Chapter 59 Vascular Infection
Primary Arterial Infections
The first published experience with primary arterial infection is generally credited to Koch, who reported a patient with a ruptured superior mesenteric artery (SMA) aneurysm in 1851.1 Arterial infections were not well appreciated before this time because the basic tenets of bacteriology and infection were not yet developed. Breakthroughs in human bacteriological research by Pasteur and others in the mid-19th century set the stage for a basic understanding of the pathophysiology and classification of vascular infections.
Classification and Etiology
Nearly all primary arterial infections result in the formation of aneurysms or pseudoaneurysms. Osler coined the term mycotic aneurysm in 1885 to describe infected aortic arch aneurysms containing “fresh fungal vegetation” in a patient who had concomitant aortic valve vegetations.2 While “mycotic” aneurysms apply only to the subset of individuals with infected aneurysms caused by septic emboli, the term has been loosely applied to include all infected aneurysms, regardless of etiology. This practice is confusing and prevents meaningful comparison of the numerous small series of infected aneurysms reported in the modern literature.
The most widely accepted classification of arterial infections was introduced by Wilson et al. in 1978.3 In keeping with the traditional definition introduced by Osler, the authors classified a mycotic aneurysm as one that occurred in an otherwise normal, nonaneurysmal artery as a result of septic emboli of endocardial origin. A preestablished aneurysm that became infected as a result of bacteremia is classified as an infected aneurysm. Microbial arteritis refers to infection of a normal or atherosclerotic (i.e., nonaneurysmal) artery that has become infected as a result of bacteremia. This most often results in rupture with formation of a pseudoaneurysm. Traumatic infected aneurysms include infected aneurysms due to trauma or iatrogenic injury (e.g., complications of arteriography). Contiguous arterial infection is due to direct extension of an adjacent infection into the wall of the artery, such as infected aortitis associated with vertebral osteomyelitis. The specific classification based on etiology will be acknowledged wherever appropriate in this chapter. However, for purposes of clarity and simplification, the generic term infected aneurysm will be applied to include all arterial infections discussed.
The etiology of infected aneurysms has changed in the past 150 years. In the pre-antibiotic era, 86% of patients with arterial infections had evidence of endocarditis.4 Following widespread use of antibiotics, the incidence of infective endocarditis has relegated the true mycotic aneurysm to a rare entity. In 1984, Brown and associates reported a collective series of infected aneurysms based on a search of the English literature.5 The etiology could be determined in 75% of the 180 subjects. The authors separated the reported experience into cases occurring before 1965 and those occurring since that time. Accuracy of the results suffers because of important differences in diagnosis and reporting frequency between the two periods. Nevertheless, this collective experience remains the largest comparative analysis of infected aneurysms to date. In the earlier part of the series, endocarditis was still the leading cause of infected aneurysms, but arterial trauma of all types became the leading cause after 1965. The authors attributed this etiological shift to a substantial change in the pattern of antibiotic use for treatment of sepsis and trauma.5 Other explanations include an increased prevalence of intravenous drug abuse and the widespread application of transarterial interventional procedures. The increase in interventional procedures was particularly notable after the pioneering work of Grunzig and others in the 1970s. The enthusiasm for endovascular technology seen in recent years suggests that arterial trauma may soon become an even more important cause of arterial infection.
Pathogenesis of Infected Aneurysms
The basic mechanisms leading to formation of infected aneurysms have been studied most extensively in the abdominal aorta. Chronic uninfected abdominal aortic aneurysms (AAAs) are thought to form as a result of both destruction of medial elastin and adventitial collagen (see Chapter 37).6 Elastolytic matrix metalloproteinases (MMPs) play a central role, leading to initial aortic dilation.6 Collagenase MMP leads to collagen failure that eventually leads to gross enlargement and eventual rupture of the AAA. The extensive transmural infiltration typically seen in noninfected AAAs suggests that inflammation and immune responses play a significant role in AAA formation.7 Interestingly, infectious causes have also been postulated: histochemical studies have demonstrated that as many as 55% of AAAs harbor Chlamydia pneumoniae.7 Thus, it appears that infected and noninfected AAAs may represent two ends of the same spectrum, with progression to infection depending on organism virulence and host resistance.
A number of important clinical differences have been noted between infected and uninfected aneurysms of the abdominal aorta. In contrast to chronic AAA, infected AAAs follow a more rapid course, have a predilection for the suprarenal aorta, and may be isolated to a small segment of an otherwise normal aorta. Recent studies have revealed a number of interesting findings related to the etiology of infected AAA. First, Buckmaster et al. have shown that elastolytic activity is derived from host leukocytes, not from the infectious organisms.8 However, infectious organisms play a central role in collagen degradation. Many bacterial isolates produce collagenases,9 and bacteria are capable of activating the collagenase promoter in macrophage-like cells.10 Furthermore, a variety of bacterial proteases activate MMP-1, MMP-8, and MMP-9.9 Therefore, infected AAAs appear be the consequence of bacterial proteases causing rapid collagen breakdown in a previously normal aorta.9 The collagenase activity may be relatively localized, leading to formation of a saccular AAA or pseudoaneurysm in an otherwise normal appearing vessel. Collagenase activity may also be intensive, which may explain the rapid course associated with infected AAAs. The reason infected AAAs are predisposed to the suprarenal aorta has not been elucidated.
Pathological findings of infected aortic aneurysms have been described by Hsu and Lin.11 Typical findings included aortic atherosclerosis, acute suppurative inflammation, neutrophil infiltration, and bacterial clumps. Two thirds of patients in their series showed acute inflammation superimposed on severe chronic atherosclerosis; the remainder showed atherosclerosis with chronic inflammation or pseudoaneurysms.
Anatomical Location
In their classic 1984 study, Brown and associates documented 243 infected aneurysms in 180 patients in the following distribution: 38% femoral artery, 31% abdominal aorta, 8% SMA, 7% brachial artery, 6% iliac artery, 5% carotid artery, 3% ulnar/radial arteries, 1% hepatic artery, less than 1% subclavian artery, and less than 1% popliteal artery.5 Notably, there were no cases involving the suprarenal or thoracoabdominal aorta in this series. More recent reports have documented a much higher prevalence of infected aortic aneurysms involving the segments proximal to the renal arteries,12–15 suggesting the aorta is the most frequent site of involvement.
Bacteriology
Approximately 75% of clinically infected aneurysms are associated with positive culture results.5 There have been significant shifts in the bacteriological patterns of infected aneurysms that have paralleled the changes in etiology. Prior to 1960, gram-positive organisms predominated, particularly Streptococcus pneumoniae, Streptococcus, and Staphylococcus aureus.3 More recent series have reported a higher prevalence of gram-negative organisms, paralleling the increasing number of arterial infections due to bacteremia, particularly Salmonella species.13,16 Gram-negative sepsis in elderly patients is a frequent clinical scenario in these circumstances.17 It appears that the bacteriological pattern is continuing to evolve: several reports have identified S. pneumoniae as an increasingly frequent cause of infected aortic aneurysms.14,18,19 The prevalence of organisms associated with opportunistic infections such as fungus and Mycobacterium species may also be on the rise owing to the increasing prevalence of chronic diseases associated with impaired immunity.
Salmonella infections deserve special emphasis. For uncertain reasons, Salmonella organisms have a tendency to infect the abdominal aorta. Up to 36% of all infected aneurysms of the aorta are due to Salmonella species; conversely, 65% of Salmonella vascular infections are localized to the aorta.20 Humans become infected by ingestion of contaminated food or water. Of the patients who develop Salmonella gastroenteritis, a small proportion will develop bacteremia that may result in extraintestinal seeding and infection. The interval between the onset of gastrointestinal symptoms and development of aortic infection may be several weeks.20 Diagnosis can be difficult because the signs and symptoms are nonspecific: more than half of patients have ruptured aneurysms before the diagnosis is made.20 Common isolates from aortic wall specimens have included Salmonella choleraesuis, Salmonella typhimurium, and Salmonella enteritidis. Salmonella aortitis is an extremely morbid condition that has historically been associated with mortality rates of 50% and a high rate of reinfection after revascularization.20 However, this trend appears to be reversing: in a more recent series, Hsu et al. documented an 11% mortality rate and showed that Salmonella was a predictor of survival compared to other microorganisms.21 Much of this effect may have been due to the fact that the Salmonella infected patients in this series were significantly younger than patients who died with other infections.
Syphilitic aneurysms, once a common cause of death due to aortic arch rupture, are now very rare. In a review of the literature on infected aortic aneurysms, Leon and Mills reported that syphilitic aneurysms occur most commonly in the ascending and arch aorta; they are uncommon below the sixth vertebral body.22 These aneurysms result from the intense inflammatory response associated with the treponemes that lodge in vasa vasorum.
Infected Aortic Aneurysms
The definition of aortic aneurysm infection has been confounded by the fact that 10% to 15% of patients with uninfected chronic AAA will have positive culture results from intraluminal thrombus removed at the time of aneurysm repair. Fortunately, positive culture results are not associated with an increased risk of late graft infection.23,24 However, this underscores the fact that the definition of infected AAA relies on other criteria such as operative findings (inflammation and purulence), clinical symptoms (fever, pain, leukocytosis), aneurysm architecture (saccular or localized), and positive aneurysm wall culture.13
Modern series of infected aortic aneurysms reveal that the majority of affected patients have comorbid conditions associated with immunosuppression such as diabetes, chronic renal failure, chronic steroid use, human immunodeficiency virus (HIV), or cancer.13,25 Nearly 50% of patients have had a recent documented infection such as pneumonia or urinary tract infection,13 and several reports have documented direct extension of vertebral osteomyelitis.14,26,27
Features
Infected aneurysms of the aorta may involve any segment from the ascending arch to the distal infrarenal area. In a series of 43 patients with infected aortic aneurysms treated at the Mayo Clinic over a 25-year period, Oderich et al. documented a wide distribution of lesions.13 No segment of the aorta was spared. As can be seen in Figure 59-1, 40% of infected aneurysms were localized to the infrarenal aorta, and the remaining lesions were almost evenly distributed in the juxta/pararenal, paravisceral, thoracoabdominal, and descending thoracic segments. A similar distribution has been observed by others.14,28,30
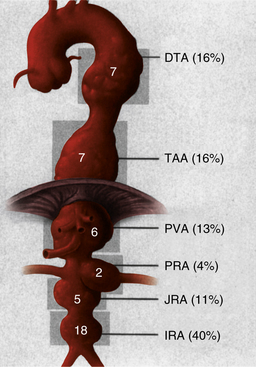
Figure 59-1 Distribution of infected aortic aneurysms in a series of 43 patients treated at the Mayo Clinic.
(From Oderich GS, Panneton JM, Bower TC, et al: Infected aortic aneurysms: aggressive presentation, complicated early outcome, but durable results. J Vasc Surg 34:900, 2001, with permission from the Society for Vascular Surgery and the American Association for Vascular Surgery.)13
The majority of aortic infections can be classified as bacterial aortitis.11 Most lesions are saccular and well localized in an otherwise normal-appearing aorta (Fig. 59-2). This appearance is highly suggestive of a ruptured aortic pseudoaneurysm, which is pathognomonic for aortic infection. In a minority of cases, infection of a preexisting aortic aneurysm may make diagnosis difficult. In these circumstances, suspicion of infection is completely reliant on clinical information.
Diagnosis
The majority of patients with infected aortic aneurysms are symptomatic. In the series by Oderich et al., 93% of patients had symptoms, the most common of which were fever, back pain, and leukocytosis (Table 59-1).13 Blood cultures are positive in approximately 75% of cases.13 The degree of symptomatology may be an important indicator of prognosis. Recent data suggest that the systemic inflammatory response syndrome (SIRS), a marker for sepsis, is associated with increased morbidity and mortality in patients with infected aortic aneurysms.14,29 Diagnostic criteria for SIRS include the presence of two or more of the following: body temperature above 38 °C or below 36 °C; heart rate over 90 beats/min; respiratory rate over 20 breaths/min or a partial pressure of carbon dioxide in arterial blood (PaCO2) less than 32 torr; and white blood cell (WBC) over 12,000 cells/mm3, less than 4000 cells/ mm3, or 10% immature (band) forms.29
Table 59-1 Clinical Presentation and Laboratory Findings in 43 Patients Treated at the Mayo Clinic for Infected Aortic Aneurysms
| No. Patients (%) | |
|---|---|
| Symptomatic | 40 (93) |
| Fever | 33 (77) |
| Pain (abdominal or back) | 28 (65) |
| Leukocyte count >12,000/mm3 | 23 (54) |
| Chills | 22 (51) |
| Sweats | 12 (28) |
| Enlarging aneurysm | 12 (28) |
| Nausea/vomiting or diarrhea | 10 (25) |
| Pulsatile mass | 7 (16) |
| Hemodynamic instability | 3 (7) |
Adapted from Oderich GS, Panneton JM, Bower TC, et al: Infected aortic aneurysms: aggressive presentation, complicated early outcome, but durable results. J Vasc Surg 34:900, 2001.13
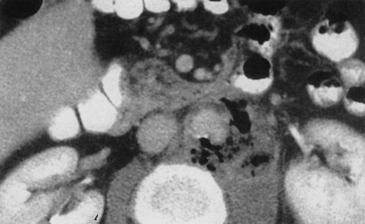
Diagnosis of infected aortic aneurysms relies on a number of imaging techniques. The presence of a saccular aneurysm in a patient with typical symptoms is pathognomonic, and the diagnosis is confirmed if blood cultures are positive. Gas bubbles and periaortic fat stranding on magnetic resonance imaging (MRI) or enhanced computed tomography (CT) scans are also diagnostic for infection (Fig. 59-3) but are not universally present. More subtle signs include periaortic stranding, proximity to abnormal fluid collections or nearby infections such as vertebral osteomyelitis, and rapid aneurysm expansion over several days.15 Indium-111-labeled WBC scans have been used in some patients, but the 80% sensitivity and specificity of this test suggest that it has limited usefulness. Angiography has been recommended to localize the infection and plan appropriate operative treatment in all patients. As an alternative, newer imaging techniques such as CT angiography (CTA) can be used to assess periaortic tissues, localize the infectious process, and evaluate vascular anatomy. This technique has the advantage of being minimally invasive compared to standard angiography, and it essentially combines two tests into one.
Treatment
Treatment of infected aortic aneurysms is surgical. Although treatment with broad-spectrum antibiotics may “sterilize” an infected aneurysm, the aneurysm still requires treatment because of the significant risk of rupture. In a series of 22 high-risk patients treated with antibiotics but without resection, Hsu et al. reported that 50% had in-hospital mortality, and 59% of those who survived to leave the hospital died of aneurysm rupture in late follow-up.30
The urgent nature of infected aortic aneurysms cannot be overstated because of the potential for rupture with exsanguination. In a recent series, nearly half of all patients with infected AAAs had already ruptured at the time of surgery, including free ruptures in 20%.31 Once the diagnosis is confirmed with appropriate imaging, preoperative preparation should be completed as rapidly as possible—preferably within 2 or 3 hours. Patients with hemodynamic instability should be transported immediately to the operating room. Stable patients should be admitted to an intensive care unit for rapid fluid repletion, institution of broad-spectrum intravenous antibiotics, and placement of appropriate monitoring devices. Blood should be typed and crossmatched, and at least four units of packed red blood cells should be available in the operating room.
Surgical treatment of infected aortic aneurysms depends on the location and extent of the infection. The most common operation is ligation and débridement of the infected arterial segment, with revascularization of the lower extremities using grafts brought through uninfected tissues remote from the infected site (extra-anatomical bypass).28,32 For example, infected aneurysms of the infrarenal aorta can be treated with ligation of the abdominal aorta distal to the renal arteries and revascularization of the lower extremities using axillofemoral bypass grafts. However, proximal aortic aneurysms involving the renal arteries, visceral aorta, or descending thoracic aorta are much more complicated. These aortic segments require direct revascularization to preserve blood flow to the kidneys, intraabdominal organs, and the spinal cord. In these cases, aortic débridement with direct revascularization of the affected segment within the infected bed (in situ reconstruction) is appropriate.
In situ reconstruction has been performed with prosthetic grafts, but the reinfection rate of up to 20% makes this option unattractive except in patients who are unstable at the time of operation.33,34 Prolonged administration of antibiotics is generally recommended in these patients.35 As an alternative, a rifampin-bonded gelatin-impregnated Dacron has been recommended in patients with arterial infections caused by susceptible organisms such as S. aureus36; however, these grafts are not effective against methicillin-resistant S. aureus or Escherichia coli infections.37 Additional alternatives include human allografts and autogenous vein grafts. The midterm results of cryopreserved human allografts are encouraging: Brown et al. reported a series of 52 patients with infected aortas or aortic grafts who underwent replacement with cryopreserved allografts.38 At 20 months follow-up, three patients had graft thrombosis or stenosis, and one developed a recurrent ilioenteric fistula; the remainder had no evidence of aneurysmal change or reinfection. The experience with autogenous superficial femoral-popliteal vein (SFPV) grafts is also encouraging. Several small series attest to the negligible reinfection rates and excellent durability associated with in situ SFPV reconstruction.14,39 The early enthusiasm for this option is buoyed by excellent experience using the SFPV to replace infected prosthetic grafts of the aorta (see later discussion).
The modern enthusiasm for endovascular therapy has extended to infected aortic aneurysms. Several small series have suggested that placement of endografts in combination with long-term antibiotics represent definitive treatment of infected aortic aneurysms with similar outcomes to open aortic repair.40,41 However, these data are countered by reports of a high rate of graft infection requiring removal, or extension of the aortic infection to more proximal segments.42,43 This appears to be a particular risk in patients with Salmonella infections. Therefore, we suggest that endovascular repair might be a good option to treat acute complications from infected aortic aneurysms such as aerodigestive fistulas. However, these grafts should not be considered definitive treatment, and additional surgical therapy is indicated after resolution of the acute problem.42
Infected Femoral Artery Aneurysms
Femoral Artery Infections Associated with Invasive Procedures
Compared to alternative approaches involving direct puncture of the axillary artery or the abdominal aorta, the transfemoral approach is associated with the lowest risk of complications. Modern results indicate that the overall arterial complication rate of transfemoral catheterizations is less than 1%; the risk of femoral artery infection is exceedingly rare.44,45 However, the increasing popularity of transcatheter techniques suggests that the absolute number of patients with catheter-related complications is likely to rise.
In an effort to reduce the incidence of pseudoaneurysms, many interventionalists use percutaneous closure devices to mechanically seal the arterial puncture site. Compared to manual compression, these devices have resulted in earlier mobilization and discharge of patients after arterial catheterization. However, these devices have been associated with a slightly higher risk of femoral artery infection. In a recent meta-analysis, Biancari et al. reported that the risk of access site infection is 0.6% after closure devices, compared to 0.2% without closure devices.46 Risk factors for infection include diabetes, obesity, therapeutic intervention, and groin hematoma.
Primary femoral artery infections present as pseudoaneurysms. Common findings include pain at the puncture site, fever with chills, and a pulsatile groin mass. The onset of symptoms may be delayed up to several weeks after the original puncture. Gram-positive organisms predominate, especially S. aureus, but gram-negative bacteria are common isolates. Treatment involves institution of broad-spectrum antibiotics, débridement of the infected arterial segment, removal of all closure device material, and excision of grossly infected adjacent tissues. Direct revascularization with autogenous saphenous vein has been associated with a high risk of reinfection and vein graft blowout.47 Alternatively, use of SFPV has been associated with excellent durability and resistance to infection in two small series.48,49 Simple ligation of the common femoral artery (CFA) may be preferable if the femoral bifurcation is uninvolved.50 If revascularization is necessary, the bypass should be routed through extra-anatomical tissues to avoid infected areas. A transobturator bypass is ideally suited to this situation.51
Femoral Artery Infections in Intravenous Drug Abusers
Approximately three fourths of all admissions for accidental intravenous drug injections involve the lower extremity, and the femoral artery is the most common site of involvement.52 Most patients present with a painful, pulsatile groin mass, often associated with overlying cellulitis. The most commonly cultured organism is S. aureus. Appropriate treatment involves ligation of the affected arterial segment to reduce the risk of hemorrhage and débridement of all grossly contaminated tissue to remove the septic focus. The advisability of subsequent revascularization remains controversial.
Avoidance of revascularization rarely leads to amputation. Earlier reports documented an 11% amputation rate when one artery was ligated and a 33% amputation rate after triple-vessel ligation.53 More recent reports suggest that the incidence of amputation is much lower. Ting and Cheng performed routine ligation in 34 infected femoral pseudoaneurysms, including 24 that involved the femoral bifurcation.54 The mean postoperative ankle-brachial index was .43 after triple ligation and .52 with single-vessel ligation. Although 88% of patients had some degree of intermittent claudication after discharge, there were no instances of delayed limb loss. Cheng and colleagues reported a similar rate of claudication after single- or triple-vessel ligation, but one patient (5%) required above-knee amputation.55 Mousavi et al. reported the results of femoral artery ligation for infected pseudoaneurysms in 134 illicit drug users. There were no amputations in this series.56
Modern consensus is that infected femoral aneurysms in drug addicts are best treated with ligation alone. Most patients will suffer some degree of claudication, but the risk of early and late amputation is low. Immediate revascularization should be limited to cases in which no Doppler signal is detected at the ankle after femoral artery ligation.57 In the vast majority, staged revascularization should be considered in patients with limiting claudication after the infection has been completely cleared. The known propensity for prosthetic graft infection from a remote injection site suggests that autogenous tissue is preferable in these circumstances. In the absence of usable saphenous vein, the SFPV represents an excellent alternative.48
Infected Aneurysms of the Superior Mesenteric Artery
The SMA is the third most common site of visceral aneurysms from all causes, but it is the most common site for infected aneurysms in the splanchnic circulation.58,59 Original studies from more than 20 years ago reported that approximately 60% of SMA aneurysms had an infectious etiology,60 but this proportion appears to be decreasing. More recent series have reported an infectious etiology in 5% to 33% of reported cases.58,59,61 Infected SMA aneurysms usually occur secondary to subacute bacterial endocarditis, and the most commonly isolated organism is nonhemolytic Streptococcus.58,62
Most infected SMA aneurysms occur in patients younger than 50 years; men and women are equally affected.58,60 Only 10% of patients are completely asymptomatic.58 Some degree of abdominal discomfort is present in two thirds, and up to half have a tender, mobile, pulsatile mass.60 Fever, nausea, vomiting, gastrointestinal hemorrhage, and jaundice may also be present.
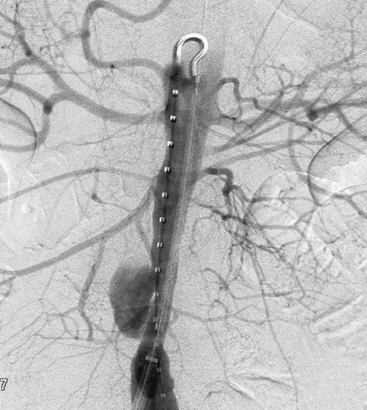
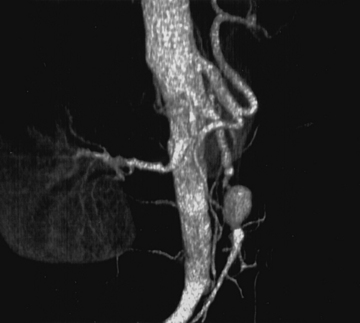
Superior mesenteric artery aneurysms tend to occur within 5 cm of the SMA origin, but any segment may be affected. Aneurysms may be suspected when vascular calcifications are seen on plain radiographs of the abdomen. Diagnosis should be confirmed with appropriate imaging studies that also localize the extent of the aneurysm such as standard mesenteric angiography or CTA (Fig. 59-4). Most infected SMA aneurysms are single, with variable involvement of visceral branches.
The natural history of infected SMA aneurysms is one of progression and eventual rupture; in fact, rupture has occurred at the time of presentation in 38% to 50% of patients.58 Reported mortality rate after rupture approaches 30%.61 Treatment includes transabdominal exploration and ligation of the arterial segments proximal and distal to the aneurysm. Complete excision is hazardous due to the proximity of the superior mesenteric vein and pancreas; therefore, débridement should be limited to exposed portions of the aneurysm wall and to the aneurysm sac contents. In the vast majority of cases, extensive mesenteric collateralization preserves bowel viability after SMA ligation. Therefore, ligation with resection of short segments of nonviable intestine is almost always appropriate. Direct revascularization is necessary in approximately 15% of cases. Bypass should be performed with autogenous tissue; we have found the SFPV to be an excellent alternative with superior patency compared to saphenous vein.63
Infected Carotid Artery Aneurysms
Infected aneurysms are rare in the extracranial carotid circulation. In a series of 67 carotid artery aneurysms treated over a 35-year period at the Texas Heart Institute, only one was infected.64 Most patients present with fever and a tender pulsatile neck mass. Medial deviation of the pseudoaneurysm may lead to clinical findings suggestive of a parapharyngeal mass. Before the antibiotic era, most carotid artery infections were the consequence of direct spread from pharyngeal infections. Most lesions are currently due to septicemia from bacterial endocarditis.
Gram-positive organisms, especially S. aureus and S. pyogenes, are common isolates from infected carotid aneurysms, but Salmonella infections have also been reported.65,66 Treatment involves ligation of all infected segments, even if this requires ligation of the internal and external carotid branches. Owing to the potential for graft disruption and exsanguinating hemorrhage, revascularization is rarely indicated. To prevent propagation of the internal carotid thrombus into the middle cerebral artery circulation, systemic anticoagulation with warfarin is recommended. Anticoagulation should theoretically be continued until the thrombus becomes stable, a period not longer than 6 weeks. Although most patients can be expected to tolerate internal carotid ligation without sequelae, temporary occlusion with a balloon catheter should be performed in the preoperative period. Patients who develop neurological deficits during balloon occlusion should be considered for prophylactic extracranial-intracranial bypass through remote uninfected tissues. As an alternative, some patients may benefit from hypertensive therapy combined with hypervolemia and hemodilution.66 To decrease the risk of exsanguination during carotid exposure, Wales et al. have advocated staged repair using a covered endovascular stent to temporarily exclude the pseudoaneurysm before proceeding to early definitive surgical management.67
Other Infected Aneurysms
Aside from femoral artery aneurysms, infected aneurysms of the lower extremity are exceedingly rare. The vast majority of infected popliteal artery aneurysms are a consequence of septic embolization from infective endocarditis.68 Mycotic aneurysms of the tibioperoneal trunk and tibial vessels have also been reported. Most infections involve gram-positive organisms such as Streptococcus, but Salmonella species have been recovered in a significant number of recently reported cases. The most common presentation is rupture, although thrombosis with foot ischemia has also been described. Treatment involves excision of the infected arterial segment and revascularization using autogenous bypass grafts.
Prosthetic Graft Infections
Risk Factors and Pathogenesis
The pathogenesis of graft infections is multifactorial and partly related to the site of implantation. Contamination prior to implantation due to failed sterilization techniques or breaks in packaging is thought to occur very infrequently. Likewise, gross breaks in sterile technique are rare. Most graft infections occur as a result of unrecognized bacterial contamination at the time of implantation. Exposure of the graft material to surrounding skin is a likely source; viable bacteria remain in the dermis of the skin despite antiseptic preparation.69 Graft contamination can also occur from remote infections such as cellulitis or pyelonephritis. Wet gangrene of a toe can increase the risk of infection in a prosthetic femoropopliteal bypass graft. Similarly, concurrent intraabdominal procedures such as cholecystectomy or appendectomy can expose an aortic graft to the patient’s enteric flora, thereby increasing the risk of graft infection.
A number of specific risk factors have been associated with aortic graft infections. Colonic ischemia following the repair of ruptured and nonruptured aortic aneurysms is associated with a high risk of graft infection due either to direct contamination or to hematogenous seeding from bacterial translocation.70 It has long been recognized that graft infections are more common after emergency repair of ruptured aortic aneurysms compared to elective operations.71 The emergent nature of ruptured AAA repair likely leads to inadvertent breaks in sterile techniques as the surgical team rushes to gain vascular control. In addition, patients presenting with either acute occlusion of the aorta or rupture of an aortic aneurysm are at high risk to develop postoperative SIRS. This SIRS response leads to an initial production of a proinflammatory cytokine response followed by a compensatory antiinflammatory cytokine response. This response renders the patient immunocompromised and at risk for nosocomial infections. Theoretically, this immunocompromised state may contribute to the increased risk of graft infection by hematogenous seeding during episodes of bacteremia.
Aortic grafts are uniquely prone to primary bacterial colonization at the time of aortic aneurysm repair. As already noted, many studies have demonstrated that the mural thrombus found in aneurysms is frequently colonized with bacteria. Macbeth et al. found that up to 43% of arterial walls were culture-positive for bacteria at the time of surgery.72 In this series, all of the aortic graft infections (.9%) occurred exclusively in patients with positive aortic wall cultures. The most common isolate was Staphylococcus epidermidis (71%) followed by Streptococcus species (13%) and other isolates (16%).72 Similarly, Buckels et al. in 1985 found that graft infection occurred more frequently in patients with positive cultures of aortic contents compared to those with negative cultures.73 More contemporary studies have failed to confirm an absolute association between positive aortic cultures and subsequent graft infections. Farkas et al. reported positive cultures in 37% of 500 aortic aneurysms.24 However, only one patient with a positive culture developed a graft infection. In contrast, 6 of 296 patients with negative cultures developed aortic graft infections during follow-up.24 Based on these observations, it is clear that colonization of the mural thrombus by bacteria plays, at most, a minor role in the pathogenesis of aortic graft infection. Results from more contemporary experiences may be due to the consistent use of perioperative antibiotic therapy.
Local and regional factors may also play a role in development of graft infections. Use of groin incisions (e.g., aortobifemoral bypass) can more than double the risk of graft infection compared to aortic grafts that remain completely intraabdominal (e.g., aortobiiliac bypass or aortic tube graft repairs). This may be related to the local environment of the groin, an area associated with one of the highest concentrations of Staphylococcus species found on the body.69 In addition, dissection in the groin disrupts abundant femoral lymphatics, leading to risk of lymph leak, groin wound breakdown, and direct graft contamination. Furthermore, the lymphatic system transports bacteria from distal sites of infection to the groin lymph nodes; opening these channels exposes the graft to potential contamination. Translocated bacteria have been demonstrated in animals and humans. In patients presenting with complex foot infections, positive lymphatic cultures at the time of amputation have been demonstrated in up to 20% of patients.74 Cultures of groin lymph nodes at the time of vascular reconstruction have revealed bacteria in 11%. However, these cultures do not correlate with subsequent groin wound infections.75
Graft material also plays a role in the pathogenesis of graft infections. The immune system responds to the foreign body by walling off the offending agent. The initial response is an acute inflammation, with influx of neutrophils followed by macrophages. These inflammatory cells produce cytokines and release proteases in an attempt to eliminate the foreign body. This initial response has a negative effect on bacterial survival; however, if the inoculum is large, some bacteria may survive. The graft interstices may offer a safe haven for bacteria and allow them to survive the initial inflammatory phase. After the acute inflammatory response, a reparative phase begins. This stage is characterized by fibroblasts depositing collagen in response to locally secreted cytokines. A resulting connective tissue barrier shields bacterium from detection and obliteration by immune competent cells. This results in a closed space for the bacteria to thrive and grow on exudative proteins existing in an acidic and ischemic environment. In the absence of infection, the reparative phase culminates in tissue ingrowth and incorporation of the graft. However, if bacterial colonization is present, the graft fails to incorporate, and chronic inflammation continues. Failure to incorporate may be due to fibroblast inhibition by the bacterial components found in the perigraft fluid.76 This results in the failure to obliterate the closed space around the graft and failure of incorporation. Bacteria are left to thrive in this closed space, eventually becoming an abscess. This can manifest as perigraft fluid that may express through incisions with sinus tract formation. In addition, the artery may be degraded at suture lines, resulting in pseudoaneurysm formation.
Aortic Graft Infection
Over the last 2 decades, refinements in diagnosis and operative management of aortic graft infections have resulted in improved mortality and morbidity. Mortality during the early experience of aortic graft infection approached 50%, and limb loss rates were as high as 75%. With refinement in technique, the respective mortality and limb loss rates have decreased to 20% or less in many series.77
Incidence
The exact incidence of graft infection following aortic reconstruction is not precisely known because most series are retrospective and suffer from lack of inclusive follow-up. The best estimates suggested that the incidence of graft infection following aortic reconstructions ranges from 1% to 5%.77–79 More contemporary data can be abstracted from the U.K. Small Aneurysm Trial, a randomized study of AAA repair in patients with small aneurysms. Although this study did not directly report the incidence of aortic graft infection, a number of graft-related complications were documented. Three patients in the trial had late aortic rupture following AAA repair, and four patients died following development of aortoenteric fistula. Most of these complications can be assumed to represent complications from aortic graft infection and represent 2% of the total patients.80 The true incidence of aortic graft infection remains inadequately described, but most authorities agree the incidence is quite low and certainly less than 2% in the contemporary experience.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree