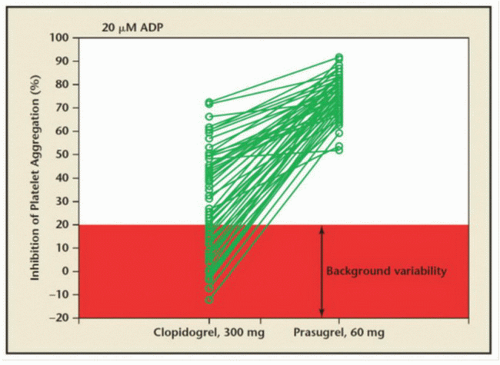
Dosing for Percutaneous Coronary Intervention
Preclinical and clinical pharmacodynamic studies suggest that 80% inhibition of platelet aggregation by light transmission aggregometry should be the target for clinically effective antiplatelet activity.
94 The level of platelet inhibition varies between the three GP IIb/IIIa inhibitors following the recommended bolus and infusions.
95 In general, the bolus and infusion regimen of abciximab and the double bolus and infusion regimen of eptifibatide are associated with rapid and profound inhibition of platelet function.
95,
96,
97 Several studies have documented that the FDA-approved bolus and infusion regimen for tirofiban achieves suboptimal levels of platelet inhibition for up to 4 to 6 hours, and this suboptimal level of inhibition likely accounts for inferior clinical results in the PCI setting.
97The clinical relevance of these pharmacodynamic observations was tested in the TARGET (Tirofiban and ReoPro Give Similar Efficacy) trial, which randomized 5,308 patients to tirofiban (10 µg/kg bolus followed by an infusion of 0.15 µg/kg/ minute for 18 to 24 hours post PCI) or abciximab before undergoing PCI with the intent to perform stenting.
98 The primary endpoint was a composite of death, nonfatal MI, or urgent target vessel revascularization (TVR) at 30 days. The primary endpoint (6.0% versus 7.6%,
P = 0.038) as well as the incidence of MI (5.4% versus 6.9%,
P = 0.04) were significantly lower in the abciximab group compared to the tirofiban group, respectively. There was no significant difference in the rate of major bleeding between the two groups. Increasing the tirofiban bolus 2.5-fold (25 mg/kg) appears to enhance platelet inhibition and improve PCI outcomes compared to the FDA-approved dosing regimen.
99 The degree of platelet inhibition appears central to the efficacy of GP IIb/IIIa inhibitors; achieving >95% platelet inhibition 10 minutes after the bolus in patients undergoing PCI was associated with a 55% reduction in MACE compared to those patients with <95% platelet inhibition.
100
Evidence for Use in Percutaneous Coronary Intervention Patients
The landmark trial demonstrating efficacy of GP IIb/IIIa inhibition in the balloon angioplasty setting is the Evaluation of IIb/IIIa platelet receptor antagonist 7E3 in Preventing Ischemic Complications (EPIC) trial.
104 In this study, high-risk patients undergoing balloon angioplasty were randomized to abciximab bolus and infusion versus abciximab bolus alone versus placebo. The group treated with abciximab bolus and
infusion had a 35% lower rate of death, MI, or unplanned urgent revascularization at 30 days compared to the placebo group (8.3% versus 12.8%,
P = 0.008). No significant benefit with abciximab bolus alone was observed, suggesting that shorter duration of platelet inhibition was insufficient to favorably affect clinical outcomes. Major bleeding complications occurred in an unacceptably high proportion of patients treated with abciximab compared to placebo (major bleeding 14% versus 7%, transfusion 15% versus 7%, respectively). Procedural modifications, including performing front-wall arterial access only, reducing arterial sheath size from 8F to 6F, reducing heparin dosing to target activated clotting time (ACT) 200 to 250 seconds rather than >300 seconds, removing sheaths as soon as possible (ACT
< 180 seconds) rather than overnight, and abandoning the use of routine venous sheaths, successfully reduced major bleeding complications to less than 1.0% to 1.5% in subsequent trials.
The benefit of GP IIb/IIIa inhibition patients undergoing elective stent placement has been shown in two large, randomized controlled trials.
101,
102 The Evaluation of IIb/IIIa inhibitor for Stenting (EPISTENT) trial randomized 2,399 patients to stent plus placebo, stent plus abciximab, or balloon angioplasty plus abciximab.
103 The primary 30-day endpoint, a combination of death, MI, or urgent revascularization, occurred in 10.8% of patients in the stent plus placebo group, 5.3% of those in the stent plus abciximab group (HR 0.48;
P < 0.001), and 6.9% in the group undergoing balloon angioplasty plus abciximab (HR 0.63;
P = 0.007). These benefits were maintained at 6 months
104 and 1 year,
105 with a significant reduction in 1-year mortality in patients treated with stent plus abciximab compared with stent without the IIb/IIIa inhibitor (2.4% versus 1.0%,
P = 0.037). No significant differences in bleeding complications were noted among the various groups.
The Enhanced Suppression of the Platelet IIb/IIIa Receptor with Integrilin Therapy (ESPRIT) trial randomized 2,064 patients undergoing stenting to eptifibatide (180 µg/kg bolus followed by a 2.0 µg/kg/hour infusion, with a second bolus of 180 µg/kg given 10 minutes after the first bolus) or placebo.
102 In this trial, patients were administered a loading dose of clopidogrel or ticlopidine on the day of the procedure. The trial was terminated early for efficacy. The primary endpoint—a composite of death, MI, urgent revascularization, or thrombotic bailout at 48 hours—was reduced by 37% with eptifibatide (10.5% versus 6.6%,
P = 0.0017). Death or MI at 48 hours was significantly reduced with eptifibatide compared to placebo (5.5% versus 9.2%, RRR = 40%,
P = 0.0013). These benefits were maintained at 6 months
106 and 1 year.
107 Major bleeding was rare, but occurred more frequently in patients receiving eptifibatide compared to placebo (1.3% versus 0.4%, respectively;
P = 0.027).
Three large, randomized clinical trials have evaluated each of the three GP IIb/IIIa inhibitors in UA/NSTEMI patients undergoing PCI. In the c7E3 Fab Antiplatelet Therapy in Unstable Refractory Angina (CAPTURE) trial, patients (
n = 1,265) with refractory angina undergoing PCI were randomly assigned to receive abciximab 18 to 24 hours prior to PCI and for 1 hour after completion of the procedure or placebo.
108 MACE was reduced in the abciximab group compared to placebo (15.9% versus 11.3%,
P = 0.012, respectively). There was a significant increase in major bleeding (1.9% versus 3.8%,
P = 0.043) and the need for transfusion (3.4% versus 7.1%,
P = 0.005) in the abciximab group compared to placebo. The Platelet Receptor Inhibition in Ischemic Syndrome Management in Patients Limited by Unstable Sign and Symptoms (PRISM-PLUS) trial randomized 1,915 patients with UA/NSTEMI to tirofiban alone, tirofiban plus heparin, or placebo infusion plus heparin.
101 The combination of tirofiban and heparin led to a 32% risk reduction in the rate of death, MI, or recurrent refractory ischemia at 7 days compared with heparin alone (12.9% versus 17.9%, respectively,
P = 0.004). Tirofiban was also beneficial in the subgroup of patients undergoing PCI (RRR of death or MI at 30 days = 0.44).
In the Platelet Glycoprotein IIb/IIIa in UA: Receptor Suppression Using Integrilin Therapy (PURSUIT) trial, 10,948 patients with UA/NSTEMI were randomized to eptifibatide or placebo.
109 The primary endpoint of 30-day death or MI was reduced in those patients receiving eptifibatide versus placebo (14.2% versus 15.7%,
P = 0.042, respectively). This treatment benefit was more pronounced among patients undergoing PCI within 72 hours of presentation (11.6% versus 16.7%,
P = 0.01, respectively). Moderate or severe hemorrhage was more common in the eptifibatide group (12.8% versus 9.9%,
P < 0.001, respectively).
Four trials support the use of abciximab in primary PCI for STEMI, reducing MACE at 30 days by 35% to 54%.
110,
111,
112,
113 In the Abciximab before Direct Angioplasty and Stenting in Myocardial Infarction Regarding Acute and Long-Term Followup (ADMIRAL) trial, 300 patients with acute MI were randomized to abciximab plus stenting or stenting alone prior to angiography.
112 At 30 days, the primary endpoint—a composite of death, reinfarction, or urgent TVR—occurred in 6% of the patients in the abciximab group, as compared with 14.6% of those in the placebo group (P = 0.01). This beneficial effect was sustained at 6 months (7.4% versus 15.9%,
P = 0.02) in ADMIRAL, but not in the two other trials.
110,
111,
112,
113Upstream administration of GP IIb/IIIa inhibitors prior to PCI in patients presenting with STEMI has been studied in six trials. In a metaanalysis of these trials, TIMI grade 3 flow (20.3% [84/413] versus 12.2% [51/418]) was significantly more common in the early (prior to transfer to the cardiac catheterization laboratory) group compared with the late (catheterization laboratory) group (odds ratio [OR], 1.69; 95% CI, 1.28 to 2.22;
P < 0.001; and OR, 1.85; 95% CI, 1.26 to 2.71;
P < 0.001, respectively). The early administration of GP IIb/IIIa inhibitors was associated with a 28% reduction of mortality from 4.7% to 3.4%, which was not significant but consistent with similar trends for reinfarction and the composite ischemic endpoint. Thus, early administration of GP IIb/IIIa inhibitors in STEMI appeared to improve coronary patency with favorable trends for clinical outcomes. However,
the prospective, randomized EARLY-ACS trial showed no significant benefit of upstream initiation compared to cardiac catheterization laboratory initiation of eptifibatide.
114GP IIb/IIIa inhibitors have been shown to reduce major adverse cardiac events (death, MI, and urgent revascularization) by 35% to 50% in patients undergoing PCI
115 (
Table 5.1). Although no single study demonstrated a significant reduction in mortality alone with GP IIb/IIIa inhibitors, a metaanalysis suggests that these agents as a class reduce death by 20% to 30%
119 (
Figure 5.3). The mechanism by which GP IIb/IIIa inhibitors reduce long-term mortality is unclear, and cannot be explained solely by their ability to reduce periprocedural death or MI. Investigators have postulated that this therapy may also be associated with significant anti-inflammatory properties that may favorably influence the course of atherothrombotic disease,
116,
117 a contention that remains unproven at this time.
Although GP IIb/IIIa inhibitors have demonstrated ability to reduce adverse cardiovascular events, in the modern era of thienopyridine use and stenting, the clinical benefit of routine GP IIb/IIIa inhibitors has been inconsistent. The CADILLAC (Controlled Abciximab and Device Investigation to Lower Late Angioplasty Complications) trial demonstrated that STEMI patients treated with stenting did not benefit as much as patients treated with balloon angioplasty.
113 In lowrisk elective PCI patients that were preloaded with 600 mg of clopidogrel, abciximab increased bleeding and did not improve ischemic outcomes.
118,
119 Although there is no benefit to routine GP IIb/IIIa inhibitor use in thienopyridinetreated low-risk elective PCI cohorts, data do justify targeted GP IIb/IIIa inhibitor use in high-risk patients. In the Intracoronary Stenting and Antithrombotic Regimen: Rapid Early Action for Coronary Treatment-2 (ISAR-REACT-2) study abciximab reduced ischemic endpoints without increasing bleeding in high-risk ACS patients.
120Although older studies demonstrated reduction in ischemia when GP IIb/IIIa inhibitors were used upstream before PCI,
121,
122 a recent study performed in the thienopyridine and stenting era showed that in ACS patients undergoing invasive management, upstream GP IIb/IIIa inhibitor use increased bleeding to the same degree that it reduced ischemic events compared to a strategy of provisional GP IIb/IIIa inhibitor use at the time of PCI.
114 Similarly, the Facilitated Intervention with Enhanced Reperfusion Speed to Stop Events (FINESSE) trial demonstrated that in ST-segment elevation MI patients, early abciximab use increased bleeding without clinical benefit when compared to provisional use during PCI.
Table 5.2 summarizes data from trials examining GP IIb/IIIa inhibitor use in the modern era of thienopyridine preloading and stenting.