Chapter 64 Vascular Trauma
Vascular trauma remains one of the most significant challenges in the management of injured patients. The advent of trauma systems and improved prehospital care have resulted in an increasing number of patients with what were previously fatal vascular injuries arriving at trauma centers still alive, but in immediate danger of death.1,2 Vascular trauma is not always obvious and timely recognition may be difficult in patients with multiple injuries.3 The time urgency and high-risk potential in managing these injuries requires an organized approach to deliver appropriate care in a timely fashion. Because so much is at stake, this is a high-risk clinical environment requiring systematic planning, preparation, and the use of practice guidelines for successful early recognition and effective treatment.
General Approach To Vascular Trauma
Mechanism of Injury
Vascular injury can be produced by a blunt or penetrating mechanism. Penetrating injury tends to be more discrete or focal, whereas blunt injury is more diffuse, with injury not only to the vascular structures but also to the bone, muscle, and nerves. The diffuseness of the blunt injury not only affects the major vascular conduit, but also disrupts smaller vessels that would normally provide collateral flow. As a result, ischemia is worsened or exaggerated. Penetrating injury is generally classified as low velocity (<2500 ft/sec; e.g., stab wound, fragment injury, handgun wound) or high velocity (>2500 ft/sec; e.g., military rifle wound).4 High-velocity weapons produce significantly more tissue damage than low-velocity weapons for three main reasons5,6:
In addition to the acute pathophysiology produced by hemorrhage and thrombosis, direct trauma can produce subacute, chronic, or occult injuries. The most common are arteriovenous fistula and pseudoaneurysm. An arteriovenous fistula typically occurs after penetrating trauma that causes injury to an artery and vein in close proximity. The high-pressure flow from the artery will follow the path of least vascular resistance into the vein, producing local, regional, and systemic signs and symptoms. These include local tenderness and edema, regional ischemia from steal, and congestive heart failure if the fistula enlarges.7 A pseudoaneurysm is a result of a puncture or laceration of an artery that bleeds into and is controlled by the surrounding tissue. The artery remains patent; blood flows into and out of the pseudoaneurysm, much like flow from the ocean into a tide pool. These can enlarge and produce local compressive symptoms, erode adjacent structures or, rarely, be a source of distal emboli. Initially, they can be clinically occult, but with time become symptomatic.
Not all arterial injuries require operative management. During the past 2 decades, it has been convincingly demonstrated that patients with a normal vascular physical examination and asymptomatic nonocclusive intimal flaps, segmental arterial narrowing, small (<2-cm) false aneurysms, or small arteriovenous fistulas discovered on arterial imaging (i.e., duplex scanning or arteriography) have a benign clinical course.8 Approximately 10% of these minimal injuries will progress to require a surgical or endovascular repair and most within the first week after injury. Thus, it is imperative that these patients have close follow-up with physical examination and duplex imaging, if needed. In rare cases, when a nonocclusive minimal injury increases in size or becomes symptomatic, requiring operative or endovascular repair, morbidity is not increased by the delay.
Clinical Presentation
Extremity vascular trauma may be immediately apparent on presentation because of external hemorrhage, hematoma, or obvious limb ischemia. A history of penetrating trauma associated with hypotension, pulsatile bleeding, or a large quantity of blood at the scene suggest vascular injury. Blunt trauma is also capable of causing significant vascular injury that can be overlooked when serious head, chest, or abdominal injuries are present. Extremity fractures may result in vascular injury. Supracondylar humerus fractures can be associated with brachial artery injury and knee dislocation carries a significant risk of popliteal artery injury.9 Crush injuries of the extremity without fracture may also result in vascular injury.
A relatively small number of vascular injuries present in a delayed fashion, without initial findings. These are limited to thrombosis of a previously partially disrupted but initially patent vessel, distal emboli from an intimal tear of the arterial wall with formation of platelet debris and, least commonly, rupture or expansion of a pseudoaneurysm that was initially small and contained by the outer arterial wall and local tissue.9 Local signs of hematoma, diminished pulses, and the presence of patterns of associated injuries should indicate the presence of these vascular injuries. A thorough history and physical examination and appropriate adjunctive imaging studies will result in an effective initial diagnosis and result in a decrease in the frequency of these delayed presentations.
Diagnosis
Physical Examination
Vascular injury can produce systemic symptoms of hypotension, tachycardia, and altered mental status because of hypovolemic shock produced by hemorrhage. As a result, vascular injury can be life-threatening and attention must initially be directed to the primary survey using the principles of advanced trauma life support (ATLS).10 The airway must be assessed, adequate oxygenation and ventilation ensured, and intravenous access achieved. Once this is completed and resuscitation is underway, the secondary survey is undertaken. A thorough history is obtained and careful physical examination is performed. This examination must include a careful inspection of the injured sites and wounds, complete sensory and motor assessment, and pulse examination of each extremity. The presence of a hematoma, bruit, or thrill must be noted. If distal pulses are diminished or absent, ankle or wrist systolic blood pressure should be determined with a continuous wave Doppler device and compared with the uninjured side. A significant difference in systolic blood pressure (>10 mm Hg) between extremities may be an indication of vascular injury. Patients with hard findings of vascular injury (Box 64-1) should be taken directly to the operating room.
Box 64-1 Findings of Vascular Injury
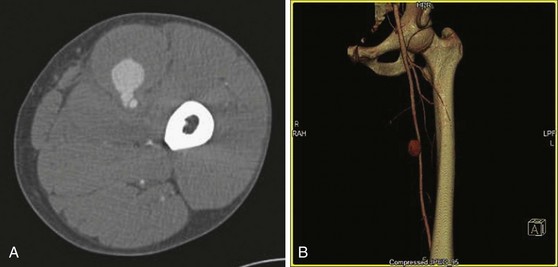
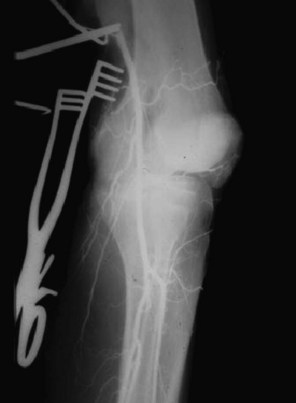
In patients without hard findings, but with soft findings (see Box 64-1), vascular imaging can be used to rule out the need for operation. Additionally, patients with hard findings but with multilevel injuries in the same extremity may also need imaging. Catheter arteriography is sensitive and specific for the diagnosis of extremity vascular injuries (Fig. 64-1). Computed tomography angiography (CTA) with the latest generation scanners has proved to be an acceptable alternative to formal arteriography (Fig. 64-2).11,12 Although this imaging technique requires a contrast infusion, it does not require arterial catheterization, is easily performed and is less costly and time-consuming than conventional angiography.
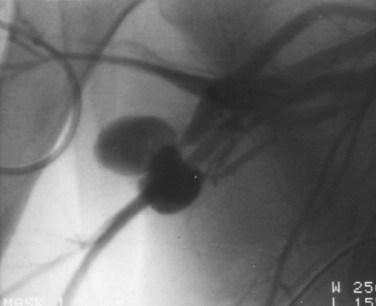
Some severely injured patients must be taken to the operating room to treat severe life-threatening associated injuries (e.g., subdural hematoma, ruptured spleen). In such cases, it is not prudent to delay operative therapy to obtain formal vascular imaging. An arteriogram can be obtained in the operating room by cannulating the artery proximal to the suspected vascular injury, injecting 20 to 25 mL of full-strength contrast, and taking an x-ray or using fluoroscopy (Fig. 64-3).13,14 If doubt remains about the presence of a vascular injury and imaging studies and other diagnostic tests are inconclusive, there is a role for operative exploration and direct assessment of the artery. Routine operative exploration in the stable patient with soft signs, however, has a 5% to 30% incidence of morbidity, occasional mortality, and low diagnostic yield.15 These patients are better served with formal vascular imaging.
Treatment
Minimal Vascular Injury and Nonoperative Management
A small, nonocclusive intimal flap is the most common clinically insignificant minimal vascular injury. The likelihood that it will progress to cause occlusion or distal embolization is approximately 10% to 15%.8,16 This progression, if it occurs, will be early in the postinjury course. Spasm is another common minimal vascular injury. This finding should resolve promptly after initial discovery. Failure of the return of normal extremity perfusion pressure indicates that a more serious vascular injury is present and intervention is needed. Small pseudoaneurysms are more likely to progress to the point of needing repair and must be actively followed with Duplex color flow imaging. Arteriovenous fistulas always enlarge over time and should be promptly repaired.
Endovascular Management
The use of endovascular therapy for the treatment of atherosclerotic arterial disease has become widespread. Endoluminal stent deployment for occlusive lesions and stent graft for aortic aneurysms have been used successively in select patients; there is now a strong tendency to generalize from this elective experience in older patients with atherosclerosis to the treatment of younger patients with acute vascular injuries. Most major trauma centers have some experience with endoluminal treatment of acute vascular lesions. However, the evidence to support these approaches is not well developed and there have been problems.17–19 A review of available evidence combined with common sense should help identify the appropriate role of endovascular management of traumatic injuries.9,18
Torso Injuries
Endovascular techniques offer various options for hemorrhage control in the torso. Intra-arterial catheter-directed embolization has become a mainstay of the management of solid organ hemorrhage in the abdomen.20,21 Whether used as the sole treatment or in combination with open procedures, this approach has been effective for liver, spleen, and kidney injuries. Less commonly used intra-arterial balloon occlusion for proximal control is a promising adjunct to open repair.22,23 These techniques are quick, accurate, and easily performed. The major obstacle to their widespread popularity has been the reluctance of surgeons to adopt catheter skills or partner with interventional radiologists to bring these techniques to the trauma operating room (OR). It does not require a dedicated endovascular suite to perform these techniques. A digital C arm and the proper catheters transforms any OR to an endovascular-capable room.
The early use of catheter-directed control of hemorrhage associated with pelvic fracture is an effective method of limiting blood loss and improving outcome.24 This approach is well tolerated and has proven superior to open attempts at hemorrhage control by packing in most patients. Unstable patients benefit from an immediate trip to the operating room. If intraoperative endovascular capability is available, a combined approach may offer the best results.
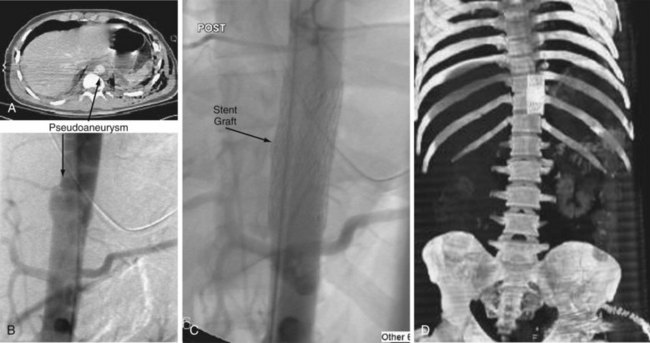
Enthusiasm for stent graft management of great vessel injuries in the chest has steadily grown. The success of stent grafts for the treatment of aneurysm disease in the infrarenal aorta has led to the use of similar devices to treat contained thoracic aortic lacerations following blunt trauma. The initial results have been encouraging but are not without complications.18 Lifelong CT imaging is necessary because of the possibility of delayed endoleak and possible loss of device fixation as the aorta enlarges over time. There is a promising role for covered stents in proximal branches of the aorta in the thorax and abdomen. In stable injuries at risk for delayed hemorrhage or thrombosis, carefully placed stents have the potential to lower morbidity compared with open procedures that require extensive operative dissection for exposure and control. Endoluminal management with stent grafts appears most effective in those torso injuries that are surgically inaccessible, with the potential for significant hemorrhage in stable patients (Fig. 64-4). These techniques should only be used in centers with an active elective endovascular practice that has experience in treating trauma patients.
Cerebrovascular Injuries
Endovascular techniques offer advantages in anatomic regions in which direct operative control is difficult or impossible. For example, hemorrhage from a penetrating injury at the base of the skull is extremely difficult to control. Catheter-directed placement of coil, balloon, or hemostatic agent in the injured carotid or vertebral artery could be lifesaving. Initially, stent placement appeared to be less effective than anticoagulation in partially occluded injuries without associated hemorrhage.17,25 However, the role of stents in cerebrovascular trauma has yet to be defined and may prove safe.26 This use of endoluminal interventions, however, requires significant expertise and experience. If such experience does not exist at the receiving hospital, consideration should be given to transferring the patient to a medical center with experience in this mode of therapy.
Extremity Vascular Injury
The use of stent grafts in the extremities is becoming more common 27,28 The long-term results, however, have not been documented and caution should be used when considering this type of treatment. Covered stents can be used to improve intraluminal diameter in partially occluded traumatized vessels with favorable early patency rates; however, they are prone to occlusion and long-term outcomes are as yet unknown. Autologous vein interposition grafts have excellent long-term patency rates and remain the gold standard for vascular repairs in the extremities.
Catheter-directed therapies for controlling hemorrhage from large branch vessels in the extremities are often effective and sufficient to manage these injuries.29 Endoluminal treatment is not advocated for pseudoaneurysms of the extremity arteries. Small pseudoaneurysms are likely to resolve without any intervention and large pseudoaneurysms are best treated with open techniques because the risk of arterial thrombosis or distal embolization is high with this endovascular intervention.
Surgical Treatment
Preparation for Operative Management
Successful operative management of vascular injuries requires a systematic approach, with careful preparation. This begins with airway control, adequate intravenous access, and availability of blood products. However, these blood products should not be administered before obtaining control of hemorrhage unless the patient is profoundly hypotensive.30–32 If the blood pressure (BP) is below approximately 80 to 90 mm Hg, the goal should be to provide adequate volume restoration with O-negative packed cells and type AB fresh-frozen plasma infusion to support transport to the operating room for definitive hemorrhage control without delay. Volume infusion that raises the blood pressure above a systolic pressure of 90 to 100 mm Hg may increase bleeding and negatively affect outcome, particularly if the infusion delays transport to the OR.
The operative management of extremity vascular injuries must be carefully orchestrated with the overall care of the patient. The choice between definitive repair and damage control should be made as soon as possible in patients with life-threatening torso injuries or severe head injuries. This includes coordinating two surgical teams to work simultaneously to care for the torso injury and extremity vascular injury at the same time. Associated injuries to the soft tissue and bone require a coordinated assessment and treatment with orthopedic and plastic surgery consultants. These specialists should be involved as early as possible to facilitate any additional imaging or diagnostic procedures prior to proceeding to the OR. The conduct of the operation should also be discussed with these colleagues.32 For example, the use of damage control procedures with shunt placement, followed by orthopedic stabilization, can remove the sense of urgency to restore blood flow. Extensive soft tissue injuries may compromise the proper coverage of vascular repairs and fracture fixation. The advice and assistance of a plastic and reconstructive surgeon can be helpful in obtaining coverage of exposed grafts and fractures.
Vascular Exposure and Control
Proximal control is the first priority in the exposure of vascular injuries.33 In the torso, chest injuries with life-threatening hemorrhage are best approached through a fourth intercostal space anterolateral thoracotomy that can be extended across the sternum into the third intercostal space of the right chest to create a clamshell incision. Thoracic outlet and proximal neck vascular injuries may require median sternotomy, with extension above the clavicle, up along the ipsilateral sternocleidomastoid muscle. For abdominal vascular injuries, a generous xiphoid to pubis incision is needed for adequate exposure. Proximal control for aortic injuries can be obtained just below the aortic hiatus of the diaphragm or may require a left anterolateral thoracotomy to clamp the distal thoracic aorta.
There are various adjunctive measures that can obtain temporary control. In the resuscitation bay, insertion and inflation of a Foley catheter in a wound in the neck or extremities with active hemorrhage can obtain temporary control and allow for safe transfer to the operating room.30 In the OR, insertion of Fogarty balloon tip catheters under direct vision at sites in the artery above or below the injury also serves to gain control in difficult to reach anatomic areas.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree