Chapter 16 Pulmonary Stenosis with Interatrial Communication
In 1769, Giovanni Battista Morgagni1 described pulmonary stenosis with a patent foramen ovale, and in 1848, Thomas Peacock2 published “Contraction of the Orifice of the Pulmonary Artery and Communication Between the Cavities of the Auricles by a Foramen Ovale.”
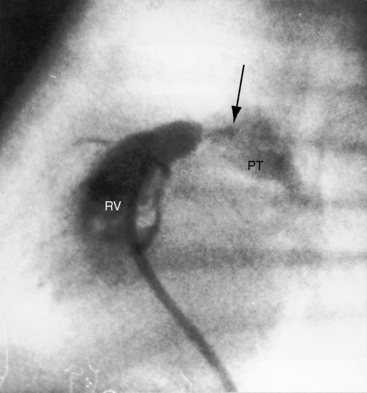
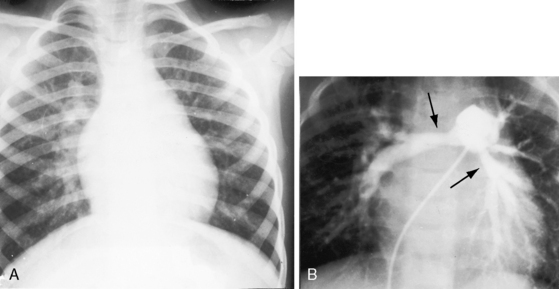
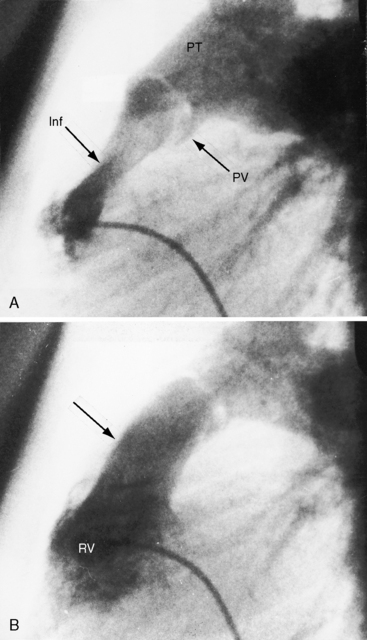
Pulmonary stenosis with reversed interatrial shunt has been called the triologie de Fallot.3 Right ventricular outflow obstruction resides in a stenotic mobile dome-shaped pulmonary valve (Figure 16-1) and is occasionally represented by stenosis of the pulmonary artery and its branches (Figure 16-2).4–7 Infundibular obstruction takes the form of secondary hypertrophic subpulmonary stenosis (Figure 16-3).8–11 The interatrial communication is by a patent foramen ovale or an ostium secundum atrial septal defect,4,6,8,12–15 less commonly by an ostium primum16 or sinus venosus atrial defect,14 or much less commonly by anomalous pulmonary venous connection.17 This chapter deals with pulmonary valve stenosis and a patent foramen ovale or a nonrestrictive ostium secundum atrial septal defect.4
Severe pulmonary valve stenosis with a right-to-left shunt through a patent foramen ovale is more common than pulmonary valve stenosis with a nonrestrictive atrial septal defect, irrespective of the direction of the shunt.4 A restrictive interatrial communication is almost always a patent foramen ovale, the shunt is right-to-left, and pulmonary stenosis is necessarily severe.4,18 A nonrestrictive interatrial communication is almost always an ostium secundum atrial septal defect, the shunt is left-to-right, and pulmonary stenosis is necessarily mild to moderate.4–6,8,12
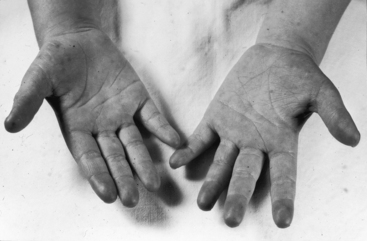
The physiologic consequences of pulmonary stenosis with an interatrial communication depend on the degree of obstruction to right ventricular outflow and the size of the interatrial communication.4,18 Patients with pulmonary stenosis and a right-to-left interatrial shunt (Figure 16-4) almost always have a severely stenotic pulmonary valve and a patent foramen ovale (see previous).4 Patients with pulmonary stenosis and a left-to-right interatrial shunt almost always have a mild to moderate pulmonary valve stenosis and a nonrestrictive ostium secundum atrial septal defect (see previous).4
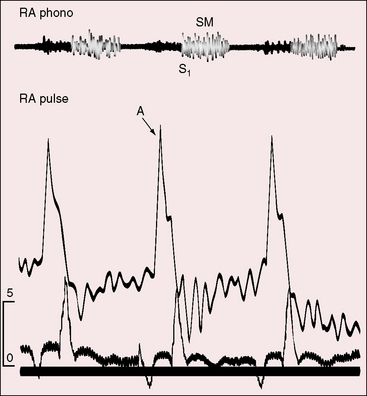
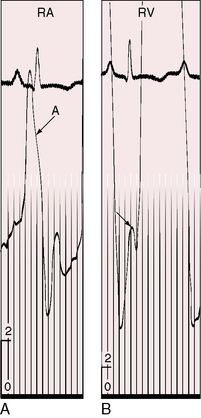
Severe pulmonary stenosis with right ventricular hypertrophy results in an increase in force of right atrial contraction (Figures 16-5 and 16-6) that generates a presystolic right-to-left interatrial shunt. High right atrial pressure stretches the margins of the foramen ovale and increases its patency. When right atrial blood escapes through the interatrial communication, pulmonary flow tends to fall reciprocally.
A nonrestrictive atrial septal defect with a large left-to-right shunt and mild to moderate pulmonary valve stenosis clinically resembles an isolated atrial septal defect (see Chapter 15). The small gradients generated by hyperkinetic right ventricular ejection across a normal pulmonary valve can generate a small gradient that should not be mistaken for mild pulmonary stenosis.
history
Familial pulmonary valve stenosis with atrial septal defect has been reported in a mother and her two children.19 Severe pulmonary stenosis with a right-to-left shunt across a patent foramen ovale is clinically analogous to isolated severe pulmonary valve stenosis, except for cyanosis that can be present at birth13,18 or can develop in childhood, puberty, or young adulthood.6,8,13,20 Infants usually come to attention because of a murmur; although in neonates with pinpoint pulmonary valve stenosis (see Figure 16-1), the murmur is disarmingly soft. Symptoms can be appreciable even when cyanosis is mild13,21 because right ventricular pressure can exceed systemic before the right-to-left interatrial shunt is established. However, among the author’s patients are a teenager who became cyanotic only when he engaged in sports,8 a woman with severe pulmonary stenosis and reversed interatrial shunt who was in good health until age 40 years,14 and a woman with a gradient of 120 mm Hg who underwent surgical repair at age 58 years (see Figure 16-5).
Giddiness, light-headedness, and syncope are provoked by exertion. Large jugular venous A waves (see Figure 16-5) are sometimes subjectively sensed, especially after effort or excitement. Chest pain that resembles angina pectoris is attributed to ischemia in the high-pressure hypertrophied right ventricle. Death is usually from right ventricular failure and less commonly from hypoxia, cerebral abscess, or infective endocarditis.5,6
Mild to moderate pulmonary stenosis with a nonrestrictive atrial septal defect clinically resembles an isolated nonrestrictive ostium secundum atrial septal defect (see Chapter 15), except for the conspicuous murmur of pulmonary stenosis.18 Relatively asymptomatic survival into the sixth and seventh decades is not uncommon. A 59-year-old man had a nonrestrictive sinus venous defect and calcific pulmonary stenosis.14
Physical appearance
Retarded growth and development are consequences of right ventricular failure that begins in infancy or early childhood. Cyanosis associated with pulmonary stenosis and a patent foramen ovale can be obvious or evident only during exercise.8,22,23 Mild or intermittent right-to-left shunts are sometimes manifested by highly colored cheeks or by distinctive erythema of the fingertips and toes (see Figure 16-4).8,20,21 The central cyanosis of a right-to-left shunt must be distinguished from the peripheral cyanosis of diminished skin blood flow caused by the low cardiac output of severe pulmonary stenosis. Peripheral cyanosis is accompanied by cold hands and feet and tends to be more pronounced in the lower extremities. When skin blood flow is improved by warmth, peripheral cyanosis diminishes or may vanish, and central cyanosis becomes more evident.
Children with a nonrestrictive atrial septal defect and mild to moderate pulmonary stenosis may have a delicate gracile body habitus, with weight more affected than height (see Figure 15-14). Noonan’s syndrome (see Figure 11-8C) is a distinctive appearance associated with dysplastic pulmonary valve stenosis that may coexist with an atrial septal defect.
jugular venous pulse
When pulmonary stenosis coexists with a nonrestrictive atrial septal defect, the hypertrophied right ventricle is less distensible, the right atrium contracts with greater force, and the A wave is prominent. The contour and height of the jugular pulse are determined by the presence and degree of pulmonary stenosis, not by the atrial defect. When pulmonary stenosis is severe enough to reverse the shunt, the jugular venous A wave is large, even giant (see Figures 16-5 and 16-6A). With the advent of right ventricular failure, the mean jugular venous pressure rises, and with it the V wave, especially if tricuspid regurgitation coexists.
Precordial movement and palpation
Pulmonary stenosis with a right-to-left interatrial shunt is associated with precordial palpation similar to severe isolated pulmonary valve stenosis (see Chapter 11). A nonrestrictive atrial septal defect with mild to moderate pulmonary stenosis is associated with precordial signs similar to an isolated atrial septal defect of equivalent size, except for a systolic thrill of coexisting pulmonary valve stenosis.23
Auscultation
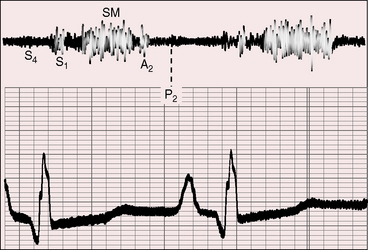
Severe pulmonary valve stenosis with a right-to-left interatrial shunt is accompanied by auscultatory signs analogous to isolated severe pulmonary stenosis (see Chapter 11). The systolic murmur is maximal in the second left intercostal space (Figure 16-7); radiates upward and to the left; extends up to or beyond the aortic component of the second heart sound, which it obscures (Figures 16-7 and 16-8A and D);24 and has a late systolic peak with a kite-shaped configuration.24,25 In neonates with critical pulmonary valve stenosis and right ventricular failure, the murmur is short and soft (see previous), and the pulmonary component of the second heart sound is delayed, soft, or absent (see Figures 16-7 and 16-8D). The most prominent auscultatory sign is then a holosystolic murmur of tricuspid regurgitation (see Figure 16-5).
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree