view is 120 degrees; the angle of deflection is 180 degrees upward and 130 degrees downward. A narrower instrument with an external diameter of 4.9 mm and an instrument channel of 2.2 mm easily passes through a bronchus narrowed by stricture or tumor, and its 180-degree upward deflection facilitates examination of the often difficult to reach apical subsegment.
Table 17-3A Standard Viewing Fiberoptic Bronchoscope Specifications | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Table 17-3B Videofiberoptic Bronchoscopes | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Videobronchoscopes provide a significantly larger image with higher resolution than is seen in standard flexible bronchoscopes. However, these images can be viewed only with a videoprocessor and monitor. For these superior images, however, these bronchoscopes may have a larger outer diameter (Table 17-3). These scopes are fully immersible and most can be autoclaved. The active endoscopist should have a variety of flexible bronchoscopes available for diagnostic and therapeutic versatility (Fig. 17-7).
The Examination
Anesthetic Considerations
Intravenous access is routinely available, and supplemental oxygen is usually provided through nasal cannulas, as recommended by Prakash and Stubbs.43 Pulse oximetry, cuff blood pressure, and electrocardiographic monitoring are routinely performed. Sedation should be administered to provide patient comfort and cooperation. In several studies, however, 50% of the life-threatening complications from bronchoscopy stemmed from hypoxemia, hypercapnia, and respiratory depression
secondary to oversedation. Therefore the agents and their dosages must be individualized; their use may not be necessary in every case.
secondary to oversedation. Therefore the agents and their dosages must be individualized; their use may not be necessary in every case.
Preoperative opiates are sometimes given for their analgesic and antitussive properties. Meperidine has an elimination half-life of 3.2 hours, but clearance is decreased in patients with renal or hepatic failure or both. It, like all opiates, can cause respiratory depression and hypotension. Naloxone, a specific opioid antagonist, must be available whenever opioids are administered.
Intravenous benzodiazepines are often administered, providing anxiolysis and antegrade amnesia. Midazolam has become the preferred agent, according to a bronchoscopy survey reported by Colt and colleagues.3 It is water-soluble, with a rapid onset, short duration of action, and an elimination half-life of only 2 hours in normal subjects. Renal failure does not alter the distribution, elimination, or clearance, but liver disease is associated with prolonged sedation. The recommended dose is 0.07 mg/kg. Diazepam is not water-soluble and can cause significant if only transient phlebitis. Its elimination half-life is 24 to 57 hours in normal patients. Both agents should be used with caution, especially in elderly patients and those with limited pulmonary reserve. Respiratory depression is a major side effect of both benzodiazepines; the degree of respiratory depression is similar with both drugs. Flumazenil is a specific benzodiazepine antagonist and should be available any time these agents are used. Because its elimination half-life is only 1 hour, repeat doses or a continuous infusion may be needed. Matot and Kramer27 have reported that alfentanil–propofol is a reasonable alternative to meperidine–midazolam, particularly in patients with known coronary artery disease or who are at risk for it (see Table 17-4).
Some physicians continue to premedicate patients with an antisialagogue, such as atropine or glycopyrrolate, to reduce secretions and inhibit vasovagal responses, as documented by Williams and colleagues.61 These medications also make topical anesthesia more effective. However, according to Hasanoglu and coworkers,11 tachycardias and episodes of hypotension were
more commonly observed when atropine was administered. According to the survey reported by Colt and colleagues,3 only 62% of respondents routinely used atropine as a premedication. Far fewer used glycopyrrolate.
more commonly observed when atropine was administered. According to the survey reported by Colt and colleagues,3 only 62% of respondents routinely used atropine as a premedication. Far fewer used glycopyrrolate.
Table 17-4 Indications for Bronchoscopy | |||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Topical anesthesia is preferred for fiberoptic bronchoscopy, but general anesthesia may be considered, particularly for prolonged examinations required to identify carcinoma in situ in a patient with normal chest radiography findings. It is occasionally requested by extremely anxious patients.
The most common agents for topical anesthesia are lidocaine (1% and 2%) and tetracaine (0.5%, 1.0%, and 2.0%). Complications from topical anesthesia usually result from the administration of excessive amounts, as documented by Credle,4 Suratt,55 and Pereira38 and their associates. If carefully measured amounts are given and the endoscopist is always aware of the total milligram dosage instilled, reactions are minimized.
Lidocaine is a safe agent with a recommended dose of 0.2 to 0.3 mL of 1%/kg, but larger amounts have been given without serious side effects. It is important to remember that the extent of absorption and bioavailability depends on tissue vascularity and the technique of application as well as the total dose administered, according to Mainland and associates.24 Lidocaine should always be titrated slowly, and patients must be monitored for mental status changes. The first sign of toxicity is usually central nervous system excitation or seizure before cardiovascular collapse. The duration of action is short.
Tetracaine is another effective topical anesthetic, but side effects frequently occur when a dose of 80 mg is exceeded. The duration of action is prolonged, and the first sign of toxicity may be sudden cardiovascular collapse.
The combination of benzocaine–tetracaine is a popular topical anesthetic agent with rapid onset of action that is useful in rapid intubation. It is often administered in a propellant spray with a tolerance dose delivered in only 2 seconds. Dosing is therefore difficult to regulate, and this agent should be avoided when flexible bronchoscopy is being performed. Complications of all topical anesthetic agents include methemoglobinemia, whereby patients become cyanotic owing to the oxidation of iron from its normal reduced state, leaving it unavailable for oxygen binding and transport. Elderly, infant, and anemic patients are particularly susceptible, as documented by Karim and coinvestigators.17 Intravenous methylene blue (1 mg/kg) is the recommended initial definitive treatment. Rarely, exchange transfusion or hemodialysis is necessary.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree