In the randomized PLATINUM trial, the PROMUS Element platinum-chromium everolimus-eluting stent (PtCr-EES; Boston Scientific, Natick, Massachusetts) was noninferior to the XIENCE V cobalt-chromium everolimus-eluting stent (CoCr-EES; Boston Scientific and Abbott Vascular, Santa Clara, California) for the primary end point of 1-year target lesion failure. This study reports the 3-year outcomes. Patients (n = 1,530) with 1 or 2 de novo native coronary artery lesions (baseline vessel diameter ≥2.50 mm to ≤4.25 mm and length ≤24 mm) were randomized 1:1 to PtCr-EES versus CoCr-EES. Three-year follow-up was available in 93.9% (703 of 749) of patients with CoCr-EES and 96.7% (733 of 758) of patients with PtCr-EES. Comparing CoCr-EES with PtCr-EES, 3-year rates of death (4.3% vs 3.7%, hazard ratio [HR] 0.88, 95% confidence interval [CI] 0.52 to 1.48, p = 0.62), cardiac death (1.9% vs 1.2%, HR 0.63, 95% CI 0.27 to 1.45, p = 0.27), myocardial infarction (2.5% vs 2.3%, HR 0.92, 95% CI 0.48 to 1.79, p = 0.81), ischemia-driven target lesion revascularization (4.9% vs 3.5%, HR 0.72, 95% CI 0.43 to 1.20, p = 0.21), and Academic Research Consortium definite or probable stent thrombosis (0.5% vs 0.7%, HR 1.23, 95% CI 0.33 to 4.57, p = 0.76) were not significantly different. In conclusion, 3-year results of the PLATINUM randomized, controlled, clinical trial demonstrate comparable safety and efficacy outcomes of the PROMUS Element PtCr-EES and the XIENCE V CoCr-EES.
Everolimus-eluting stents have become the most commonly used drug-eluting stents (DES), in part because of data from clinical trials showing significantly reduced rates of revascularization, myocardial infarction (MI), and stent thrombosis with the PROMUS or XIENCE V everolimus-eluting, cobalt-chromium stent (Boston Scientific, Natick, Massachusetts and Abbott Vascular, Santa Clara, California) compared with early generation paclitaxel-eluting stents. The PROMUS Element platinum-chromium everolimus-eluting stent (PtCr-EES) uses the same drug and polymer formulation as PROMUS or XIENCE V but combines them with a novel metal alloy and stent design intended to improve deliverability and conformability, increase radial strength and radiopacity, and reduce recoil compared with previous generation DES. One-year data from the PLATINUM clinical trial demonstrated that the PtCr-EES was noninferior (p = 0.001) to the XIENCE V cobalt-chromium everolimus-eluting stent (CoCr-EES) with 1-year target lesion failure (TLF) rates of 2.9% (CoCr-EES) versus 3.4% (PtCr-EES) and no significant differences between stent types in the rates of death, MI, ischemia-driven revascularization, or definite or probable stent thrombosis. Given the potential for late restenosis and stent thrombosis, long-term follow-up data are important and have not yet been published in the context of a randomized controlled trial. The present analysis presents the 3-year follow-up of the PLATINUM randomized, controlled, clinical trial, which compared the PtCr-EES and CoCr-EES for the treatment of de novo atherosclerotic coronary artery lesions.
Methods
The PtCr-EES (manufactured as PROMUS Element by Boston Scientific) incorporates a novel, thin-strut, platinum-enriched metal alloy platform coated with the same durable, biocompatible, inert fluorocopolymer and antiproliferative agent as the comparator CoCr-EES (manufactured as XIENCE V by Abbott Vascular; also distributed as PROMUS by Boston Scientific). Release kinetics, arterial tissue levels, and vascular responses of the 2 devices are similar. The PtCr-EES stent has been extensively studied in preclinical assessments and has also been shown to have low late loss and incomplete apposition rates in angiographic and intravascular ultrasound analyses in humans.
Detailed methods have been previously described. Briefly, patients with stable or unstable angina pectoris or documented silent ischemia and 1 or 2 de novo native coronary artery lesions (baseline vessel diameter ≥2.50 mm to ≤4.25 mm, length ≤24 mm, and diameter stenosis ≥50% to <100% with Thrombolysis In Myocardial Infarction flow 2 or 3 by visual estimate) were eligible for inclusion. A total of 1,530 patients were randomized 1:1 to PtCr-EES versus CoCr-EES at 132 clinical sites. Randomization was stratified by the presence or absence of medically treated diabetes mellitus, the intent to treat 1 versus 2 target lesions, and study site. Exclusion criteria included acute or recent MI, left ventricular ejection fraction <30%, left main or ostial location, significant bifurcation disease, chronic total occlusion, and target vessel thrombus. Periprocedural use of unfractionated heparin (or alternatives such as enoxaparin or bivalirudin) was required per protocol; glycoprotein IIb/IIIa inhibitor use was left to investigator discretion. Daily aspirin was required indefinitely after stenting. Oral clopidogrel 75 mg/day was required for at least 6 months after stent placement in all patients and for at least 12 months in those not at high risk of bleeding. Ticlopidine or prasugrel (in accordance with approved country-specific labeling) were allowed as alternatives to clopidogrel.
Follow-up was scheduled for 1, 6, 12, and 18 months and annually from 2 to 5 years. Patients who did not receive either study stent were only followed to the first year after index procedure. Repeat angiographic follow-up was performed only for clinical indications.
The study was approved by the institutional review board or ethics committee at each participating center, and all eligible patients provided written informed consent. The study is registered with www.clinicaltrials.gov (NCT00823212).
All case report form data were 100% verified by on-site study monitors. A blinded independent clinical events committee adjudicated all death, MI, target vessel revascularization (TVR), and stent thrombosis events. All reported and adjudicated adverse events were regularly evaluated by an independent data safety and monitoring committee. Angiographic data were analyzed by an independent core angiographic laboratory (Beth Israel Deaconess Medical Center, Boston, Massachusetts).
Detailed definitions have been previously described. TLF was defined as the composite of cardiac death related to the target vessel, MI related to the target vessel, or ischemia-driven target lesion revascularization (TLR). MI was defined as (1) the development of new Q waves in ≥2 leads of duration ≥0.04 seconds with creatine kinase-MB or troponin levels elevated above normal or (2) in the absence of new Q waves, elevation of total creatine kinase levels >3× normal (peri–percutaneous coronary intervention) or >2× normal (spontaneous) with elevated creatine kinase-MB or troponin >3× normal (peri–percutaneous coronary intervention) or >2× normal (spontaneous). Elevated troponin levels also required at least one of the following: (1) electrocardiographic changes indicative of new ischemia, (2) imaging evidence of new loss of viable myocardium, or (3) new regional wall motion abnormality to meet the criteria of MI. Similar criteria were required for the diagnosis of MI after coronary artery bypass grafting, with a creatine kinase-MB or troponin threshold of >5× normal. Ischemia-driven TLR or TVR was defined as revascularization of the target lesion or vessel with ≥50% stenosis by quantitative coronary angiography if associated with clinical or functional ischemia or stenosis ≥70% by quantitative coronary angiography without documented ischemia. Cardiac death included death due to any of the following causes: acute MI, cardiac perforation and/or pericardial tamponade, arrhythmia or conduction abnormality, cerebrovascular attack at hospital discharge or cerebrovascular attack suspected of being related to the procedure, death due to a complication of the procedure, or any death in which a cardiac cause could not be excluded. Stent thrombosis was defined according to the definite or probable Academic Research Consortium criteria.
Baseline, lesion, and procedural data were analyzed in the intention-to-treat analysis set (including all patients who underwent randomization, regardless of treatment actually received). Three-year follow-up data were analyzed in patients who received either study stent (n = 1,507), as patients who did not receive a study stent were only monitored for 1 year. Data are presented as mean ± SD or n/N (%). Treatment groups were compared with a 2-sided chi-square or Fisher’s exact test for categorical variables and Student t test for continuous variables. Three-year event rates are reported as time-to-event estimates using the Kaplan-Meier product limit method. Landmark analyses of TLF, TLR, and all-cause death or MI before 1 year and in the follow-up period from 1 to 3 years were also conducted. Patients who did not have at least 366 days of follow-up were excluded from the 1 to 3-year landmark analyses. For any patient experiencing events in both the 0 to 1-year and 1 to 3-year intervals, only the earliest event in each time interval contributed to the 0 to 1-year and 1 to 3-year rates, respectively. Hazard ratio and 95% confidence interval were derived from Cox partial likelihood method. p Values are from the log-rank test comparing the time-to-event curves with those 3 years after procedure. Interaction p values were calculated by a chi-square test of the treatment-by-subgroup interaction in a logistic regression model. All statistical analyses were conducted using SAS software, version 8.2 or above (SAS Institute Inc., Cary, North Carolina).
Results
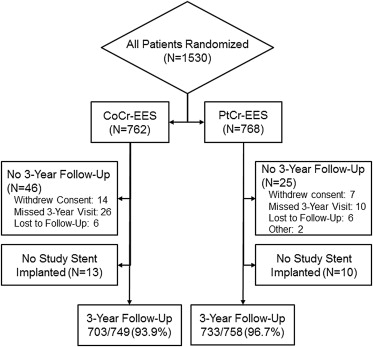
Three-year follow-up was available in 93.9% (703 of 749) of patients with CoCr-EES and 96.7% (733 of 758) of patients with PtCr-EES ( Figure 1 ). As reported previously, baseline demographics and patient medical history were well matched between the randomized study groups ( Table 1 ). Maximum dilation pressure was significantly higher in the PtCr-EES group. This might be due to the differences in nominal inflation pressures between the 2 stent platforms (9 atm for CoCr-EES and 12 atm for PtCr-EES). The difference might also result from the greater radiopacity of the PtCr-EES in that operators may be better able to detect stent underexpansion or malapposition and may have used higher postdilation pressures in an attempt to optimize stent deployment. There was a significantly higher rate of unplanned (bailout) stenting in the CoCr-EES group (9.8%) compared with the PtCr-EES group (5.9%; p = 0.004), due primarily to an increased incidence of inadequate lesion coverage (geographic miss; 3.4% vs 1.4%, p = 0.01), with a significantly greater number of stents per target lesion ( Table 1 ). No other procedural differences were observed.
| Variable | CoCr-EES (n = 762 Patients and 841 Lesions) | PtCr-EES (n = 768 Patients and 853 Lesions) | RR (95% CI) | p Value |
|---|---|---|---|---|
| Demographic features | ||||
| Age (yrs) | 63.1 ± 10.3 (762) | 64.0 ± 10.3 (768) | — | — |
| Men | 542/762 (71.1) | 550/768 (71.6) | 1.01 (0.94–1.07) | — |
| Hypertension ∗ | 558/762 (73.2) | 544/767 (70.9) | 0.97 (0.91–1.03) | — |
| Hyperlipidemia ∗ | 579/760 (76.2) | 598/765 (78.2) | 1.03 (0.97–1.08) | — |
| Diabetes mellitus ∗ | 191/762 (25.1) | 169/768 (22.0) | 0.88 (0.73–1.05) | — |
| Insulin treated | 48/762 (6.3) | 59/768 (7.7) | 1.22 (0.84–1.76) | — |
| Current smoker | 131/741 (17.7) | 158/751 (21.0) | 1.19 (0.97–1.47) | — |
| Previous MI | 160/760 (21.1) | 160/761 (21.0) | 1.00 (0.82–1.21) | — |
| Unstable angina | 188/762 (24.7) | 185/767 (24.1) | 0.98 (0.82–1.17) | — |
| Number of target lesions, mean | 1.10 ± 0.31 (762) | 1.11 ± 0.31 (768) | — | — |
| 1 | 684/762 (89.8) | 683/768 (88.9) | 0.99 (0.96–1.03) | — |
| 2 | 77/762 (10.1) | 85/768 (11.1) | 1.10 (0.82–1.47) | — |
| 3 | 1/762 (0.1) | 0/768 (0.0) | 0 | — |
| Target lesion measures | ||||
| Reference vessel diameter (mm) | 2.63 ± 0.49 | 2.67 ± 0.49 | — | — |
| Minimal lumen diameter (mm) | 0.74 ± 0.34 | 0.75 ± 0.35 | — | — |
| Diameter stenosis (%) | 71.9 ± 11.5 | 71.8 ± 11.5 | — | — |
| Lesion length (mm) | 12.5 ± 5.5 | 13.0 ± 5.7 | — | — |
| Procedural variables | ||||
| No. of stents per patient | 1.20 ± 0.48 | 1.16 ± 0.44 | — | 0.16 |
| No. of stents per target lesion | 1.08 ± 0.35 | 1.05 ± 0.26 | — | 0.01 |
| Max stent diameter/lesion (mm) | 3.05 ± 0.44 | 3.09 ± 0.45 | — | 0.07 |
| Total stent length/lesion (mm) | 19.7 ± 8.9 | 20.5 ± 7.0 | — | 0.06 |
| Poststent dilatation performed | 415/841 (49.3) | 425/853 (49.8) | 1.01 (0.92–1.11) | 0.84 |
| Maximum dilation pressure (atm) | 15.9 ± 3.2 | 16.3 ± 3.1 | — | 0.002 |
| Fluoroscopy time (minutes) | 11.3 ± 10.1 | 12.2 ± 11.8 | — | 0.10 |
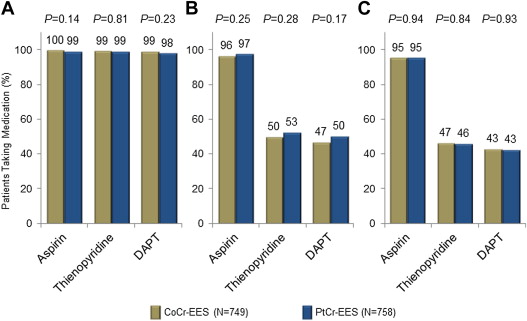
There were no statistically significant differences in dual antiplatelet medication usage between study groups through 3-year follow-up. At 3 years, 95.2% of patients treated with CoCr-ESS and 95.3% of patients treated with PtCr-EES were taking aspirin; similarly, 42.9% of patients treated with CoCr-EES and 42.7% of patients treated with PtCr-EES were taking aspirin plus a thienopyridine. Antiplatelet therapy rates at discharge, 2 years, and 3 years are shown in Figure 2 .
As listed in Table 2 , there were no statistically significant differences in clinical event rates at 3 years between the CoCr-EES and PtCr-EES groups, with the exception of a significantly lower rate of cardiac death unrelated to the target vessel in the PtCr-EES group versus the CoCr-EES group (p = 0.02). Rates of all death, cardiac death, MI, and stent thrombosis were not significantly different between groups. Kaplan-Meier analysis of the 3-year rate of all death or MI demonstrated no significant difference, with largely overlapping curves ( Figure 3 ). The rate of ischemia-driven TLR by percutaneous intervention was numerically lower in the PtCr-EES group (2.7%) compared with the CoCr-EES group (4.5%), but this difference did not reach statistical significance (p = 0.08). Three-year Kaplan-Meier curves of TLF ( Figure 4 ) and TLR ( Figure 5 ) demonstrated no significant differences. Similarly, in post hoc landmark analyses of TLF ( Figure 4 ) and TLR ( Figure 5 ), there was no significant difference between groups from 1 to 3 years. Kaplan-Meier analysis of stent thrombosis showed very similar event rates between groups at 3 years ( Figure 6 ). There were no cases of longitudinal stent compression in either arm of the PLATINUM trial through 3 years.
| Variable | CoCr-EES (n = 749) | PtCr-EES (n = 758) | HR (95% CI) | p Value |
|---|---|---|---|---|
| TLF | 7.1 (51) | 5.9 (44) | 0.84 (0.56–1.26) | 0.40 |
| All-cause death, MI, TVR | 12.7 (91) | 11.4 (84) | 0.90 (0.67–1.21) | 0.48 |
| All death | 4.3 (30) | 3.7 (27) | 0.88 (0.52–1.48) | 0.62 |
| Cardiac death | 1.9 (14) | 1.2 (9) | 0.63 (0.27–1.45) | 0.27 |
| Related to the TV | 1.3 (9) | 1.2 (9) | 0.98 (0.39–2.46) | 0.96 |
| Not related to the TV | 0.7 (5) | 0.0 (0) | 0 | 0.02 |
| Noncardiac death | 2.4 (16) | 2.6 (18) | 1.10 (0.56–2.15) | 0.79 |
| MI | 2.5 (18) | 2.3 (17) | 0.92 (0.48–1.79) | 0.81 |
| Related to the TV | 2.1 (15) | 1.5 (11) | 0.72 (0.33–1.56) | 0.40 |
| Not related to the TV | 0.4 (3) | 0.8 (6) | 1.94 (0.49–7.77) | 0.34 |
| Q-wave MI | 0.8 (6) | 0.4 (3) | 0.49 (0.12–1.95) | 0.30 |
| Non−Q-wave MI | 1.9 (14) | 1.9 (14) | 0.98 (0.47–2.05) | 0.95 |
| TVR, overall | 7.8 (55) | 6.4 (47) | 0.83 (0.56–1.22) | 0.34 |
| TLR, overall | 4.9 (35) | 3.5 (26) | 0.72 (0.43–1.20) | 0.21 |
| TLR, PCI | 4.5 (32) | 2.7 (20) | 0.61 (0.35–1.06) | 0.08 |
| TLR, CABG | 0.6 (4) | 0.8 (6) | 1.46 (0.41–5.19) | 0.55 |
| Non-TLR TVR, overall | 3.3 (23) | 3.3 (24) | 1.02 (0.57–1.80) | 0.96 |
| Cardiac death or MI | 4.1 (30) | 3.5 (26) | 0.85 (0.50–1.43) | 0.53 |
| Stent thrombosis (ARC) | 0.5 (4) | 0.7 (5) | 1.23 (0.33–4.57) | 0.76 |
| Definite | 0.5 (4) | 0.7 (5) | 1.23 (0.33–4.57) | 0.76 |
| Probable | 0 (0) | 0 (0) | — | — |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


