David A. Morrow, William E. Boden
Stable Ischemic Heart Disease
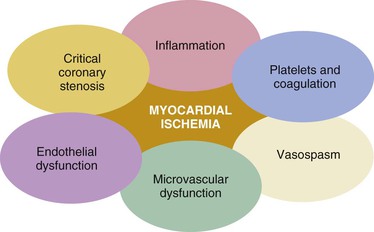
Stable ischemic heart disease (SIHD) is most commonly caused by atheromatous plaque that obstructs or gradually narrows one or more of the epicardial coronary arteries. The pathogenesis of atherosclerosis is described in Chapter 41. However, other contributors, such as endothelial dysfunction, microvascular disease, and vasospasm, may also exist alone or in combination with coronary atherosclerosis and may be the dominant cause of myocardial ischemia in some patients (Fig. 54-1).1,2 Thus the concept that ischemic heart disease (IHD) is synonymous with obstructive coronary atherosclerosis represents an overly simplified view.1,2
Factors that predispose to coronary atherosclerosis are discussed in Chapter 42, control of coronary blood flow in Chapter 49, ST-segment elevation myocardial infarction (MI) in Chapters 51 and 52, non–ST-segment elevation acute coronary syndromes (ACSs) in Chapter 53, and sudden cardiac death, another significant consequence of coronary artery disease (CAD), in Chapter 39.
The clinical findings in patients with IHD are highly variable. Chest discomfort is usually the predominant symptom in chronic (stable) angina, unstable angina, Prinzmetal (variant) angina, microvascular angina, and acute MI. However, manifestations of IHD also occur in which chest discomfort is absent or not prominent, such as asymptomatic (silent) myocardial ischemia, heart failure, cardiac arrhythmias, and sudden death. Notably, there may be features of atypical angina or anginal equivalents that characterize IHD, such as midepigastric discomfort, effort intolerance, dyspnea, and excessive fatigue, which are observed more frequently in women, older adults, and individuals with diabetes. Obstructive CAD also has nonatherosclerotic causes, including congenital abnormalities of the coronary vessels, myocardial bridging, coronary arteritis in association with the systemic vasculitides, and radiation-induced CAD. Myocardial ischemia and angina pectoris may also occur in the setting of extreme myocardial O2 demand with or without underlying obstructive CAD, as in the case of aortic valve disease (see Chapter 63), hypertrophic cardiomyopathy (see Chapter 66), and idiopathic dilated cardiomyopathy (see Chapter 65).
Magnitude of the Problem
The importance of IHD in contemporary society is attested to by the almost epidemic number of persons afflicted (see Chapter 1). It is estimated that 15,400,000 Americans have IHD, 7,800,000 of whom have angina pectoris and 7,600,000 have had MI.3 Based on data from the Framingham Heart Study, the lifetime risk for the development of symptomatic CAD after 40 years of age is 49% for men and 32% for women. In 2010, IHD accounted for 48% of all deaths caused by cardiovascular disease and was the single most frequent cause of death in American men and women; it resulted in more than one in six deaths in the United States.3 The economic cost of IHD is formidable, and in the United States in 2010 it was estimated to be $204.4 billion.3 Despite a steady decline in age-specific mortality from CAD over the past several decades, IHD is now the leading cause of death worldwide, and it is expected that the rate of CAD will only accelerate in the coming decades with the burden shifting progressively to lower socioeconomic groups; contributory factors include aging of the population, increases in the worldwide prevalence of obesity and type 2 diabetes, and a rise in cardiovascular risk factors in younger generations. The World Health Organization has estimated that by 2020, the global number of deaths from CAD will have risen from 7.6 million in 2005 to 11.1 million (see Chapter 1).4
Stable Angina Pectoris
Clinical Manifestations
Characteristics of Angina (See Chapter 11)
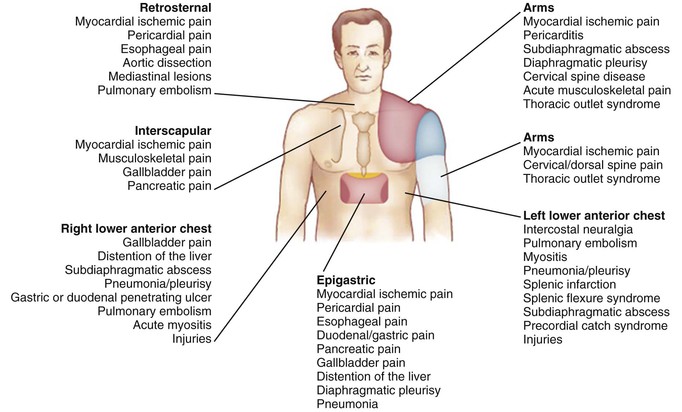
Angina pectoris is a discomfort in the chest or adjacent areas caused by myocardial ischemia. It is usually precipitated by exertion and is associated with a disturbance in myocardial function. Acute MI, which is generally associated with prolonged, severe pain occurring at rest (see Chapter 51), and unstable angina, which is characterized by an accelerated pattern of increasing frequency and tempo of angina or angina at rest (see Chapter 53), are discussed separately. Heberden’s initial description of angina as conveying a sense of “strangling and anxiety” is still remarkably pertinent. Other adjectives frequently used to describe this distress include constricting, suffocating, crushing, heavy, and squeezing. In other patients, the quality of the sensation is more vague and described as a mild pressure-like discomfort, tightness, an uncomfortable numbness, or a burning sensation. The site of the discomfort is usually retrosternal, but radiation is common and generally occurs down the ulnar surface of the left arm; the right arm and the outer surfaces of both arms may also be involved (Fig. e54-1![]() ). Epigastric discomfort alone or in association with chest pressure may occur and can masquerade as indigestion. Anginal discomfort above the mandible or below the epigastrium is rare. Anginal equivalents (i.e., symptoms of myocardial ischemia other than angina), such as dyspnea, faintness, fatigue, and eructations, are common, particularly in women and older adults. A history of abnormal exertional dyspnea may be an indicator of IHD even when angina is absent or no evidence of CAD can be found on the electrocardiogram (ECG). Nocturnal angina may be a manifestation of unstable angina but should also raise suspicion of sleep apnea (see Chapter 75). Postprandial angina, presumably caused by redistribution of coronary blood flow, may be a marker of severe CAD.
). Epigastric discomfort alone or in association with chest pressure may occur and can masquerade as indigestion. Anginal discomfort above the mandible or below the epigastrium is rare. Anginal equivalents (i.e., symptoms of myocardial ischemia other than angina), such as dyspnea, faintness, fatigue, and eructations, are common, particularly in women and older adults. A history of abnormal exertional dyspnea may be an indicator of IHD even when angina is absent or no evidence of CAD can be found on the electrocardiogram (ECG). Nocturnal angina may be a manifestation of unstable angina but should also raise suspicion of sleep apnea (see Chapter 75). Postprandial angina, presumably caused by redistribution of coronary blood flow, may be a marker of severe CAD.
The typical episode of angina pectoris usually begins gradually and reaches its maximum intensity over a period of minutes before dissipating. It is unusual for angina pectoris to reach its maximum severity within seconds, and it is characteristic that patients with angina generally prefer to rest, sit, or stop walking during episodes. Chest discomfort while walking in the cold or uphill is suggestive of angina. Features suggesting the absence of angina pectoris include pleuritic pain, chest pain localized with the tip of one finger, pain reproduced by movement or palpation of the chest wall or arms, and constant pain lasting many hours or, alternatively, very brief episodes of pain lasting seconds. Pain radiating into the lower extremities is also a highly unusual manifestation of angina pectoris.
Typical angina pectoris is relieved within minutes by rest or the use of short-acting nitroglycerin. Response to the latter is often a useful diagnostic tool, although it should be remembered that esophageal pain and other syndromes may also respond to nitroglycerin. A delay of more than 5 to 10 minutes before relief is obtained with rest and nitroglycerin suggests that the symptoms are either not caused by ischemia or are caused by severe ischemia, as with acute MI or unstable angina. The phenomenon of first-effort or warm-up angina is used to describe the ability of some patients in whom angina develops with exertion to continue subsequently at the same or even greater level of exertion without symptoms after an intervening period of rest. This attenuation of myocardial ischemia observed with repeated exertion has been postulated to be caused by ischemic preconditioning (see Chapter 49) and appears to require preceding ischemia of at least moderate intensity to induce the warm-up angina phenomenon.
Grading of Angina Pectoris
A system of grading the severity of angina pectoris proposed by the Canadian Cardiovascular Society (CCS) has gained widespread acceptance (see Table 11-1).5 The system is a modification of the New York Heart Association (NYHA) functional classification but allows patients to be categorized in more specific terms. Other grading systems include a specific activity scale developed by Goldman and associates and an anginal score developed by Califf and colleagues.6 The Goldman scale is based on the metabolic cost of specific activities and appears to be valid when used by both physicians and nonphysicians. The anginal score of Califf and coworkers integrates the clinical features and tempo of angina together with ST and T wave changes on the ECG and offers independent prognostic information beyond that provided by age, sex, left ventricular (LV) function, and coronary angiographic anatomy. A limitation of all these grading systems is their dependence on accurate patient observation and patients’ widely varying tolerance of symptoms. Functional estimates based on the CCS criteria have shown a reproducibility of only 73% and do not correlate well with objective measures of exercise performance.
Differential Diagnosis of Chest Pain
Esophageal Disorders
Common disorders that may simulate or coexist with angina pectoris are gastroesophageal reflux and disorders of esophageal motility, including diffuse spasm and nutcracker esophagus. To compound the difficulty in distinguishing between angina and esophageal pain, both may be relieved by nitroglycerin. However, esophageal pain is often relieved by milk, antacids, foods, or occasionally, warm liquids.
Esophageal Motility Disorders
Esophageal motility disorders are not uncommon in patients with retrosternal chest pain of unclear cause and should be specifically excluded or confirmed, if possible. In addition to chest pain, most such patients have dysphagia. Both IHD and esophageal disease are common clinical entities that may coexist. Diagnostic evaluation for an esophageal disorder may be indicated in patients with IHD who have a poor symptomatic response to antianginal therapy in the absence of documented severe ischemia.
Biliary Colic
Although visceral symptoms are commonly associated with myocardial ischemia (particularly acute inferior MI; see Chapter 51), cholecystitis and related hepatobiliary disorders may also mimic ischemia and should always be considered in patients with atypical chest discomfort, particularly those with diabetes. The pain is steady, usually lasts 2 to 4 hours, and subsides spontaneously, without any symptoms between attacks. It is generally most intense in the right upper abdominal area but may also be felt in the epigastrium or precordium. This discomfort is often referred to the scapula, may radiate around the costal margin to the back, or may in rare cases be felt in the shoulder and suggest diaphragmatic irritation.
Costochondritis
In 1921 Tietze first described a syndrome of local pain and tenderness, generally limited to the anterior chest wall and associated with swelling of costal cartilage. The full-blown Tietze syndrome (i.e., pain associated with tender swelling of the costochondral junctions) is uncommon, whereas costochondritis causing tenderness of the costochondral junctions (without swelling) is relatively common. Pain on palpation of these joints is usually well localized and is a useful clinical sign, although deep palpation may elicit pain in the absence of costochondritis. Local pressure should be applied routinely to the anterior chest wall during examination of a patient with suspected angina pectoris. Although palpation of the chest wall often reproduces pain in patients with various musculoskeletal conditions, it should be appreciated that chest wall tenderness may also be associated with and does not exclude symptomatic CAD.
Other Musculoskeletal Disorders
Cervical radiculitis may be confused with angina. This condition may occur as a constant ache and sometimes results in a sensory deficit. The pain may be related to motion of the neck, just as motion of the shoulder triggers attacks of pain from bursitis. Occasionally, pain mimicking angina can be caused by compression of the brachial plexus by the cervical ribs, and tendinitis or bursitis involving the left shoulder may also cause angina-like pain. Physical examination may also detect pain brought about by movement of an arthritic shoulder or a calcified shoulder tendon.
Other Causes of Angina-Like Pain
Severe pulmonary hypertension may be associated with exertional chest pain with the characteristics of angina pectoris, and indeed, this pain is thought to be caused by right ventricular ischemia that develops during exertion (see Chapter 74). Other associated symptoms include exertional dyspnea, dizziness, and syncope. Related findings on physical examination, such as a parasternal lift, a palpable and loud pulmonary component of the second sound, and right ventricular hypertrophy on the ECG, are usually readily recognized.
Pulmonary embolism is initially characterized by dyspnea as the cardinal symptom, but chest pain may also be present (see Chapter 73). Pleuritic pain suggests pulmonary infarction, and a history of exacerbation of the pain with inspiration, along with a pleural friction rub, if present, helps distinguish it from angina pectoris.
The pain of acute pericarditis (see Chapter 71) may at times be difficult to distinguish from angina pectoris. However, pericarditis tends to occur in younger patients, and the diagnosis depends on the combination of chest pain not relieved by rest or nitroglycerin, exacerbation by movement or deep inspiration, and lying flat; a pericardial friction rub, which may be evanescent; and changes on the ECG (notably PR-segment depression and/or diffuse ST-T wave abnormalities).
The classic symptom of aortic dissection is a severe, often sharp pain that radiates to the back (see Chapter 57).
Physical Examination
Many patients with SIHD have normal findings on physical examination, and thus the single best clue to the diagnosis of angina is the clinical history. Nonetheless, careful examination may reveal the presence or evidence of risk factors for coronary atherosclerosis or the consequences of myocardial ischemia (see Chapter 11).
Pathophysiology
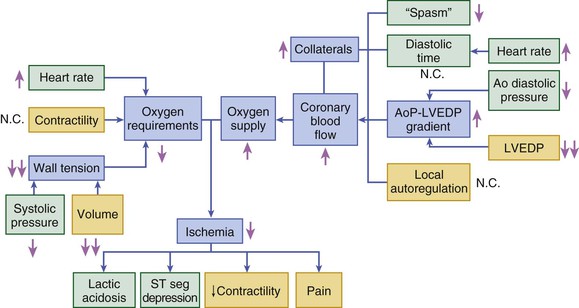
Angina pectoris results from myocardial ischemia, which is caused by an imbalance between myocardial O2 requirements and myocardial O2 supply. The former may be elevated by increases in heart rate, LV wall stress, and contractility (see Chapter 49); the latter is determined by coronary blood flow and coronary arterial O2 content (Fig. 54-2). The clinical precipitants and manifestations of supply-demand imbalance are discussed in this section. The pathobiology of atherosclerosis is discussed in Chapter 41. See Chest Pain with a Normal Coronary Arteriogram in this chapter and Chapter 49 for discussion of other abnormalities in coronary function and contributors to myocardial ischemia in the absence of critical coronary obstruction.
Angina Caused by Increased Myocardial O2 Requirements
In this condition, sometimes termed demand angina, the myocardial O2 requirement increases in the presence of a constant and usually restricted O2 supply. The increased requirement commonly stems from release of norepinephrine by adrenergic nerve endings in the heart and vascular bed, a physiologic response to exertion, emotion, or mental stress. Of great importance to the myocardial O2 requirement is the rate at which any task is carried out. Hurrying is particularly likely to precipitate angina, as are efforts involving motion of the hands over the head. Mental and emotional stress may also precipitate angina, presumably by increased hemodynamic and catecholamine responses to stress, increased adrenergic tone, and reduced vagal activity. The combination of physical exertion and emotion in association with sexual activity may precipitate angina pectoris. Anger may produce constriction of coronary arteries with preexisting narrowing, without necessarily affecting O2 demand. Other precipitants of angina include physical exertion after a heavy meal and the excessive metabolic demands imposed by chills, fever, thyrotoxicosis, tachycardia from any cause, exposure to the cold, and hypoglycemia.
Angina Caused by Transiently Decreased O2 Supply
Evidence has suggested that not only unstable angina but also chronic stable angina may be caused by transient reductions in O2 supply, a condition sometimes termed supply angina, as a consequence of coronary vasoconstriction that results in dynamic stenosis. In the presence of organic stenoses, platelet thrombi and leukocytes may elaborate vasoconstrictor substances such as serotonin and thromboxane A2. In addition, endothelial damage in atherosclerotic coronary arteries decreases production of vasodilator substances, which may result in an abnormal vasoconstrictor response to exercise and other stimuli. A variable threshold of myocardial ischemia in patients with chronic stable angina may be caused by dynamic changes in peristenotic smooth muscle tone and also by constriction of arteries distal to the stenosis. Patients with resulting “variable-threshold angina” may have good days, when they are capable of substantial physical activity, as well as bad days, when even minimal activity can cause clinical and/or electrocardiographic evidence of myocardial ischemia or angina at rest. They often complain of a circadian variation in angina that is more common in the morning. Angina on exertion and sometimes even at rest may be precipitated by cold temperature, emotion, and mental stress.
In rare instances, severe dynamic obstruction may develop alone in patients without organic obstructing lesions and can cause myocardial ischemia and angina at rest (see Prinzmetal [Variant] Angina; see also Chapters 49 and 53). On the other hand, in patients with severe fixed obstruction in one or more epicardial coronary arteries, only a minor increase in dynamic obstruction is necessary for coronary blood flow to fall below a critical level and cause myocardial ischemia.
Importance of Pathophysiologic Considerations in Configuring Therapy
The pathophysiologic and clinical correlations of ischemia in patients with SIHD may have important implications for the selection of anti-ischemic agents, as well as for their timing. The greater the contribution from increased myocardial O2 demand associated with tachycardia or increased contractility, the greater the likelihood that beta-blocking agents will be effective; nitrates and calcium channel–blocking agents, at least hypothetically, are more likely to be effective in episodes caused primarily by coronary vasoconstriction. The finding that an increase in myocardial O2 requirement precedes episodes of ischemia in most patients with chronic stable angina—that is, that they have demand angina—argues in favor of controlling the heart rate and blood pressure as a primary therapeutic approach.
Evaluation and Management
Noninvasive Testing
Biochemical Tests
In patients with SIHD, metabolic abnormalities that are risk factors for the development of CAD are frequently detected. Such abnormalities include hypercholesterolemia and other dyslipidemias (see Chapter 45), carbohydrate intolerance, and insulin resistance. Moreover, chronic kidney disease is strongly associated with risk for atherosclerotic vascular disease (see Chapter 88). All patients with established or suspected CAD warrant biochemical evaluation of total cholesterol, low-density lipoprotein (LDL) cholesterol, high-density lipoprotein (HDL) cholesterol, triglyceride, serum creatinine (estimated glomerular filtration rate [eGFR]), and fasting blood glucose levels.
Other biochemical markers have also been shown to be associated with higher risk for future cardiovascular events (see Chapter 42). Measurement of lipoprotein(a) and other lipid elements that are particularly atherogenic, such as apolipoprotein B and small dense LDL, appears to add prognostic information to the measurement of total cholesterol and LDL and may be considered a secondary target for therapy in patients who have achieved therapeutic targets for LDL.10 However, no consensus has been reached regarding routine measurement, and a simple approach based on calculation of non-HDL cholesterol (particularly in patients with triglyceride levels >200 mg/dL) may capture important information related to the presence of residual very low-density lipoprotein (VLDL) atherogenic remnants.11 Similarly, lipoprotein-associated phospholipase A2 (Lp-PLA2) is associated with risk for coronary heart disease (CHD), as well as for recurrent events, independent of traditional risk factors.12 An assay for Lp-PLA2 is available for clinical use but has not been incorporated into guidelines for routine risk assessment.13 Inhibitors of Lp-PLA2 are under investigation for the treatment of IHD.14,15 Although homocysteine has also been linked to atherogenesis and correlates with risk for CAD, prospective studies, in aggregate, have supported, at most, a modest increase in risk associated with elevated homocysteine levels and have not consistently demonstrated a relationship independent of traditional risk factors or other biochemical markers.16 Moreover, placebo-controlled trials have failed to demonstrate clinical benefit associated with folate replacement therapy as an intervention to mitigate the adverse effects of increased homocysteine levels.17 Therefore general screening for elevated homocysteine levels is not recommended.
Advances in understanding regarding the pathobiology of atherothrombosis (see Chapter 41) have generated interest in inflammatory biomarkers as noninvasive indicators of underlying atherosclerosis and cardiovascular risk. Measurement of the acute-phase protein high-sensitivity C-reactive protein (hsCRP) has shown a consistent relationship to risk for incident cardiovascular events, as well as with imaging findings of vulnerable atherosclerosis.18 The prognostic value of hsCRP is additive to traditional risk factors, including lipids19; however, its incremental clinical value for screening continues to be debated.20 Measurement of hsCRP in patients judged to be at intermediate risk by global risk assessment (10-year risk for CHD of 10% to 20%) may help direct further evaluation and therapy for primary prevention of CHD (see Chapter 42) and may be useful as an independent marker of prognosis in patients with established CAD.19 In a randomized, double-blind trial involving patients free of known atherosclerosis with an LDL level less than 130 mg/dL who were identified to be at higher vascular risk by virtue of elevated hsCRP (>2 mg/L), treatment with a 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor (statin) versus placebo significantly reduced the risk for cardiovascular death and incident atherosclerotic vascular events.21 On the basis of this finding, some professional society guidelines recommend the use of hsCRP as a risk indicator to support statin therapy for primary prevention in individuals at moderate global risk.22 Other biomarkers of inflammation, such as interleukin-6, myeloperoxidase, growth factors, and metalloproteinases, remain under study as potential markers of underlying athersclerosis.6 Genetic testing, including multigene expression from peripheral blood cells,23 is also under investigation. For example, a peripheral blood gene expression score based on expression values for 23 genes from peripheral blood cells has been developed and validated to assess the risk for obstructive CAD; a high negative predictive value with a very low rate of major adverse cardiac events over a 1-year period was demonstrated in patients with a low gene expression score.23
Biomarkers of Myocyte Injury, Ischemia, and Hemodynamic Stress
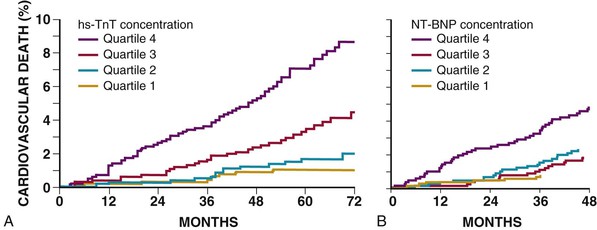
Blood levels of cardiac markers of myocardial necrosis are typically used to differentiate patients with acute MI from those with SIHD. However, with more sensitive assays for cardiac troponin, circulating biomarkers of myocyte injury have now been detected in patients with SIHD and shown to have a graded relationship with the subsequent risk for cardiovascular mortality and heart failure (Fig. 54-3A).24 Although such evidence may lead to new applications of troponin in patients with SIHD, clinical use in this population is currently not recommended.25 Biomarkers of myocardial ischemia are also under study. For example, the plasma concentration of brain natriuretic peptide (BNP) increases in response to spontaneous or provoked ischemia. Although BNP and N-terminal pro-BNP may not have sufficient specificity to aid in the diagnosis of SIHD, their concentration is associated with risk for cardiovascular events in those at risk for and with established CAD (Fig. 54-3B).26 Other novel biomarkers of hemodynamic stress, such as midregional proadrenomedullin (MR-proADM) and midregional pro–atrial natriuretic peptide (MR-proANP), also appear to provide incremental information regarding the risk for cardiovascular death in patients with SIHD and may be useful for guiding decisions regarding therapy.27 Growth differentiation factor-15,28 ST2, and galectin-329 are other biomarkers that may putatively reflect myocardial ischemia or its consequence and have been associated with outcomes in clinical studies of patients with SIHD.
Resting Electrocardiogram
Findings on the resting ECG (see Chapter 12) are normal in approximately half of patients with SIHD, and even patients with severe CAD may have a normal tracing at rest. A normal resting ECG suggests the presence of normal resting LV function and is an unusual finding in a patient with an extensive previous MI. The most common abnormalities on the ECG in patients with chronic CAD are nonspecific ST-T wave changes with or without abnormal Q waves. In patients with known CAD, however, the occurrence of ST-T wave abnormalities on the resting ECG (particularly if obtained during an episode of angina) can correlate with the severity of the underlying heart disease. This correlation explains the adverse association of ST-T wave changes with prognosis in these patients. In contrast, a normal resting ECG is a more favorable long-term prognostic sign in patients with suspected or definite CAD.
Interval ECGs may reveal the development of Q wave MIs that have gone unrecognized clinically. Various conduction disturbances, most frequently left bundle branch block and left anterior fascicular block, may occur in patients with SIHD. They are often associated with impairment of LV function, reflect multivessel CAD, and are an indicator of a relatively poor prognosis. Various arrhythmias, especially ventricular premature beats, may be present on the ECG, but they too have low sensitivity and specificity for accurately detecting CAD. LV hypertrophy on the ECG implies suggests a poor prognosis in patients with chronic stable angina. This finding implies the presence of underlying hypertension, aortic stenosis, hypertrophic cardiomyopathy, or previous MI with remodeling and warrants further evaluation, such as echocardiography to assess LV size, wall thickness, and function.
During an episode of angina pectoris, findings on the ECG become abnormal in 50% or more of patients with normal resting ECGs. The most common finding is ST-segment depression, although ST-segment elevation and normalization of previous resting ST-T wave depression or inversion (pseudonormalization) may develop. Ambulatory ECG monitoring (see Silent Myocardial Ischemia) provides a quantitative estimate of the frequency and duration of ischemic episodes during routine activities; however, its sensitivity for detecting CAD is less than that of exercise electrocardiography.