Principles and Applications of Cardiopulmonary Exercise Testing
INTRODUCTION
Cardiopulmonary exercise (CPX) testing draws on the recognition that the thorax represents a metabolic gas transport unit, the function of whose requisite components – diaphragm, heart, lungs, rib cage, and corresponding skeletal muscles – is to transport O2 to and CO2 from metabolizing tissues. Unit O2 and CO2 transport must adjust to physiological and pathophysiological stresses that augment the body’s consumption of oxygen (![]() O2) and carbon dioxide production (
O2) and carbon dioxide production (![]() CO2). During strenuous levels of muscular work, for example,
CO2). During strenuous levels of muscular work, for example, ![]() O2 may rise eightfold, accompanied by increased
O2 may rise eightfold, accompanied by increased ![]() CO2. Cardiovascular or ventilatory disease can disrupt the unit’s functional integrity. With severe disease, an abnormality in respiratory gas transport may be apparent at rest, when the body’s O2 requirements are modest. Resting function is preserved with less severe expressions of disease, but abnormal respiratory gas transport becomes apparent when the unit is stressed by an elevation in
CO2. Cardiovascular or ventilatory disease can disrupt the unit’s functional integrity. With severe disease, an abnormality in respiratory gas transport may be apparent at rest, when the body’s O2 requirements are modest. Resting function is preserved with less severe expressions of disease, but abnormal respiratory gas transport becomes apparent when the unit is stressed by an elevation in ![]() O2.
O2.
CPX testing includes the monitoring of respiratory gas exchange (![]() O2 and
O2 and ![]() CO2), minute ventilation (
CO2), minute ventilation (![]() E), and its components, tidal volume and respiratory rate, together with blood pressure, heart rate, and the electrocardiogram. CPX testing represents a useful approach in the clinical evaluation of a whole host of disorders and circumstances. This chapter addresses physiological principles and the clinical application of CPX testing in the evaluation of major disorders that impair heart or lung function. The chapter is by no means an exhaustive review. For a more detailed discussion of specific entities, the interested reader is referred to several other textbooks.1,2
E), and its components, tidal volume and respiratory rate, together with blood pressure, heart rate, and the electrocardiogram. CPX testing represents a useful approach in the clinical evaluation of a whole host of disorders and circumstances. This chapter addresses physiological principles and the clinical application of CPX testing in the evaluation of major disorders that impair heart or lung function. The chapter is by no means an exhaustive review. For a more detailed discussion of specific entities, the interested reader is referred to several other textbooks.1,2
PRINCIPLES, DEFINITIONS, AND CLINICAL APPLICATION OF CARDIOPULMONARY EXERCISE TESTING
The metabolic gas transport unit, also referred to as the “cardiopulmonary unit,” links metabolizing tissues to the atmosphere and its supply of O2. O2 transport to tissues must be precise and based upon prevailing need. CO2 produced by tissues must be eliminated into the atmosphere in an equally efficient manner.
 RESTING OXYGEN UPTAKE AND TRANSPORT
RESTING OXYGEN UPTAKE AND TRANSPORT
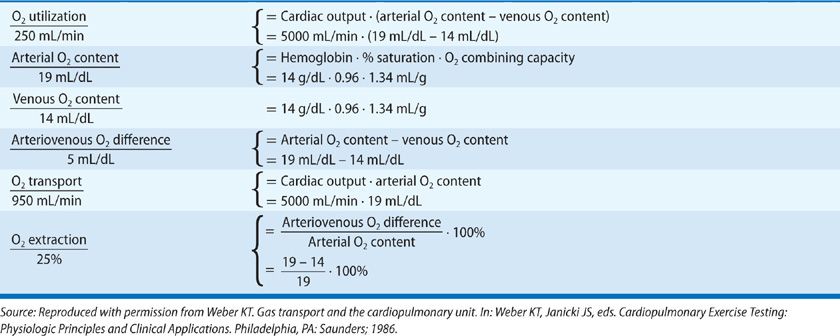
Concepts and calculations pertaining to ![]() O2 and O2 content, transport, and extraction are reviewed in Table 34-1. The heart and lungs accommodate to the metabolic requirements of tissues; they must do so on a moment-to-moment basis, according to physiological priorities. Tissue requirements for O2 dictate a certain
O2 and O2 content, transport, and extraction are reviewed in Table 34-1. The heart and lungs accommodate to the metabolic requirements of tissues; they must do so on a moment-to-moment basis, according to physiological priorities. Tissue requirements for O2 dictate a certain ![]() E and cardiac output. In an average-sized person, resting
E and cardiac output. In an average-sized person, resting ![]() O2 averages 250 mL/min or 3.5 mL/min/kg body weight (one metabolic equivalent) and is associated with a
O2 averages 250 mL/min or 3.5 mL/min/kg body weight (one metabolic equivalent) and is associated with a ![]() E of 8 to 10 L/min and cardiac output of 4 to 6 L/min. O2 transport, also termed O2 delivery, ranges between 730 and 1040 mL/min and is more than adequate to satisfy resting
E of 8 to 10 L/min and cardiac output of 4 to 6 L/min. O2 transport, also termed O2 delivery, ranges between 730 and 1040 mL/min and is more than adequate to satisfy resting ![]() O2. On average, 25% of arterial O2 content is extracted by tissues. O2 delivery and extraction each increase during physiological stress in proportion to the elevation in O2 demand. Factors that normally determine O2 availability at rest and during exercise include cardiac output, hemoglobin concentration and its percent saturation, and O2 extraction.
O2. On average, 25% of arterial O2 content is extracted by tissues. O2 delivery and extraction each increase during physiological stress in proportion to the elevation in O2 demand. Factors that normally determine O2 availability at rest and during exercise include cardiac output, hemoglobin concentration and its percent saturation, and O2 extraction.
 EXERCISE OXYGEN UPTAKE AND TRANSPORT
EXERCISE OXYGEN UPTAKE AND TRANSPORT
![]() E and O2 delivery must each rise during exercise. Strenuous work can raise
E and O2 delivery must each rise during exercise. Strenuous work can raise ![]() E 8 to 10 times its resting level. Ventilation normally poses no limitation on the ability of tissues to carry out aerobic work. By contrast, the extent to which cardiac output rises during progressive work is less dramatic. In untrained subjects, cardiac output increases four to five times its resting value. Cardiac output rises 600 mL/min for every 100 mL/min increment in
E 8 to 10 times its resting level. Ventilation normally poses no limitation on the ability of tissues to carry out aerobic work. By contrast, the extent to which cardiac output rises during progressive work is less dramatic. In untrained subjects, cardiac output increases four to five times its resting value. Cardiac output rises 600 mL/min for every 100 mL/min increment in ![]() O2. This is considered the normal “gain” setting between the heart and its cardiac output and
O2. This is considered the normal “gain” setting between the heart and its cardiac output and ![]() O2. O2 availability during physical activity is further ensured by enhanced O2 extraction and circulatory autoregulation. Reflexive and humoral influences produce vasoconstriction in less metabolically active tissues, permitting a greater apportionment of blood flow to exercising muscle.
O2. O2 availability during physical activity is further ensured by enhanced O2 extraction and circulatory autoregulation. Reflexive and humoral influences produce vasoconstriction in less metabolically active tissues, permitting a greater apportionment of blood flow to exercising muscle.
Physiological limits to the elevation in cardiac output (i.e., cardiac reserve) and O2 extraction (approximately 75%–80% of arterial O2 content) determine aerobic capacity of untrained subjects to incremental exercise. Beyond these physiological limits, any additional increment in work is not accompanied by an elevation in O2; a plateau in ![]() O2 is attained and is termed the maximal oxygen uptake (
O2 is attained and is termed the maximal oxygen uptake (![]() O2max). CPX test results, including
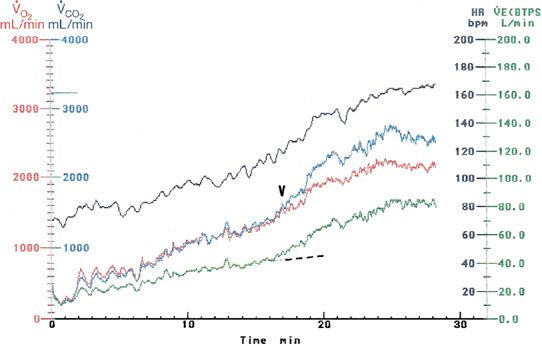
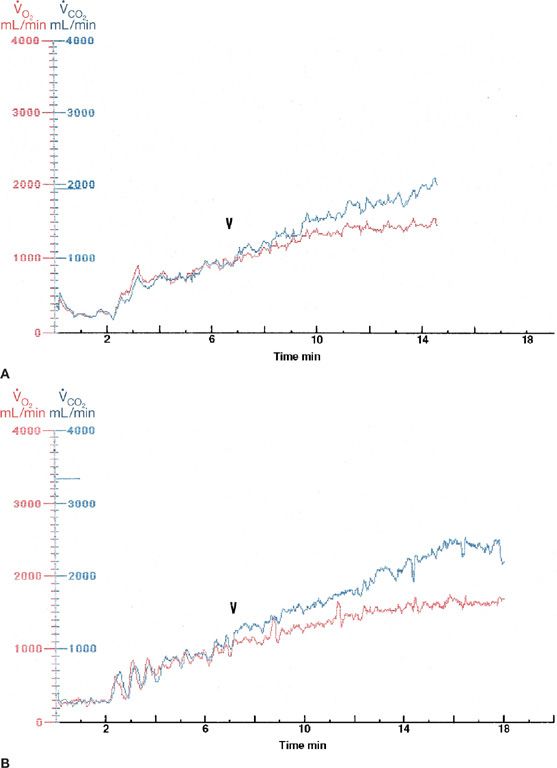
O2max). CPX test results, including ![]() O2max, are shown in Figure 34-1 for a 40-year-old man without clinically apparent heart or lung disease. Shown are individual responses in
O2max, are shown in Figure 34-1 for a 40-year-old man without clinically apparent heart or lung disease. Shown are individual responses in ![]() O2,
O2, ![]() CO2,
CO2, ![]() E, and heart rate during progressive increments in treadmill work. A
E, and heart rate during progressive increments in treadmill work. A ![]() O2max of 2198 mL/min (27.2 mL/min/kg) was attained. This is a true plateau in
O2max of 2198 mL/min (27.2 mL/min/kg) was attained. This is a true plateau in ![]() O2, with
O2, with ![]() O2 remaining invariant for 2.5 stages (5 minutes) of exercise.
O2 remaining invariant for 2.5 stages (5 minutes) of exercise.
Figure 34-1 Cardiopulmonary exercise response in a 40-year-old man without heart or lung disease. Shown are 2 minutes of standing rest, followed by incremental treadmill exercise. Individual responses (color coded) include oxygen uptake (![]() O2), carbon dioxide production (
O2), carbon dioxide production (![]() CO2), minute ventilation (
CO2), minute ventilation (![]() E), and heart rate (HR). Maximal O2 uptake, a plateau in
E), and heart rate (HR). Maximal O2 uptake, a plateau in ![]() O2 was attained after the crossover of
O2 was attained after the crossover of ![]() CO2 and
CO2 and ![]() O2 (arrowhead), representing the AT and accompanied by a disproportionate (broken line) rise in
O2 (arrowhead), representing the AT and accompanied by a disproportionate (broken line) rise in ![]() E.
E.
![]() O2max should not be equated or used synonymously with peak
O2max should not be equated or used synonymously with peak ![]() O2 achieved during symptom-limited exercise.
O2 achieved during symptom-limited exercise. ![]() O2max reflects a person’s aerobic capacity—a physiological capacity of the cardiovascular system. In an average-sized, untrained person whose maximum cardiac output and arteriovenous oxygen difference are 20 L/min and 12 mL/dL, respectively, a
O2max reflects a person’s aerobic capacity—a physiological capacity of the cardiovascular system. In an average-sized, untrained person whose maximum cardiac output and arteriovenous oxygen difference are 20 L/min and 12 mL/dL, respectively, a ![]() O2max of 2400 mL/min is expected. In athletes, a greater cardiac reserve and enhanced capacity for oxidative metabolism by trained skeletal muscle are available, providing for greater aerobic capacity. In patients with heart disease, whose ability to raise cardiac output during exercise is impaired,
O2max of 2400 mL/min is expected. In athletes, a greater cardiac reserve and enhanced capacity for oxidative metabolism by trained skeletal muscle are available, providing for greater aerobic capacity. In patients with heart disease, whose ability to raise cardiac output during exercise is impaired, ![]() O2max is proportionally reduced (see above).
O2max is proportionally reduced (see above).
 CARBON DIOXIDE PRODUCTION
CARBON DIOXIDE PRODUCTION
The right heart “accepts” metabolically produced CO2, and the alveolar exchange surface expels CO2 into the atmosphere. CO2 is a major respiratory stimulant that maintains eucapnia. Seventy-five to 80% of O2 is converted to CO2. Accordingly, resting ![]() CO2 averages 190 mL/min and represents a metabolic source of CO2. The resting
CO2 averages 190 mL/min and represents a metabolic source of CO2. The resting ![]() CO2/
CO2/![]() O2 ratio, or respiratory gas exchange ratio (R), typically ranges between 0.75 and 0.85. The absolute value of R depends on the proportion of carbohydrates and fats available from the diet.
O2 ratio, or respiratory gas exchange ratio (R), typically ranges between 0.75 and 0.85. The absolute value of R depends on the proportion of carbohydrates and fats available from the diet. ![]() O2 and
O2 and ![]() CO2 rise in proportion to one another during physical activity as long as an adequate amount of O2 is available to sustain oxidative metabolism.
CO2 rise in proportion to one another during physical activity as long as an adequate amount of O2 is available to sustain oxidative metabolism.
With strenuous levels of muscular work, ![]() O2 rises to a level where the heart is unable to provide O2 at a commensurate rate. Consequently, tissue O2 availability becomes inadequate. Working skeletal muscle enhances its use of less efficient anaerobic metabolism to derive energy. This leads to lactate production from working muscle beyond that normally produced. This non-metabolic source of CO2 is derived from rapid buffering of the lactate by bicarbonate; the CO2 generated serves as a respiratory stimulant. The accompanying increase in
O2 rises to a level where the heart is unable to provide O2 at a commensurate rate. Consequently, tissue O2 availability becomes inadequate. Working skeletal muscle enhances its use of less efficient anaerobic metabolism to derive energy. This leads to lactate production from working muscle beyond that normally produced. This non-metabolic source of CO2 is derived from rapid buffering of the lactate by bicarbonate; the CO2 generated serves as a respiratory stimulant. The accompanying increase in ![]() E maintains eucapnia and raises the respiratory gas exchange ratio above that associated with aerobic metabolism. Anaerobic metabolism during a progressive exercise test is heralded by this disproportionate rise in
E maintains eucapnia and raises the respiratory gas exchange ratio above that associated with aerobic metabolism. Anaerobic metabolism during a progressive exercise test is heralded by this disproportionate rise in ![]() E and
E and ![]() CO2 relative to
CO2 relative to ![]() O2. The corresponding level of
O2. The corresponding level of ![]() O2 at which anaerobic metabolism occurs is termed the anaerobic threshold (AT).1 The point during exercise at which
O2 at which anaerobic metabolism occurs is termed the anaerobic threshold (AT).1 The point during exercise at which ![]() CO2 exceeds
CO2 exceeds ![]() O2 and
O2 and ![]() E rises disproportionately is shown in Figure 34-1. Anaerobiosis normally occurs when 60% or more of a person’s aerobic capacity has been attained. For the 40-year-old man whose exercise response is shown in Figure 34-1, the AT occurred at a
E rises disproportionately is shown in Figure 34-1. Anaerobiosis normally occurs when 60% or more of a person’s aerobic capacity has been attained. For the 40-year-old man whose exercise response is shown in Figure 34-1, the AT occurred at a ![]() O2 of 18.8 mL/min/kg, or 69% of his
O2 of 18.8 mL/min/kg, or 69% of his ![]() O2max.
O2max.
 CLINICAL APPLICATION OF CARDIOPULMONARY EXERCISE TESTING
CLINICAL APPLICATION OF CARDIOPULMONARY EXERCISE TESTING
Patients with cardiovascular or respiratory disease of mild-to-moderate severity frequently note limiting symptoms of fatigue or breathlessness during physical activity. Because their quality of life is compromised, they seek or are referred for medical evaluation. Re-creating muscular work in a monitored setting permits an evaluation of the nature and severity of such symptoms and the relative importance of abnormal heart or lung function. This strategy provides information surpassing that available from static measures of heart and lung function, such as ejection fraction, lung volumes, or airflows, determined at rest. The continuous monitoring of ![]() O2,
O2, ![]() CO2,
CO2, ![]() E, respiratory rate, and tidal volume during incremental exercise can be performed simply and on a breath-by-breath basis. Data shown in Figure 34-1 are displayed throughout the test. The choice of a particular CPX test (see above) depends on the nature and expression of the clinical disorder and the particular problem to be addressed. For most clinical evaluations, isotonic forms of exercise are used. Isotonic work is an acceptable, negotiable, and reproducible form of exercise for patients with heart or lung disease.
E, respiratory rate, and tidal volume during incremental exercise can be performed simply and on a breath-by-breath basis. Data shown in Figure 34-1 are displayed throughout the test. The choice of a particular CPX test (see above) depends on the nature and expression of the clinical disorder and the particular problem to be addressed. For most clinical evaluations, isotonic forms of exercise are used. Isotonic work is an acceptable, negotiable, and reproducible form of exercise for patients with heart or lung disease.
It should be noted, however, that while noninvasive CPX testing may help determine the impairment in aerobic capacity, abnormalities in ventilation with exercise, and their severity in patients with lung disease, these parameters are not necessarily diagnostic. For example, ![]() O2max, AT, or exercise
O2max, AT, or exercise ![]() E do not identify the underlying structural defect responsible for a patient’s abnormal response. This may require invasive monitoring during CPX testing to identify specific hemodynamic abnormalities. Echocardiography and specialized pulmonary function studies may be required. The physician must draw upon sound clinical judgment and complementary laboratory tests to derive an understanding of the nature and severity of the heart or lung disease.
E do not identify the underlying structural defect responsible for a patient’s abnormal response. This may require invasive monitoring during CPX testing to identify specific hemodynamic abnormalities. Echocardiography and specialized pulmonary function studies may be required. The physician must draw upon sound clinical judgment and complementary laboratory tests to derive an understanding of the nature and severity of the heart or lung disease.
 NONINVASIVE TREADMILL EXERCISE
NONINVASIVE TREADMILL EXERCISE
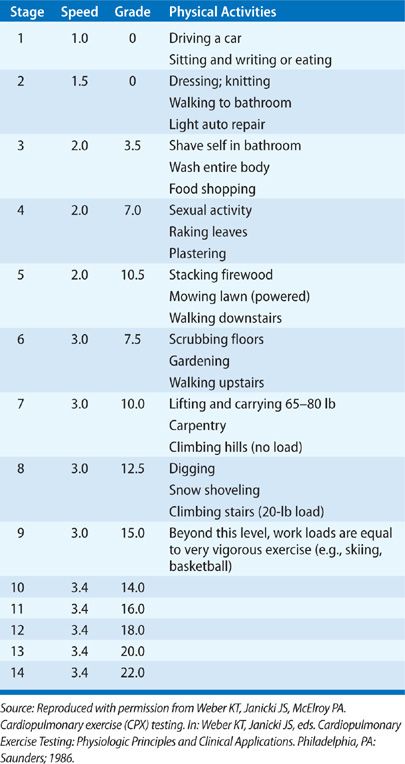
Walking represents a common daily exercise rather than a specialized skill. A patient who walks into the physician’s office or down a hospital corridor can walk on a treadmill at 1.0 or 1.5 mph, zero grade. Treadmills are programmable. The Bruce protocol, which employs marked increments in treadmill speed and slope over short periods in the evaluation of myocardial ischemia, may not be useful for patients with limited exercise tolerance. A modified Naughton protocol of gradually progressive exercise (Table 34-2) serves to stress the cardiopulmonary unit for patients with heart or lung disease who have a wide range of exercise tolerance. In this protocol, the first two stages of exercise represent very low workloads and are a warmup for patients with heart or lung disease of minor severity; the stages represent near-maximal exercise for patients with more advanced disease.
![]() O2max is defined as
O2max is defined as ![]() O2 that remains invariant (<1 mL/min/kg for 30 seconds or more) despite an increment in workload. An invariant
O2 that remains invariant (<1 mL/min/kg for 30 seconds or more) despite an increment in workload. An invariant ![]() O2 for at least two stages of exercise is preferred (Fig. 34-1).
O2 for at least two stages of exercise is preferred (Fig. 34-1). ![]() O2max follows the AT, and this definition of
O2max follows the AT, and this definition of ![]() O2max presumes that the AT has already been achieved. The AT generally occurs at 60% of a patient’s aerobic capacity.
O2max presumes that the AT has already been achieved. The AT generally occurs at 60% of a patient’s aerobic capacity. ![]() O2max associated with incremental treadmill exercise provides a greater aerobic capacity than does cycle ergometry because it works a larger group of muscles.3 A patient’s aerobic capacity to incremental treadmill exercise is used to grade the functional impairment (Table 34-3).
O2max associated with incremental treadmill exercise provides a greater aerobic capacity than does cycle ergometry because it works a larger group of muscles.3 A patient’s aerobic capacity to incremental treadmill exercise is used to grade the functional impairment (Table 34-3). ![]() O2max is an objective measure of functional status—in contradistinction to the New York Heart Association classification, which is based on perceptions and biases of the patient and physician. Treadmill O2max determination is reproducible in patients with a wide variety of cardiovascular disorders.2 A
O2max is an objective measure of functional status—in contradistinction to the New York Heart Association classification, which is based on perceptions and biases of the patient and physician. Treadmill O2max determination is reproducible in patients with a wide variety of cardiovascular disorders.2 A ![]() O2max of under 20 mL/min/kg has been selected as the cutoff for grading impaired aerobic capacity; adult men and women, including the elderly (over 65 years of age), have an expected
O2max of under 20 mL/min/kg has been selected as the cutoff for grading impaired aerobic capacity; adult men and women, including the elderly (over 65 years of age), have an expected ![]() O2max of more than 20 mL/min/kg.4
O2max of more than 20 mL/min/kg.4
The duration of symptom-free treadmill exercise should not be equated with ![]() O2max. Treadmill time suffers from not having an objective, quantitative end-point. Differences in gait and body weight create different levels of work for equivalent stages of treadmill exercise. Symptom-limited exercise time is subject to patient motivation and physician bias.5 Peak heart rate attained with exercise is also a less precise measure of
O2max. Treadmill time suffers from not having an objective, quantitative end-point. Differences in gait and body weight create different levels of work for equivalent stages of treadmill exercise. Symptom-limited exercise time is subject to patient motivation and physician bias.5 Peak heart rate attained with exercise is also a less precise measure of ![]() O2max. This is particularly true in patients with atrial fibrillation.
O2max. This is particularly true in patients with atrial fibrillation.
Determination of the AT can be defined according to one or more criteria.1 These include (1) a disproportionate rise in ![]() CO2,
CO2, ![]() E, or R relative to
E, or R relative to ![]() O2 and (2) a disproportionate rise in end-tidal CO2 relative to end-tidal O2. These criteria can best be applied to breath-by-breath respiratory gas exchange data. In our laboratory, a simpler strategy is used. The AT is identified as the level of
O2 and (2) a disproportionate rise in end-tidal CO2 relative to end-tidal O2. These criteria can best be applied to breath-by-breath respiratory gas exchange data. In our laboratory, a simpler strategy is used. The AT is identified as the level of ![]() O2 attained during treadmill work after the plots of
O2 attained during treadmill work after the plots of ![]() O2 and
O2 and ![]() CO2 cross, when R exceeds 1.0. Figure 34-1 depicts the crossover in
CO2 cross, when R exceeds 1.0. Figure 34-1 depicts the crossover in ![]() CO2 and
CO2 and ![]() O2 from breath-by-breath gas exchange data monitored throughout incremental treadmill exercise. It also demonstrates the point at which
O2 from breath-by-breath gas exchange data monitored throughout incremental treadmill exercise. It also demonstrates the point at which ![]() E rises disproportionately. Measured days or weeks apart, this noninvasive determination of the AT is reproducible in a wide range of patients with cardiac or circulatory failure and correlates with lactate threshold (see above).2,6,7
E rises disproportionately. Measured days or weeks apart, this noninvasive determination of the AT is reproducible in a wide range of patients with cardiac or circulatory failure and correlates with lactate threshold (see above).2,6,7
The normal ventilatory response to incremental treadmill exercise consists of an increase in ![]() E created by an increase in respiratory rate and tidal volume. Ventilatory reserves, represented by maximal voluntary ventilation (MVV) and vital capacity determined during routine pulmonary function testing, are only partly utilized during light, moderate, and maximal exercise by normal persons. The ratio of maximal exercise
E created by an increase in respiratory rate and tidal volume. Ventilatory reserves, represented by maximal voluntary ventilation (MVV) and vital capacity determined during routine pulmonary function testing, are only partly utilized during light, moderate, and maximal exercise by normal persons. The ratio of maximal exercise ![]() E to MVV reflects use of this ventilatory reserve. Exercise
E to MVV reflects use of this ventilatory reserve. Exercise ![]() E in normal subjects and patients with predominant cardiovascular disease rarely exceeds 50% of MVV.8 The same is true of the ratio between maximal exercise tidal volume and vital capacity. These limitations in ventilatory responses are consistent with a ventilatory effort that can be voluntarily sustained at rest without the appearance of fatigue or breathlessness.
E in normal subjects and patients with predominant cardiovascular disease rarely exceeds 50% of MVV.8 The same is true of the ratio between maximal exercise tidal volume and vital capacity. These limitations in ventilatory responses are consistent with a ventilatory effort that can be voluntarily sustained at rest without the appearance of fatigue or breathlessness.
An oximeter, worn on either an earlobe or a finger, provides noninvasive monitoring of arterial O2 saturation during exercise. This is a useful screening procedure in patients in whom O2 desaturation might be anticipated (e.g., those with congenital heart disease with right-to-left shunt, restrictive or obstructive lung disease, or pulmonary vascular disease). Normal subjects and patients with chronic cardiac or circulatory failure do not develop arterial hypoxemia (arterial O2 saturation under 90%) during exercise. In patients in whom O2 desaturation is evident from oximetry, confirmatory evidence from direct measurement of arterial blood gases during repeat exercise may be advisable.
Thus, incremental treadmill exercise can be used to determine the following: the AT with a submaximal test, the AT and ![]() O2max with a maximal test, the ventilatory response to submaximal or maximal exercise, and arterial O2 desaturation during submaximal or maximal exercise.
O2max with a maximal test, the ventilatory response to submaximal or maximal exercise, and arterial O2 desaturation during submaximal or maximal exercise.
 INVASIVE TREADMILL EXERCISE
INVASIVE TREADMILL EXERCISE
Invasive hemodynamic monitoring may be necessary to better define the nature and severity of an underlying cardiopulmonary disorder.2 A triple-lumen flotation catheter can be safely used for hemodynamic monitoring during upright exercise. The hemodynamic response to incremental treadmill exercise in normal subjects is characterized by a progressive rise in cardiac output, accomplished with minimal elevations in left and right ventricular filling pressures. The rise in cardiac output occurs because of an increment in stroke volume, which is most apparent at low and moderate workloads, and because of an elevation in heart rate, which accompanies the entire exercise response. Systemic O2 extraction rises progressively with incremental exercise to exceed 70% at maximal workloads. A rise in mixed venous lactate concentration, as observed with pulmonary arterial blood sampling, occurs when O2 extraction exceeds 60% and when the subject is working at greater than 60% of ![]() O2max.
O2max.
Systolic and mean arterial pressures rise during upright exercise. Because of skeletal muscle vasodilatation, arterial diastolic pressure remains essentially invariant during exercise. Systemic vascular resistance falls by 50% to approximately 600 dynes·s·cm–5 during incremental, isotonic treadmill exercise. In normal persons, pulmonary artery systolic, mean, and diastolic pressures rise only minimally with exercise and only with higher workloads. Pulmonary vascular resistance, like systemic vascular resistance, falls 50% to about 60 dynes·s·cm–5 during incremental isotonic exercise.
CHRONIC CARDIAC FAILURE
In physiological terms, cardiac failure is defined as an impairment in cardiac output secondary to a disease process affecting the myocardium. Ischemic heart disease and dilated cardiomyopathies are examples of disease entities that can result in chronic cardiac failure. ![]() O2max and the AT each predict cardiac reserve and, thereby, the severity of cardiac failure. These parameters further serve to objectively demonstrate a patient’s functional capacity, which is not predicted from the cardiac ejection fraction. Patients with an ejection fraction under 20% may still be able to swim.
O2max and the AT each predict cardiac reserve and, thereby, the severity of cardiac failure. These parameters further serve to objectively demonstrate a patient’s functional capacity, which is not predicted from the cardiac ejection fraction. Patients with an ejection fraction under 20% may still be able to swim.
 SYSTOLIC DYSFUNCTION
SYSTOLIC DYSFUNCTION
In patients with chronic cardiac failure, ![]() O2max attained during incremental treadmill exercise is primarily a function of maximal cardiac output.8,9 This conclusion has been confirmed by numerous studies.10–14 An impairment in aerobic capacity is gauged according to the exercise AT and
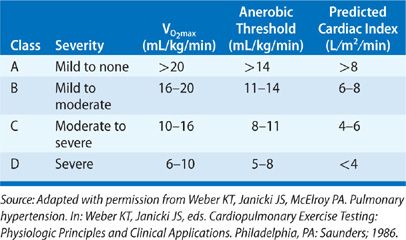
O2max attained during incremental treadmill exercise is primarily a function of maximal cardiac output.8,9 This conclusion has been confirmed by numerous studies.10–14 An impairment in aerobic capacity is gauged according to the exercise AT and ![]() O2max and assigned a functional class as reviewed in Table 34-3. These parameters are, in turn, used to predict maximal exercise cardiac index (or cardiac reserve). Examples of
O2max and assigned a functional class as reviewed in Table 34-3. These parameters are, in turn, used to predict maximal exercise cardiac index (or cardiac reserve). Examples of ![]() O2max and the AT attained by two patients with chronic cardiac failure (one class B, the other class C) are given in Figure 34-2. To measure
O2max and the AT attained by two patients with chronic cardiac failure (one class B, the other class C) are given in Figure 34-2. To measure ![]() O2max in such patients, they must be exercised to exhaustion. The AT is achieved at submaximal workloads short of exhaustion; it, too, stratifies the degree of cardiac dysfunction.
O2max in such patients, they must be exercised to exhaustion. The AT is achieved at submaximal workloads short of exhaustion; it, too, stratifies the degree of cardiac dysfunction.
Figure 34-2 Cardiopulmonary exercise test results for a 45-year-old woman (A) and a 40-year-old man (right panel), each with ischemic heart disease and chronic cardiac failure. Only ![]() O2 and
O2 and ![]() CO2 are shown, to better demonstrate the anaerobic threshold (AT) and
CO2 are shown, to better demonstrate the anaerobic threshold (AT) and ![]() O2max attained by each patient. On the left, the AT was seen with a
O2max attained by each patient. On the left, the AT was seen with a ![]() O2 of 11.6 mL/min/kg and a
O2 of 11.6 mL/min/kg and a ![]() O2max of 16.5 mL/min/kg. This represents a functional class B response. The AT and
O2max of 16.5 mL/min/kg. This represents a functional class B response. The AT and ![]() O2max are 8.5 and 13.7 mL/min/kg, respectively (B). This corresponds to functional class C.
O2max are 8.5 and 13.7 mL/min/kg, respectively (B). This corresponds to functional class C.
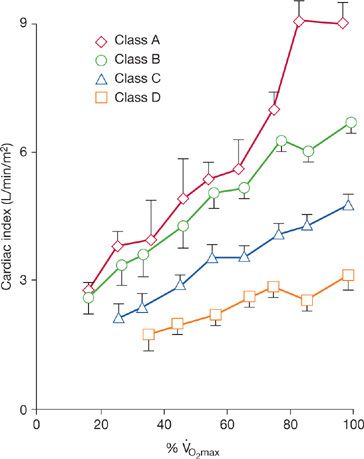
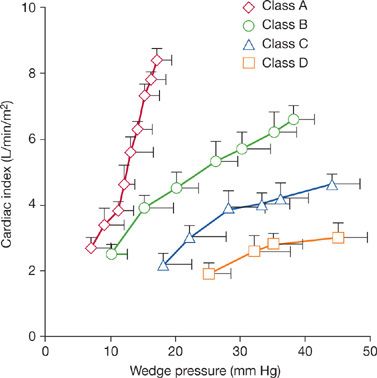
Validation of these concepts was obtained during treadmill exercise using invasive measures of cardiac output and mixed venous lactate concentration.15,16 Patients had chronic cardiac failure of varying severity (classes A to D), due to either ischemic or myopathic heart disease. In each exercise class, the arteriovenous O2 difference rose to 12 mL/dL or more at maximum exercise, corresponding to a systemic O2 extraction in excess of 70%, suggesting that O2 extraction reached maximal physiological levels. The reduction in aerobic capacity of a patient with chronic cardiac failure is, therefore, due primarily to impaired cardiac reserve. The cardiac output–O2 relation to progressive treadmill exercise for these patients is given in Figure 34-3. For each exercise class, cardiac output is presented as a percentage of ![]() O2max (set equal to 100%) that existed at rest and throughout each stage of exercise. Cardiac output rose by 600 mL/min/m2 for each dL/min/m2 increase in
O2max (set equal to 100%) that existed at rest and throughout each stage of exercise. Cardiac output rose by 600 mL/min/m2 for each dL/min/m2 increase in ![]() O2 in each class. This indicates that the heart responds to tissue O2 requirements irrespective of the severity of heart failure, but it is limited by the maximal cardiac output it can attain. Differences in cardiac output achieved at peak exercise are seen between classes. Progressive reductions in cardiac reserve are responsible for different aerobic capacities observed in these patients.
O2 in each class. This indicates that the heart responds to tissue O2 requirements irrespective of the severity of heart failure, but it is limited by the maximal cardiac output it can attain. Differences in cardiac output achieved at peak exercise are seen between classes. Progressive reductions in cardiac reserve are responsible for different aerobic capacities observed in these patients. ![]() O2max, therefore, serves as a noninvasive measure of peak exercise cardiac output and is given for each functional class in Table 34-3.
O2max, therefore, serves as a noninvasive measure of peak exercise cardiac output and is given for each functional class in Table 34-3.
Figure 34-3 Relationship between treadmill exercise cardiac index and normalized aerobic capacity for patients with chronic cardiac failure of diverse origin and severity, subdivided according to each functional class. (Reproduced with permission from Weber KT, Janicki JS. Cardiopulmonary exercise testing for evaluation of chronic cardiac failure. Am J Cardiol. 1985;55(2):22A–31A.)
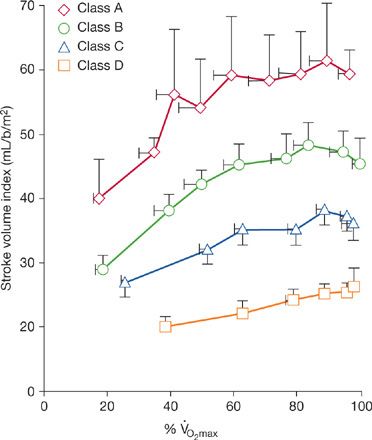
The cardiac output response to exercise is a function of the rises in stroke volume and heart rate. Responses in stroke volume for patients with chronic cardiac failure are shown in Figure 34-4 for each exercise class. In class A and B patients, stroke volume rises 50% during lighter workloads that represent less than 60% of ![]() O2max; at larger workloads, further increments in stroke volume are less apparent. A 25% rise in stroke volume occurs at submaximal exercise in class C patients, whereas in class D patients, exercise stroke volume is no different from its resting value. Exercise stroke volume is a result of several factors, including systolic wall stress, mitral or tricuspid regurgitation that may appear during exercise, and depressed myocardial contractility.
O2max; at larger workloads, further increments in stroke volume are less apparent. A 25% rise in stroke volume occurs at submaximal exercise in class C patients, whereas in class D patients, exercise stroke volume is no different from its resting value. Exercise stroke volume is a result of several factors, including systolic wall stress, mitral or tricuspid regurgitation that may appear during exercise, and depressed myocardial contractility.
Figure 34-4 Relationship between treadmill exercise stroke volume index and normalized aerobic capacity for patients with chronic cardiac failure of varying severity, as expressed by each functional class. (Reproduced with permission from Weber KT, Janicki JS. Cardiopulmonary exercise testing for evaluation of chronic cardiac failure. Am J Cardiol. 1986;55(2):22A–31A.)
For each functional class of chronic cardiac failure, the heart rate– ![]() O2 response to upright incremental exercise is represented by a common slope.17 The average slope is 3.6 beats/min for every 1-mL/min/kg increment in
O2 response to upright incremental exercise is represented by a common slope.17 The average slope is 3.6 beats/min for every 1-mL/min/kg increment in ![]() O2. Peak heart rate achieved is a function of maximal workload performed. Maximal exercise heart rate is, therefore, different for each class. In class D patients, the elevation in heart rate is the sole mechanism by which cardiac output rises during exercise.
O2. Peak heart rate achieved is a function of maximal workload performed. Maximal exercise heart rate is, therefore, different for each class. In class D patients, the elevation in heart rate is the sole mechanism by which cardiac output rises during exercise.
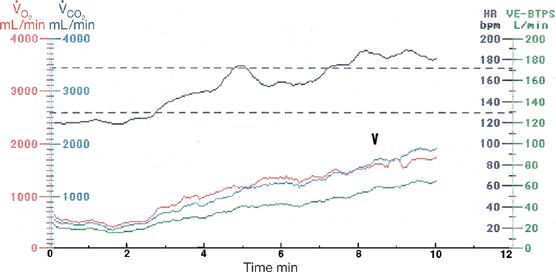
Some patients with chronic cardiac failure deviate from this heart rate– ![]() O2 relation by having an inappropriate sinus tachycardia, either at rest and throughout exercise or simply during exercise. In the presence of a reduced ejection fraction and ventricular dilation, this inappropriately rapid heart rate further compromises exercise cardiac output and reduces aerobic capacity. β-Adrenergic receptor blockade is useful in attenuating resting or exercise heart rate under these circumstances.18 Such chronotropic dysfunction (see below) to exercise may also apply to patients with chronic atrial fibrillation. An example of an inappropriate rapid heart rate relative to incremental treadmill exercise (Naughton protocol) is given in Figure 34-5 for a patient with atrial fibrillation and dilated cardiomyopathy of uncertain origin.
O2 relation by having an inappropriate sinus tachycardia, either at rest and throughout exercise or simply during exercise. In the presence of a reduced ejection fraction and ventricular dilation, this inappropriately rapid heart rate further compromises exercise cardiac output and reduces aerobic capacity. β-Adrenergic receptor blockade is useful in attenuating resting or exercise heart rate under these circumstances.18 Such chronotropic dysfunction (see below) to exercise may also apply to patients with chronic atrial fibrillation. An example of an inappropriate rapid heart rate relative to incremental treadmill exercise (Naughton protocol) is given in Figure 34-5 for a patient with atrial fibrillation and dilated cardiomyopathy of uncertain origin.
Figure 34-5 Cardiopulmonary exercise test results in a 48-year-old man with atrial fibrillation and dilated (idiopathic) cardiomyopathy. Note the rapid heart rate at rest and throughout incremental treadmill exercise. Predicted maximum heart rate range in this patient is shown by the broken lines. He achieved this rate during the first stage of exercise and exceeded it during the last stage of exercise. This is an inappropriate heart rate response. The AT is 13 mL/min/kg (arrow), in keeping with functional class B. He did not achieve ![]() O2max and, therefore, had a peak
O2max and, therefore, had a peak ![]() O2 of 15 mL/min/kg.
O2 of 15 mL/min/kg.
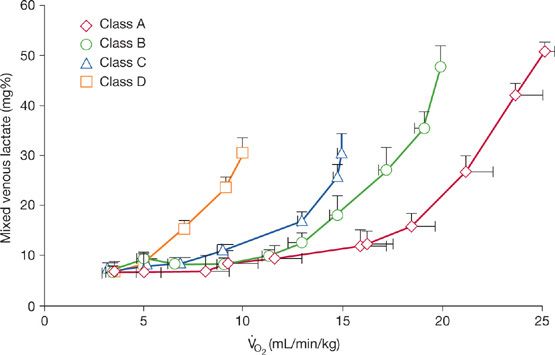
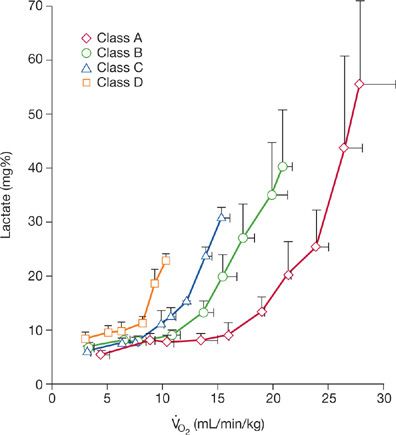
As in normal persons, lactate production appears in patients with chronic cardiac failure when systemic O2 extraction exceeds 60%. Mixed venous lactate concentration during exercise rises above resting values when 60% or more of ![]() O2max is attained.7,16 Given differences in aerobic capacity between exercise classes, different workloads are associated with this lactate threshold (Fig. 34-6). In class D patients whose cardiac output response is limited, the lactate threshold occurs at very light workloads (
O2max is attained.7,16 Given differences in aerobic capacity between exercise classes, different workloads are associated with this lactate threshold (Fig. 34-6). In class D patients whose cardiac output response is limited, the lactate threshold occurs at very light workloads (![]() O2 of 5–8 mL/min/kg). Corresponding values for class C, B, and A patients are 8 to 11 mL/min/kg, 11 to 14 mL/min/kg, and more than 14 mL/min/kg, respectively. Thus, lactate threshold and
O2 of 5–8 mL/min/kg). Corresponding values for class C, B, and A patients are 8 to 11 mL/min/kg, 11 to 14 mL/min/kg, and more than 14 mL/min/kg, respectively. Thus, lactate threshold and ![]() O2max reflect the severity of chronic cardiac failure as given in Table 34-3. A noninvasively determined AT based on measurements of respiratory gas exchange, as discussed previously, corresponds to the invasively measured lactate threshold.7,12,16,19
O2max reflect the severity of chronic cardiac failure as given in Table 34-3. A noninvasively determined AT based on measurements of respiratory gas exchange, as discussed previously, corresponds to the invasively measured lactate threshold.7,12,16,19
Figure 34-6 Relationship between mixed venous lactate concentration and ![]() O2 to incremental treadmill exercise for patients with chronic cardiac failure of varying severity, as expressed by each functional class. (Reproduced with permission from Weber KT, Janicki JS. Cardiopulmonary exercise testing for evaluation of chronic cardiac failure. Am J Cardiol. 1987;55(2):22A–31A.)
O2 to incremental treadmill exercise for patients with chronic cardiac failure of varying severity, as expressed by each functional class. (Reproduced with permission from Weber KT, Janicki JS. Cardiopulmonary exercise testing for evaluation of chronic cardiac failure. Am J Cardiol. 1987;55(2):22A–31A.)
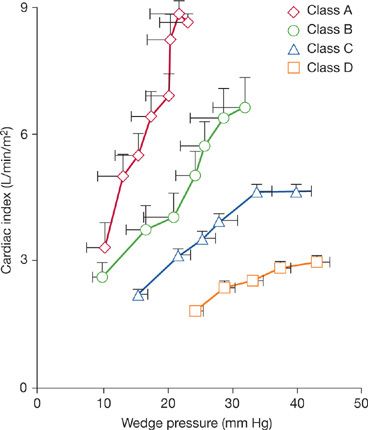
Exercise left ventricular filling pressure, as gauged from an occlusive wedge pressure recording, rises to a different degree in each exercise class with chronic cardiac failure (Fig. 34-7). In class A patients, the rise in wedge pressure during isotonic exercise rarely exceeds 18 mm Hg. This resembles a normal response. In class B patients, more dramatic elevations in exercise wedge pressure – to 25 mm Hg or higher – are frequently noted. Resting filling pressure is increased in class C and D patients; a further rise may be seen during upright exercise, often to levels in excess of 30 mm Hg. Despite these marked levels of pulmonary venous pressure, patients do not develop evidence of pulmonary congestion after exercise. Moreover, elevations in wedge pressure neither would predict exercise cardiac reserve and aerobic capacity nor are responsible for exertional dyspnea in these patients. Dyspnea corresponds with the lactate threshold and a disproportionate rise in ![]() E relative to
E relative to ![]() O2.8,16,20,21 Patients can be encouraged to exercise to exhaustion, attaining
O2.8,16,20,21 Patients can be encouraged to exercise to exhaustion, attaining ![]() O2max in the presence of dyspnea. In patients with acute cardiac failure, pulmonary congestion and dyspnea correlate with the elevation in wedge pressure; pulmonary edema occurs when hydrostatic pressure exceeds the colloidal osmotic pressure of 25 mm Hg and these patients should not be exercised.
O2max in the presence of dyspnea. In patients with acute cardiac failure, pulmonary congestion and dyspnea correlate with the elevation in wedge pressure; pulmonary edema occurs when hydrostatic pressure exceeds the colloidal osmotic pressure of 25 mm Hg and these patients should not be exercised.
Figure 34-7 Relationship between treadmill exercise cardiac index and occlusion wedge pressure in patients with chronic cardiac failure, subdivided according to functional class. (Reproduced with permission from Weber KT, Janicki JS. Cardiopulmonary exercise testing for evaluation of chronic cardiac failure. Am J Cardiol. 1988;55(2):22A–31A.)
![]() E rises appropriately during incremental exercise in patients with chronic cardiac failure. The response in
E rises appropriately during incremental exercise in patients with chronic cardiac failure. The response in ![]() E most closely corresponds to
E most closely corresponds to ![]() CO2 throughout exercise (aerobic and anaerobic work) and is sufficient to sustain alveolar ventilation, thereby preventing hypoxemia and hypercapnia. Maximum
CO2 throughout exercise (aerobic and anaerobic work) and is sufficient to sustain alveolar ventilation, thereby preventing hypoxemia and hypercapnia. Maximum ![]() E attained with exercise is less than 50% of MVV. Thus, these patients do not exhaust their ventilatory reserve in responding to exercise, even when their pulmonary compliance may be adversely elevated as a result of chronic pulmonary congestion and elevations in pulmonary venous pressure that appear with exercise. To minimize the work of breathing during exercise, class C and D patients use a pattern of rapid, shallow breathing to increase
E attained with exercise is less than 50% of MVV. Thus, these patients do not exhaust their ventilatory reserve in responding to exercise, even when their pulmonary compliance may be adversely elevated as a result of chronic pulmonary congestion and elevations in pulmonary venous pressure that appear with exercise. To minimize the work of breathing during exercise, class C and D patients use a pattern of rapid, shallow breathing to increase ![]() E. Thus, the rise in tidal volume during exercise above its resting value is modest and compatible with a substantial portion of each breath being wasted in ventilation of anatomic dead space.8 The response of class A and B patients more closely approximates that of healthy persons, in whom respiratory rate rises progressively during incremental exercise and the rise in tidal volume occurs early during the transition from rest to low-level exercise.
E. Thus, the rise in tidal volume during exercise above its resting value is modest and compatible with a substantial portion of each breath being wasted in ventilation of anatomic dead space.8 The response of class A and B patients more closely approximates that of healthy persons, in whom respiratory rate rises progressively during incremental exercise and the rise in tidal volume occurs early during the transition from rest to low-level exercise.
 DIASTOLIC DYSFUNCTION
DIASTOLIC DYSFUNCTION
In 30% or more of patients with symptomatic heart failure, primary diastolic dysfunction is held responsible. The ejection fraction is normal or only minimally impaired in these patients. Diastolic dysfunction relates to an inability of the left ventricle to accommodate left atrial and pulmonary venous blood flow during diastole without a marked increase in filling pressure. Abnormal diastolic relaxation and filling typically appear in patients with chronic ischemic heart disease (with previous myocardial infarction), in those with hypertensive heart disease, and especially in elderly women. Responsible mechanisms are thought to include abnormal tissue structure, as occurs with fibrous tissue accumulation or amyloid infiltration and abnormal calcium handling by sarcoplasmic reticulum. Factors extrinsic to the myocardium may also be contributory. Examples include the interaction between the pressure- or volume-overloaded right ventricle with the left ventricle and leftward shift of the interventricular septum and the interplay between the heart and pericardium.
Invasive CPX testing, together with incremental bicycle exercise, has been used to address the hemodynamic response of patients with primary diastolic dysfunction.22 Most patients studied had systemic hypertension, and many were elderly; all had a clinical history of pulmonary congestion. Peak exercise ![]() O2 was reduced, owing to a reduction in exercise cardiac output and stroke volume responses; arteriovenous O2 difference rose above 10 mL/dL. The level of
O2 was reduced, owing to a reduction in exercise cardiac output and stroke volume responses; arteriovenous O2 difference rose above 10 mL/dL. The level of ![]() O2 achieved with exercise correlated with peak cardiac output response. In comparison to age-matched controls, expected exercise-associated increments in left ventricular end-diastolic volume were not seen and were accompanied by increased left ventricular filling pressure. Thus, abnormalities in diastolic filling abrogated the Frank–Starling mechanism, thereby restricting the rise in exercise cardiac output; this finding may serve to explain symptoms of fatigue and breathlessness that these patients experience on exertion.
O2 achieved with exercise correlated with peak cardiac output response. In comparison to age-matched controls, expected exercise-associated increments in left ventricular end-diastolic volume were not seen and were accompanied by increased left ventricular filling pressure. Thus, abnormalities in diastolic filling abrogated the Frank–Starling mechanism, thereby restricting the rise in exercise cardiac output; this finding may serve to explain symptoms of fatigue and breathlessness that these patients experience on exertion.
Primary diastolic dysfunction has been observed in patients following cardiac transplantation23 in whom there is an abnormal blunting of stroke volume and heart rate responses to exercise. Despite the slower exercise heart rate in the transplanted, denervated heart, in which diastolic filling periods would accordingly be longer, diastolic dysfunction is present, limiting the exercise cardiac output response. Abnormal diastolic function has also been observed in the elderly and contributes to impaired exercise cardiac output response.24
 CHRONOTROPIC DYSFUNCTION
CHRONOTROPIC DYSFUNCTION
Cardiac reserve in exercise depends not only on systolic and diastolic functions but also on heart rate and rhythm, including a coordinated contraction of the atria and ventricles. CPX testing has been used to address the contribution of abnormal heart rate and rhythm on the AT and ![]() O2max, broadly categorized here as chronotropic dysfunction. This includes abnormal sinus tachycardia, bradyarrhythmias, atrioventricular dissociation, and atrial fibrillation. CPX testing has proved useful in the evaluation of pacemaker function and technique.25 Improvements in the AT at submaximal levels of work have been demonstrated for single-chamber, activity-triggered pacing compared with fixed-rate atrial or ventricular pacing. It too can help determine the optimum upper rate limit in heart failure patients with pacemakers. This can be determined by the highest pacing rate which still produces an increase in oxygen consumption.26 With recent advancements in pacemaker technology, cardiac resynchronization therapy (CRT) is being increasingly offered to patients with heart failure. In a study involving CRT patients undergoing CPX testing, significant increments in peak
O2max, broadly categorized here as chronotropic dysfunction. This includes abnormal sinus tachycardia, bradyarrhythmias, atrioventricular dissociation, and atrial fibrillation. CPX testing has proved useful in the evaluation of pacemaker function and technique.25 Improvements in the AT at submaximal levels of work have been demonstrated for single-chamber, activity-triggered pacing compared with fixed-rate atrial or ventricular pacing. It too can help determine the optimum upper rate limit in heart failure patients with pacemakers. This can be determined by the highest pacing rate which still produces an increase in oxygen consumption.26 With recent advancements in pacemaker technology, cardiac resynchronization therapy (CRT) is being increasingly offered to patients with heart failure. In a study involving CRT patients undergoing CPX testing, significant increments in peak ![]() O2,
O2, ![]() O2 at AT and all ventilation and metabolic parameters were noted. Patients with baseline
O2 at AT and all ventilation and metabolic parameters were noted. Patients with baseline ![]() O2 of less than 14 mL/min/kg had the most benefit.27 Similarly, patients with severe heart failure and atrial fibrillation had better hemodynamic performance with chronic biventricular pacing than left ventricular pacing alone.28
O2 of less than 14 mL/min/kg had the most benefit.27 Similarly, patients with severe heart failure and atrial fibrillation had better hemodynamic performance with chronic biventricular pacing than left ventricular pacing alone.28
Some patients with chronic cardiac failure deviate from this heart rate– ![]() O2 relation by having inappropriate sinus tachycardia, either at rest and throughout exercise, or simply during exercise. In the presence of reduced ejection fraction and ventricular dilation, this inappropriately rapid heart rate further compromises exercise cardiac output and reduces aerobic capacity. Under these circumstances, β-adrenergic receptor blockade is useful in attenuating the resting or exercise heart rate. Such chronotropic dysfunction (see below) to exercise may also apply to patients with chronic atrial fibrillation. An example of an inappropriate rapid heart rate during incremental treadmill exercise (Naughton protocol) is given in Figure 34-5 for a patient with atrial fibrillation and dilated cardiomyopathy of uncertain origin.29
O2 relation by having inappropriate sinus tachycardia, either at rest and throughout exercise, or simply during exercise. In the presence of reduced ejection fraction and ventricular dilation, this inappropriately rapid heart rate further compromises exercise cardiac output and reduces aerobic capacity. Under these circumstances, β-adrenergic receptor blockade is useful in attenuating the resting or exercise heart rate. Such chronotropic dysfunction (see below) to exercise may also apply to patients with chronic atrial fibrillation. An example of an inappropriate rapid heart rate during incremental treadmill exercise (Naughton protocol) is given in Figure 34-5 for a patient with atrial fibrillation and dilated cardiomyopathy of uncertain origin.29
 SURVIVAL AND PROGNOSIS
SURVIVAL AND PROGNOSIS
Various gas exchange parameters have been used to assess prognosis in patients with heart failure, including AT, peak ![]() O2, and
O2, and ![]() E/
E/![]() CO2 slope (a marker of ventilatory efficiency).13,30–33
CO2 slope (a marker of ventilatory efficiency).13,30–33 ![]() O2 at AT and
O2 at AT and ![]() CO2 slope are less subject to patient motivation or premature cessation of exercise and hence are more useful parameters. Class D patients with little or no exercise cardiac reserve (see Table 34-3) with AT <8 mL/min/kg have a marked reduction in 1- and 2-year survival as contrasted to Class A and B patients with respective exercise cardiac index responses of >8 and 6 to 8 L/min/m2.13,30,31
CO2 slope are less subject to patient motivation or premature cessation of exercise and hence are more useful parameters. Class D patients with little or no exercise cardiac reserve (see Table 34-3) with AT <8 mL/min/kg have a marked reduction in 1- and 2-year survival as contrasted to Class A and B patients with respective exercise cardiac index responses of >8 and 6 to 8 L/min/m2.13,30,31 ![]() O2 at AT combined with
O2 at AT combined with ![]() E/
E/![]() CO2 is a better prognostic indicator than peak
CO2 is a better prognostic indicator than peak ![]() O2 alone.34 A low peak PaCO2 with exercise is responsible for the prognostic power of
O2 alone.34 A low peak PaCO2 with exercise is responsible for the prognostic power of ![]() E/
E/![]() CO2 slope and by itself is also an independent predictor of prognosis.35 Resting end-tidal CO2 has been shown to be a predictor of cardiac-related events.36 Another
CO2 slope and by itself is also an independent predictor of prognosis.35 Resting end-tidal CO2 has been shown to be a predictor of cardiac-related events.36 Another ![]() O2 kinetics parameter that is a strong predictor of survival and less dependent on motivation is the mean response time (
O2 kinetics parameter that is a strong predictor of survival and less dependent on motivation is the mean response time (![]() O2 deficit/Δ
O2 deficit/Δ ![]() O2).37 In the recovery period, slow normalization of
O2).37 In the recovery period, slow normalization of ![]() O2 is associated with poor prognosis.38
O2 is associated with poor prognosis.38
The fluctuations in breathing patterns and its association with prognosis have also been studied in heart failure. Cyclic fluctuations in ![]() E at rest that persist during effort (external oscillatory ventilation) are associated with poor prognosis, whereas oscillations at rest alone are not.39–41
E at rest that persist during effort (external oscillatory ventilation) are associated with poor prognosis, whereas oscillations at rest alone are not.39–41
 EFFICACY OF MEDICATIONS
EFFICACY OF MEDICATIONS
The response to various heart failure medications on gas exchange and hemodynamics has been studied. Patients with heart failure taking spironolactone had a significant increase in peak oxygen consumption, DLCO, and membrane diffusing capacity.42,43 In a study in which losartan was added to an ACE inhibitor, there was a significant increase in peak ![]() O2 and exercise capacity;44 however, in another study in which candesartan was added, there was no increase in the peak
O2 and exercise capacity;44 however, in another study in which candesartan was added, there was no increase in the peak ![]() O2 or exercise capacity.45 Peak
O2 or exercise capacity.45 Peak ![]() O2 has been used as a prognostic marker in heart failure, but in a study involving patients with chronic heart failure taking carvedilol, peak
O2 has been used as a prognostic marker in heart failure, but in a study involving patients with chronic heart failure taking carvedilol, peak ![]() O2 was not found to be a useful prognostic marker.46
O2 was not found to be a useful prognostic marker.46
 EXERCISE TRAINING
EXERCISE TRAINING
Exercise training leads to an improvement in exercise tolerance and peak ![]() O2 in patients with heart failure who have left ventricular dysfunction.47 Exercise training in moderate stable heart failure results in favorable qualitative, rather than quantitative, changes in skeletal muscle.48 Correction of maximum oxygen uptake for skeletal muscle mass, rather than total body mass, is a more sensitive measure of changes associated with exercise training.48 Only progressive/increasing workload seems to markedly improve oxygen uptake.49
O2 in patients with heart failure who have left ventricular dysfunction.47 Exercise training in moderate stable heart failure results in favorable qualitative, rather than quantitative, changes in skeletal muscle.48 Correction of maximum oxygen uptake for skeletal muscle mass, rather than total body mass, is a more sensitive measure of changes associated with exercise training.48 Only progressive/increasing workload seems to markedly improve oxygen uptake.49
 ISCHEMIC HEART DISEASE
ISCHEMIC HEART DISEASE
Ischemia can be diagnosed with the help of ST segment changes during incremental exercise on treadmill or an ergometer during CPX testing. The sensitivity and specificity of ST changes for ischemia is not high. Parameters of gas exchange on CPX testing can be used to improve the diagnostic ability of the exercise-induced ST changes. Using O2 pulse flattening duration and Δ ![]() O2/Δ work rate slope with ST changes, the sensitivity and specificity for diagnosing ischemia improved from 46% to 66%, respectively, to 87% and 74%, respectively.50 Myocardial stress/rest scintigraphy was used as the standard for detecting ischemia in this study. In another study, exercise cardiac output estimated from
O2/Δ work rate slope with ST changes, the sensitivity and specificity for diagnosing ischemia improved from 46% to 66%, respectively, to 87% and 74%, respectively.50 Myocardial stress/rest scintigraphy was used as the standard for detecting ischemia in this study. In another study, exercise cardiac output estimated from ![]() O2 at AT correlated with multivessel coronary artery disease, adverse cardiac events and clinically driven revascularization.51 In postmyocardial infarction patients undergoing 3 weeks of exercise training, a significant improvement in
O2 at AT correlated with multivessel coronary artery disease, adverse cardiac events and clinically driven revascularization.51 In postmyocardial infarction patients undergoing 3 weeks of exercise training, a significant improvement in ![]() O2 was found.52
O2 was found.52
Parameters of gas exchange measured during CPX testing have proven useful in the diagnosis of ischemia. Myocardial ischemia produces transient depression of left ventricular systolic function and, consequently, a reduction in stroke volume and cardiac output. Graphically, this decline in cardiac output should be reflected in a decline in the rate of increase in oxygen consumption per workload (Δ ![]() O2/Δ Work Rate [WR] in watts). Thus, a blunted slope of Δ
O2/Δ Work Rate [WR] in watts). Thus, a blunted slope of Δ ![]() O2/Δ WR should reflect myocardial ischemia. This analysis of gas exchange has been compared to SPECT myocardial scintigraphy. CPX testing demonstrates improved sensitivity and specificity in the detection of myocardial ischemia. Another valid parameter for the detection of myocardial ischemia is a flattening of the graph of O2 pulse (mL/beat).50 As is the case with heart failure, peak
O2/Δ WR should reflect myocardial ischemia. This analysis of gas exchange has been compared to SPECT myocardial scintigraphy. CPX testing demonstrates improved sensitivity and specificity in the detection of myocardial ischemia. Another valid parameter for the detection of myocardial ischemia is a flattening of the graph of O2 pulse (mL/beat).50 As is the case with heart failure, peak ![]() O2 and AT are prognosticators of adverse prognosis in patients with ischemic heart disease.
O2 and AT are prognosticators of adverse prognosis in patients with ischemic heart disease.
CPX testing has been utilized to assess the presence of right ventricular dysfunction in patients following an inferior myocardial infarction. Right ventricular dysfunction was analyzed utilizing echocardiographic parameters. Patients with right ventricular infarction had significantly lower oxygen consumption than those who had suffered an inferior infarction without right ventricular involvement.53
CHRONIC CIRCULATORY FAILURE
Circulatory failure, in physiological terms, refers to an inability of the heart to raise its cardiac output in a manner commensurate with prevailing ![]() O2. Responsible factors are extrinsic to the myocardium and include such entities as valvular heart disease, intrinsic pulmonary vascular disease, pericardial disease, and anemia.
O2. Responsible factors are extrinsic to the myocardium and include such entities as valvular heart disease, intrinsic pulmonary vascular disease, pericardial disease, and anemia.
 VALVULAR HEART DISEASE
VALVULAR HEART DISEASE
Mitral or aortic valve disease may alter the functional integrity of the cardiopulmonary unit by impairing the heart’s ability to increase cardiac output in accordance with ![]() O2. Pathophysiological alterations within the unit that result from chronic valvular disease and that determine the clinical course and outcome following valve replacement include right heart overload and structural remodeling of the pulmonary vasculature and lung interstitium. The more marked the preoperative impairment in cardiac reserve, the poorer the long-term prognosis. Similarly, the greater the elevation in pulmonary vascular resistance, the more delayed is its return to normal levels and the slower the postoperative abatement of symptoms. The decision for surgical intervention requires an assessment of cardiopulmonary status—one that can be assessed noninvasively and monitored over time to detect a decline in cardiac reserve. Noninvasive CPX testing serves this purpose. Because of the heightened risk of syncope and the myocardial ischemia and arrhythmias that can occur during exercise in patients with aortic valvular stenosis, these patients should exercise with extreme caution, if at all.
O2. Pathophysiological alterations within the unit that result from chronic valvular disease and that determine the clinical course and outcome following valve replacement include right heart overload and structural remodeling of the pulmonary vasculature and lung interstitium. The more marked the preoperative impairment in cardiac reserve, the poorer the long-term prognosis. Similarly, the greater the elevation in pulmonary vascular resistance, the more delayed is its return to normal levels and the slower the postoperative abatement of symptoms. The decision for surgical intervention requires an assessment of cardiopulmonary status—one that can be assessed noninvasively and monitored over time to detect a decline in cardiac reserve. Noninvasive CPX testing serves this purpose. Because of the heightened risk of syncope and the myocardial ischemia and arrhythmias that can occur during exercise in patients with aortic valvular stenosis, these patients should exercise with extreme caution, if at all.
Incompetence of the mitral and aortic valves is an example of a disorder that can result in chronic circulatory failure. Each creates a volume overload on the left ventricle. The onset of ventricular dysfunction is generally unpredictable and may initially appear only during vigorous levels of physical activity. As dysfunction progresses, symptoms appear at lower levels of activity and, finally, at rest.
Resting cardiac output is often not distinguishable among class A, B, C, or D patients with mitral or aortic regurgitation. Cardiac reserve is reduced, however, and, accordingly, so is aerobic capacity.2 No impairment in systemic O2 extraction has been reported. Thus, as in chronic cardiac failure, any observed decrease in aerobic capacity must be due to a decline in maximal forward cardiac output. To the extent that cardiac output can rise, the exercise cardiac output– ![]() O2 relation is preserved among these classes, averaging 600 mL/min/m2 for every dL/min/m2 rise in
O2 relation is preserved among these classes, averaging 600 mL/min/m2 for every dL/min/m2 rise in ![]() O2. Responses in cardiac output and wedge pressure for each exercise class are given in Figure 34-8. As in chronic cardiac failure, marked elevations in wedge pressure are seen in class C and D patients; this is also true for class B patients with mitral or aortic regurgitation. However, these patients do not develop clinical evidence of pulmonary congestion following exercise, and dyspnea correlates with the lactate threshold. Exercise wedge pressure does not presage aerobic capacity or functional class in these patients.
O2. Responses in cardiac output and wedge pressure for each exercise class are given in Figure 34-8. As in chronic cardiac failure, marked elevations in wedge pressure are seen in class C and D patients; this is also true for class B patients with mitral or aortic regurgitation. However, these patients do not develop clinical evidence of pulmonary congestion following exercise, and dyspnea correlates with the lactate threshold. Exercise wedge pressure does not presage aerobic capacity or functional class in these patients.
Figure 34-8 Relationship between treadmill exercise cardiac index and wedge pressure in patients with chronic mitral or aortic regurgitation, divided according to functional class. (Reproduced with permission from Weber KT, Janicki JS (eds). Cardiopulmonary Exercise Testing: Physiologic Principles and Clinical Applications. Philadelphia: WB Saunders; 1986.)
AT can be used as an alternative measure in patients with valvular incompetence who are unable to attain ![]() O2max. The lactate threshold occurs at 60% to 70% of the patient’s aerobic capacity and corresponds to a level of systemic O2 extraction of 60% or more. Figure 34-9 depicts the response in mixed venous lactate concentration as a function of O2 for each exercise class with mitral or aortic regurgitation. As in patients with chronic cardiac failure, the lactate threshold occurs at progressively lower levels of work as the severity of valvular disease increases.2 The invasively measured lactate threshold correlates well with the valve obtained using noninvasive respiratory gas exchange measurements (see above).
O2max. The lactate threshold occurs at 60% to 70% of the patient’s aerobic capacity and corresponds to a level of systemic O2 extraction of 60% or more. Figure 34-9 depicts the response in mixed venous lactate concentration as a function of O2 for each exercise class with mitral or aortic regurgitation. As in patients with chronic cardiac failure, the lactate threshold occurs at progressively lower levels of work as the severity of valvular disease increases.2 The invasively measured lactate threshold correlates well with the valve obtained using noninvasive respiratory gas exchange measurements (see above).
Figure 34-9 Relationship between mixed venous lactate concentration and ![]() O2 observed during incremental treadmill exercise in patients with chronic mitral or aortic regurgitation. As in chronic cardiac failure, the lactate threshold (lactate >12 mg/dL) occurs at different levels of
O2 observed during incremental treadmill exercise in patients with chronic mitral or aortic regurgitation. As in chronic cardiac failure, the lactate threshold (lactate >12 mg/dL) occurs at different levels of ![]() O2, depending on functional class. (Reproduced with permission from Weber KT, Janicki JS (eds). Cardiopulmonary Exercise Testing: Physiologic Principles and Clinical Applications. Philadelphia: WB Saunders; 1986.)
O2, depending on functional class. (Reproduced with permission from Weber KT, Janicki JS (eds). Cardiopulmonary Exercise Testing: Physiologic Principles and Clinical Applications. Philadelphia: WB Saunders; 1986.)
The reduced mitral valve orifice that accompanies rheumatic mitral valvular stenosis leads to left atrial chamber enlargement, pulmonary venous hypertension, and right heart pressure overload. Pulmonary vascular resistance in most patients ranges between 200 and 600 dynes·s·cm–5. Mitral stenosis is responsible for reduced left ventricular filling at rest and during exercise. An exercise-associated rise in heart rate reduces the diastolic filling period to further curtail left ventricular filling.
Cardiac output fails to rise appropriately with exercise in patients with chronic circulatory failure due to mitral stenosis.2 For most symptomatic patients, cardiac output fails to rise appropriately during symptom-limited exercise because of a limited stroke volume response. Systemic O2 extraction increases markedly with exercise, as do pulmonary capillary wedge and mean pulmonary artery pressures. Preoperative assessment of mitral stenosis should include not only calculation of mitral valve area but also exercise test-determined cardiac reserve and functional status. A decision regarding surgery should be based on these objective measures and clinical judgment, not simply on a laboratory-based calculation of reduced valve area.
A preoperative assessment of valvular surgery patients by CPX testing can help predict the degree of postoperative recovery. Preoperative peak ![]() O2 of 19 mL/min/kg and greater in patients undergoing surgery for mitral and aortic regurgitation correlates with higher percentage of patients attaining NYHA Functional Class I at 1 year after surgery.54,55
O2 of 19 mL/min/kg and greater in patients undergoing surgery for mitral and aortic regurgitation correlates with higher percentage of patients attaining NYHA Functional Class I at 1 year after surgery.54,55 ![]() O2max along with AT has been used to follow progress of postoperative rehabilitation and training in valve surgery patients.56,57 Exercise parameters can be helpful in assessing patients with valvular disease patients in whom there is a discrepancy between symptoms and echocardiographic data. A
O2max along with AT has been used to follow progress of postoperative rehabilitation and training in valve surgery patients.56,57 Exercise parameters can be helpful in assessing patients with valvular disease patients in whom there is a discrepancy between symptoms and echocardiographic data. A ![]() O2max less than 75% predicted in moderate-to-severe mitral stenosis correlates with higher transvalvular gradients and higher pulmonary artery pressures at the end of exercise than does a
O2max less than 75% predicted in moderate-to-severe mitral stenosis correlates with higher transvalvular gradients and higher pulmonary artery pressures at the end of exercise than does a ![]() O2max greater than 75% of max predicted.58
O2max greater than 75% of max predicted.58
In the absence of pulmonary arterial hypertension in patients undergoing mitral valve replacement, the presence of significant tricuspid regurgitation is associated with lower ![]() O2max and AT and steeper
O2max and AT and steeper ![]() E/
E/![]() CO2 than when significant tricuspid regurgitation is absent.59
CO2 than when significant tricuspid regurgitation is absent.59
 HYPERTROPHIC CARDIOMYOPATHY
HYPERTROPHIC CARDIOMYOPATHY
Hypertrophic cardiomyopathy, the best known genetically determined cardiovascular condition, is marked by significant left ventricular hypertrophy, which may be asymmetric, concentric, or localized (apical). Two major clinical concerns in the disorder are the risk of sudden death and the difficulty in making the diagnosis when the classic asymmetric septal hypertrophy and the classic hemodynamic findings are absent. Exercise testing, in general, is a simple, but imprecise, method to screen for malignant dysrhythmias. CPX testing has been useful in distinguishing the physiologic left ventricular hypertrophy of athletes from hypertrophic cardiomyopathy.60 Peak ![]() O2 is significantly higher in the athletes. A peak
O2 is significantly higher in the athletes. A peak ![]() O2 20% above predicted
O2 20% above predicted ![]() O2max separates this group from those with genetically proven hypertrophic cardiomyopathy. A number of patients with hypertrophic cardiomyopathy experience symptoms of fatigue and dyspnea which do not correlate with hemodynamic findings. CPX testing can separate real from perceived symptoms. The percent predicted peak
O2max separates this group from those with genetically proven hypertrophic cardiomyopathy. A number of patients with hypertrophic cardiomyopathy experience symptoms of fatigue and dyspnea which do not correlate with hemodynamic findings. CPX testing can separate real from perceived symptoms. The percent predicted peak ![]() O2, combined with a quality of life questionnaire, appears to be helpful in making this distinction.61
O2, combined with a quality of life questionnaire, appears to be helpful in making this distinction.61
 OBSTRUCTIVE SLEEP APNEA
OBSTRUCTIVE SLEEP APNEA
CPX testing can safely be performed in patients with sleep apnea to evaluate abnormalities in gas exchange and the response to continuous positive airway pressure therapy. Patients with moderate or severe obstructive sleep apnea have impaired exercise capacity, low peak ![]() O2, and low AT.62 The abnormal parameters on CPX testing can be improved with continuous positive airway pressure therapy. In one study involving severe sleep apnea, 2 months of nasal continuous positive airway pressure treatment resulted in higher right ventricular ejection fraction, peak
O2, and low AT.62 The abnormal parameters on CPX testing can be improved with continuous positive airway pressure therapy. In one study involving severe sleep apnea, 2 months of nasal continuous positive airway pressure treatment resulted in higher right ventricular ejection fraction, peak ![]() O2, peak
O2, peak ![]() O2, AT, and oxygen pulse.63
O2, AT, and oxygen pulse.63
Patients with heart failure who have central sleep apnea have an increased mortality rate compared to those without it. Patients with heart failure often lack the classical symptoms of central sleep apnea and, hence, its presence may be underestimated. Treating central sleep apnea in heart failure has beneficial effects on cardiac function. Patients with heart failure with central sleep apnea have a highly augmented ventilatory response to exercise. This is manifested by a significantly increased slope of ![]() E/
E/![]() CO2, which correlates with the severity of sleep apnea. Thus, patients with heart failure who have an increased
CO2, which correlates with the severity of sleep apnea. Thus, patients with heart failure who have an increased ![]() E/
E/![]() CO2 slope should be considered for a full sleep study to confirm the presence of sleep apnea.64
CO2 slope should be considered for a full sleep study to confirm the presence of sleep apnea.64
 CONGENITAL HEART DISEASE
CONGENITAL HEART DISEASE
Patients with cyanotic congenital heart disease have limitations in exercise tolerance. CPX testing can be used to objectively assess their exercise limitation and ventilatory efficiency.
In a study of 25 adults with uncorrected cyanotic congenital heart disease, peak oxygen uptake and PaO2 were significantly reduced compared with normal subjects, while PaCO2 was only slightly reduced. Ventilatory efficiency, expressed as ![]() E/
E/![]() CO2, was found to be markedly impaired at rest and during exercise.
CO2, was found to be markedly impaired at rest and during exercise. ![]() E/
E/![]() CO2 correlated more strongly with patients’ symptoms than hypoxemia and peak oxygen uptake.65 For the corresponding NYHA class, patients with adult congenital heart disease and patients with heart failure had no significant differences in peak
CO2 correlated more strongly with patients’ symptoms than hypoxemia and peak oxygen uptake.65 For the corresponding NYHA class, patients with adult congenital heart disease and patients with heart failure had no significant differences in peak ![]() O2.66
O2.66
CPX testing has also been used for assessing the response to transcatheter closure of atrial septal defect in adult patients. Improved peak oxygen uptake, peak oxygen pulse, and vital capacity have been reported following closure.67 In one study, improvements in the prolonged ![]() O2 slope and
O2 slope and ![]() CO2 slope were noted, reflecting improvement in recovery from maximal exercise.68
CO2 slope were noted, reflecting improvement in recovery from maximal exercise.68
Tricuspid valve surgery in patients with Ebstein’s anomaly produces reduced right ventricular volumes, increased pulmonary blood flow, and increased left ventricular filling and cardiac output. This is reflected by an increased ![]() O2max.69
O2max.69
 PULMONARY HYPERTENSION
PULMONARY HYPERTENSION
Pulmonary hypertension is expressed as an abnormal elevation in resting or exercise pulmonary artery pressure (see Chapter 72). Chronic left heart failure with attendant elevated left atrial pressure remains the most common cause of pulmonary venous hypertension. Pulmonary arterial hypertension (PAH) accompanies intrinsic pulmonary vascular disease or arteriolar vasoconstriction associated with hypoxemia due to intrinsic lung disease. PAH creates right ventricular pressure overload and an impediment to left ventricular filling. Accordingly, exercise cardiac output is compromised and aerobic capacity declines. PAH represents an example of chronic circulatory failure.
Patients with PAH have been studied with elective right heart catheterization using a triple-lumen flotation catheter and subsequent exercise testing.2 Resting and peak treadmill exercise hemodynamic responses are given in Table 34-4. At rest, right heart and pulmonary arterial pressures exceeded the normal range. Right ventricular systolic pressure at rest was in excess of 50 mm Hg, and in one-quarter of patients it approximated or exceeded left ventricular (and systemic arterial) systolic pressure. Resting wedge pressure was normal in these patients. Calculated pulmonary vascular resistance exceeded the upper range of normal (170 dynes·s·cm–5) in all patients; in more than one-third, it was above 1000 dynes·s·cm–5, approximating systemic vascular resistance.