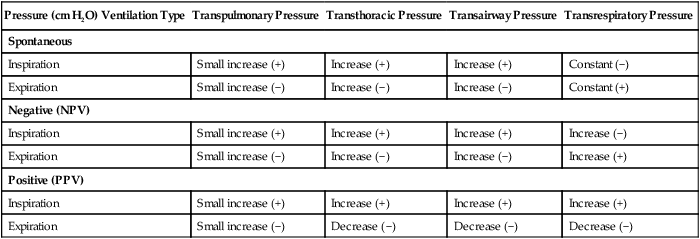
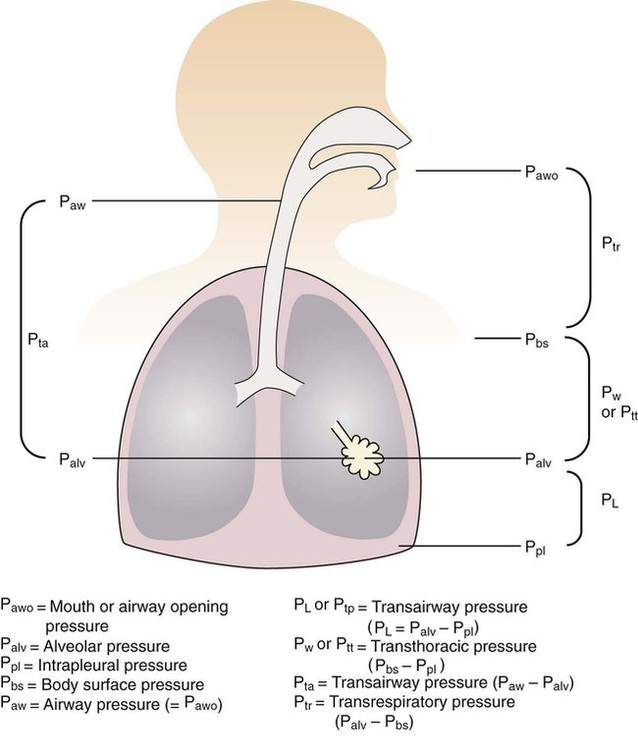
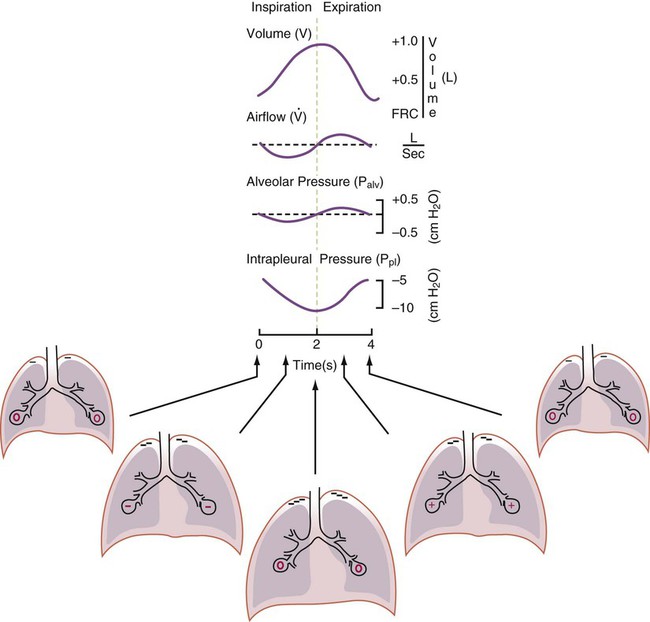
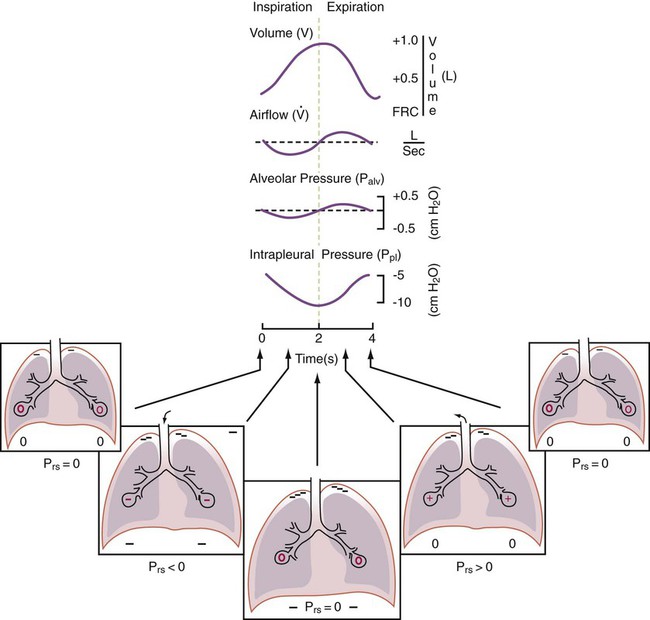
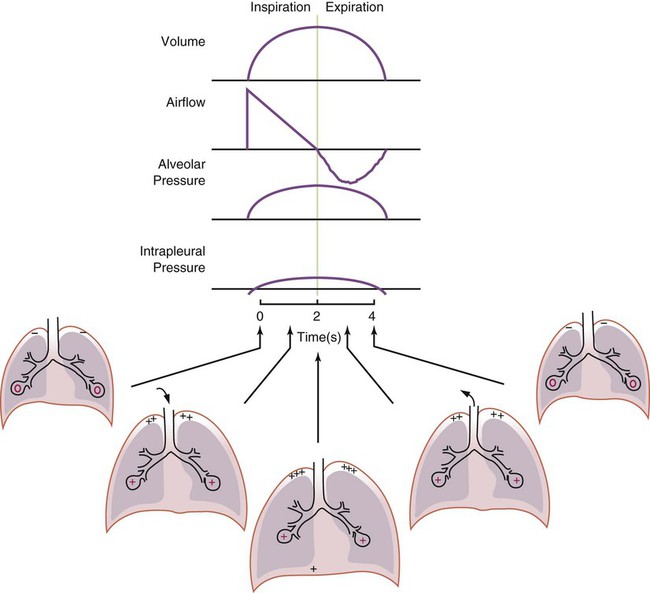
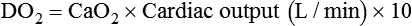After reading this chapter you will be able to: For gas to flow through the airway, a pressure gradient must exist. The airways begin at the mouth and end at the alveoli, so mouth pressure (pressure at the airway opening [Pawo]) and alveolar pressure (Palv) are important in describing gas flow, as are intrapleural pressure (Ppl) and body surface pressure or atmospheric pressure (Pbs). In addition, intraabdominal pressure (Pab) affects the impact of Ppl change on diaphragm movement. Ppl is the pressure in the pleural space, the virtual space between the visceral and parietal pleurae, and is usually negative in relation to Palv. Figure 43-1 shows a graphic model of the respiratory system with these pressures identified as points in space. Mathematical models relating pressure, volume, and flow corresponding to this graphic model are constructed using pressure differences. The various components of the graphic model are defined as everything that exists between these points in space. The respiratory system is everything that exists between the airway opening and the body surface. The associated pressure difference is transrespiratory pressure (Ptr), defined as Pawo − Pbs. The components of transrespiratory pressure correspond to the components of the graphic model. The airways are represented by transairway pressure (Pta), defined as Pawo − Palv. The lungs are represented by the transalveolar pressure: (PL = Palv − Ppl). The chest wall is represented by trans–chest wall pressure: (Ptcw = Ppl − Pbs). If the lungs and chest wall are lumped together, they can be represented by transthoracic pressure: (Ptt = Palv − Pbs). Another pressure gradient not defined in Figure 43-1 that also affects gas movement is the transdiaphragmatic pressure (Pdi). This pressure gradient is the difference between intraabdominal pressure and pleural pressure and affects diaphragmatic movement: (Ppl − Pab). Once these pressures and pressure gradients are understood, the differences between spontaneous ventilation, positive pressure ventilation (PPV), and negative pressure ventilation (NPV) become evident. Spontaneous breathing is normally an autonomic phenomenon. In other words, we do not think about breathing; it is controlled by the autonomic nervous system. Not until our breathing is stressed do we consider the effort to breathe or the energy expended. At end-exhalation, intrapleural pressure is slightly negative. Alveolar, mouth, and body surface pressures are zero. The diaphragm contracts in response to stimulation of the phrenic nerve via the respiratory center in the medulla of the brain. When the diaphragm contracts, it descends into the abdominal cavity, decreasing intrapleural pressure. When intrapleural pressure becomes more negative, alveolar pressure becomes negative as well. The effects of spontaneous breathing on the pressure gradients are shown in Table 43-1. Under normal circumstances, a decrease in intrapleural pressure results in decreased alveolar pressure, increased transairway pressure, and inspiration of the tidal volume (VT) (Figure 43-2). TABLE 43-1 Changes in Airway Pressure Gradients During Spontaneous, Negative, and Positive Pressure Ventilation VT and flow during spontaneous ventilation may be described by the equation of motion.1,2 The equation of motion describes the relationship between muscle pressure (analogous to pleural pressure in spontaneous breathing), compliance, resistance, flow, and volume as follows: Mechanical NPV is similar to spontaneous breathing. NPV decreases pleural pressure (Ppl) during inspiration by exposing the chest to subatmospheric pressure. Negative pressure at the body surface (Pbs) is transmitted first to the pleural space and then to the alveoli (Palv). Because the airway opening remains exposed to atmospheric pressure during NPV, a transairway pressure gradient is created. Gas flows from the relatively high pressure at the airway opening (zero) to the relatively low pressure in the alveoli (negative). As with spontaneous breathing, alveolar expansion during NPV is determined by the magnitude of the transpulmonary pressure gradient. During expiration in both spontaneous breathing and NPV, the lungs and chest wall passively recoil to their resting end expiratory levels. As this recoil occurs, pleural pressure becomes less negative, and alveolar pressure increases above atmospheric pressure (Figure 43-3). This increase in alveolar pressure reverses the transairway pressure gradient. As Palv becomes greater than Pawo, gas flows from the alveoli to the airway opening. The effects of NPV on the pressure gradients are shown in Table 43-1. PPV causes air to flow into the lungs because of an increase in airway pressure, not a decrease in pleural pressure as occurs during spontaneous breathing and NPV (Figure 43-4). However, similar to spontaneous breathing and NPV, PPV causes an increase in Ptp, which allows gas to flow into the lungs. Gas flows into the lungs because pressure at the airway opening (Pawo) is positive, and alveolar pressure (Palv) is initially zero or less positive. Alveolar pressure rapidly increases during the inspiratory phase of PPV. The increased alveolar pressure expands the airways and alveoli. Because alveolar pressure is greater than pleural pressure (Ppl) during PPV, positive pressure is transmitted from the alveoli to the pleural space, causing pleural pressure to increase during inspiration. Depending on the compliance and resistance of the lungs, pleural pressure may markedly exceed atmospheric pressure during a portion of inspiration. These changes in pleural pressure during PPV can lead to significant physiologic changes (see later section). Pressure gradients during PPV are similar to pressure gradients during spontaneous breathing and NPV except that they are created by a positive pressure at the airway opening instead of a negative pressure in the pleural space (see Table 43-1). All pressure gradients change in the same direction as during NPV and spontaneous breathing except the transrespiratory pressure, which changes in the opposite direction. Similar to spontaneous breathing, the recoil force of the lungs and chest wall, stored as potential energy during the positive pressure breath, causes passive exhalation. As gas flows from the alveoli to the airway opening, alveolar pressure decreases to atmospheric level, while pleural pressure is restored to its normal subatmospheric level (see Figure 43-4). Spontaneous ventilation results in gas distribution mainly to the dependent and peripheral zones of the lungs. PPV tends to reverse this normal pattern of gas distribution, and most of the delivered volume is directed to nondependent lung zones (Figure 43-5). This phenomenon is caused partly by the inactivity of the diaphragm and chest wall during PPV. Although these structures actively facilitate gas movement during spontaneous breathing, inactivity of these structures during PPV impedes ventilation to dependent lung zones. An increase in ventilation to the nondependent zones of the lung, where there is less perfusion, increases the ventilation/perfusion ( Bicarbonate has been used as therapy for metabolic acidosis. If it is administered, bicarbonate quickly combines with hydrogen ions and dissociates to form CO2 and water, a reaction that may increase WOB. Generally, bicarbonate administration is not recommended until acidosis is severe (pH < 7.2). When necessary, bicarbonate is administered according to the following formula:2 Metabolic alkalemia is defined as a normal PaCO2 with an elevated pH (>7.45) and an increased bicarbonate level (>26 mEq/L) and base excess (>+2 mEq/L). With metabolic alkalemia, in an effort to compensate for the increased pH, the patient tries to decrease minute ventilation. If weaning is attempted when the patient has a metabolic alkalemia, the patient may continue to hypoventilate, and weaning may fail. As with metabolic acidemia, the underlying cause should be determined and managed. Common causes of metabolic alkalosis include hypochloremia or hypokalemia secondary to gastrointestinal loss, diuretics, or steroid administration. See Chapter 13 for details on acid-base balance. When a mechanical ventilator is used to improve arterial oxygenation by increasing FiO2 or PEEP, CaO2 increases. However, the increase in CaO2 represents only part of tissue O2 delivery because O2 delivery is defined by CaO2 and cardiac output, as follows:2 where 10 is a constant for converting deciliters to milliliters. Normal tissue O2 delivery is approximately 990 ml/min because the normal CaO2 is approximately 20 vol%, and the normal cardiac output is approximately 5 L/min. When PaO2, CaO2, and cardiac output are adequate, so is tissue O2 delivery. When PEEP is needed to improve PaO2, it must be used cautiously because PEEP increases intrathoracic pressure. When intrathoracic pressure is increased, pleural pressure around the heart also increases, and the increase can affect the mechanical activity of the heart and impede venous return and decrease cardiac output. As discussed in Chapter 44, careful titration of PEEP must include monitoring the cardiovascular status of the patient. Optimal PEEP provides adequate arterial oxygenation and tissue O2 delivery.
Physiology of Ventilatory Support
 Discuss the pressures and pressure gradients that affect gas delivery during spontaneous breathing, negative pressure ventilation (NPV), and positive pressure ventilation (PPV).
Discuss the pressures and pressure gradients that affect gas delivery during spontaneous breathing, negative pressure ventilation (NPV), and positive pressure ventilation (PPV).
 Identify the effects of mechanical ventilation on oxygenation, ventilation, and lung mechanics.
Identify the effects of mechanical ventilation on oxygenation, ventilation, and lung mechanics.
 Describe the currently available modes of mechanical ventilation.
Describe the currently available modes of mechanical ventilation.
 Discuss the indications and physiologic effect of positive end expiratory pressure (PEEP).
Discuss the indications and physiologic effect of positive end expiratory pressure (PEEP).
 Describe the cardiovascular effects of PPV and NPV.
Describe the cardiovascular effects of PPV and NPV.
 Describe the effects of PPV on other body systems.
Describe the effects of PPV on other body systems.
 Identify and list the complications and hazards of providing mechanical ventilatory support.
Identify and list the complications and hazards of providing mechanical ventilatory support.
 Discuss how to minimize adverse effects of mechanical ventilation.
Discuss how to minimize adverse effects of mechanical ventilation.
Pressure and Pressure Gradients
Airway, Alveolar, and Intrathoracic Pressure, Volume, and Flow During Spontaneous Ventilation
Pressure (cm H2O) Ventilation Type
Transpulmonary Pressure
Transthoracic Pressure
Transairway Pressure
Transrespiratory Pressure
Spontaneous
Inspiration
Small increase (+)
Increase (+)
Increase (+)
Constant (−)
Expiration
Small increase (−)
Increase (−)
Increase (−)
Constant (+)
Negative (NPV)
Inspiration
Small increase (+)
Increase (+)
Increase (+)
Increase (−)
Expiration
Small increase (−)
Increase (−)
Increase (−)
Increase (+)
Positive (PPV)
Inspiration
Small increase (+)
Increase (+)
Increase (+)
Increase (+)
Expiration
Small increase (−)
Decrease (−)
Decrease (−)
Decrease (−)
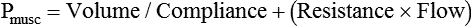
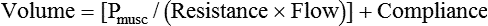
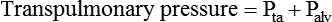
Airway, Alveolar, and Intrathoracic Pressure, Volume, and Flow During Negative Pressure Mechanical Ventilation
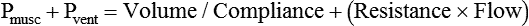
Airway, Alveolar, and Intrathoracic Pressure, Volume, and Flow During Positive Pressure Mechanical Ventilation
Effects of Mechanical Ventilation on Ventilation
Increased Minute Ventilation
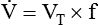
Decreased Ventilation/Perfusion Ratio
 ) ratio, effectively increasing physiologic dead space. The increase in P(A − a)O2 often observed with PPV is caused by areas of low
) ratio, effectively increasing physiologic dead space. The increase in P(A − a)O2 often observed with PPV is caused by areas of low 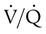 ratio.
ratio.
Changes in Acid-Base Balance
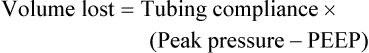
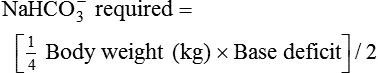
Effects of Mechanical Ventilation on Oxygenation
Increased Inspired Oxygen
Increased Tissue Oxygen Delivery
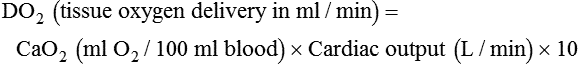

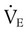 ) is the product of tidal volume (VT) and ventilatory rate (f):
) is the product of tidal volume (VT) and ventilatory rate (f):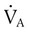 ) is inversely related to PaCO2 as defined by the following relationship:
) is inversely related to PaCO2 as defined by the following relationship: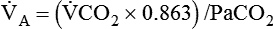
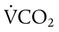 is carbon dioxide (CO2) production.
is carbon dioxide (CO2) production.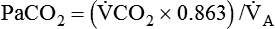
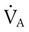 decreases or
decreases or 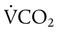 increases, PaCO2 increases, and hypercapnic respiratory failure follows; mechanical ventilation may be indicated in this setting. Because mechanical ventilation increases ventilation, PaCO2 can be decreased to the desired level depending on the total ventilatory rate.
increases, PaCO2 increases, and hypercapnic respiratory failure follows; mechanical ventilation may be indicated in this setting. Because mechanical ventilation increases ventilation, PaCO2 can be decreased to the desired level depending on the total ventilatory rate.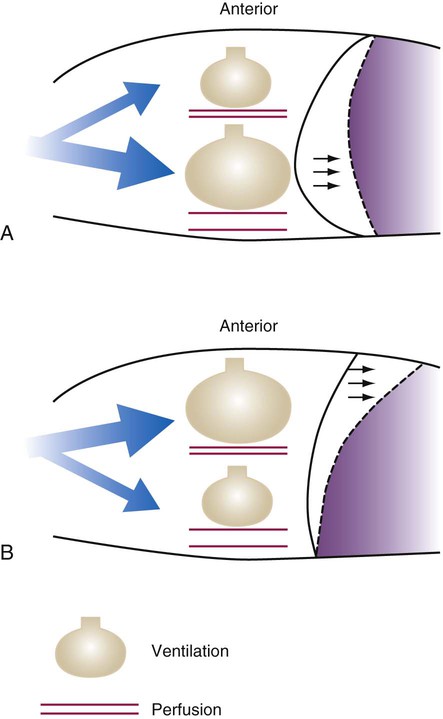
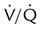 ratio. Partly because of diaphragmatic inactivity, PPV reverses this normal pattern of gas distribution, and most delivered volume is directed to the upper lung zones. B, An increase in ventilation to the upper lung zones, where there is less perfusion, increases the
ratio. Partly because of diaphragmatic inactivity, PPV reverses this normal pattern of gas distribution, and most delivered volume is directed to the upper lung zones. B, An increase in ventilation to the upper lung zones, where there is less perfusion, increases the 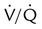 ratio, effectively increasing physiologic dead space. At the same time, higher alveolar pressure in the better ventilated upper lung zones diverts blood flow away from these areas to the areas receiving the least ventilation. The result is areas of low
ratio, effectively increasing physiologic dead space. At the same time, higher alveolar pressure in the better ventilated upper lung zones diverts blood flow away from these areas to the areas receiving the least ventilation. The result is areas of low 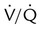 ratio and impaired oxygenation.
ratio and impaired oxygenation. 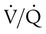 ratio in the bases and dependent lung zones mainly as a result of ventilation being primarily distributed to nondependent lung zones. The
ratio in the bases and dependent lung zones mainly as a result of ventilation being primarily distributed to nondependent lung zones. The 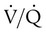 ratio is also decreased in nondependent lung zones because of the effect of PPV on perfusion. PPV can compress the pulmonary capillaries. This compression increases pulmonary vascular resistance and decreases perfusion. Minimal blood
ratio is also decreased in nondependent lung zones because of the effect of PPV on perfusion. PPV can compress the pulmonary capillaries. This compression increases pulmonary vascular resistance and decreases perfusion. Minimal blood 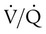 ratio in those areas and increases the P(A − a)O2.
ratio in those areas and increases the P(A − a)O2.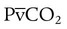 of 45 mm Hg. Under normal circumstances, CO2 moves out of the blood at the pulmonary capillary interface; the result is a PaCO2 of 40 mm Hg. In the event of a decrease in alveolar ventilation or an increase in CO2 production, PaCO2 increases. Mechanical ventilation can increase minute volume and alveolar ventilation and reduce P
of 45 mm Hg. Under normal circumstances, CO2 moves out of the blood at the pulmonary capillary interface; the result is a PaCO2 of 40 mm Hg. In the event of a decrease in alveolar ventilation or an increase in CO2 production, PaCO2 increases. Mechanical ventilation can increase minute volume and alveolar ventilation and reduce P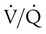 ratio. In emphysema, formation of bullae is coincident with the destruction of pulmonary capillaries; the result is large areas of poorly perfused but ventilated alveoli. Pulmonary emboli may completely occlude pulmonary vessels; the result is lack of perfusion to alveoli distal to the blockage.
ratio. In emphysema, formation of bullae is coincident with the destruction of pulmonary capillaries; the result is large areas of poorly perfused but ventilated alveoli. Pulmonary emboli may completely occlude pulmonary vessels; the result is lack of perfusion to alveoli distal to the blockage.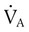 ) are inadequate to meet the needs of the body. Respiratory acidemia can occur when the VT is low, even though an accompanying mandatory rate is high.
) are inadequate to meet the needs of the body. Respiratory acidemia can occur when the VT is low, even though an accompanying mandatory rate is high.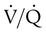 ratio or hypoventilation is more responsive to increased FiO2 than hypoxemia caused by a diffusion defect or shunt. Hypoxemia caused by hypoventilation responds well to an increase in FiO2, but alveolar ventilation can be restored only by improved ventilation. Hypoxemia caused by diffusion defect and shunt generally respond better to an increase in PEEP than to an increase in FiO2. The fact that PaO2 responds well to increased FiO2 generally indicates that a low
ratio or hypoventilation is more responsive to increased FiO2 than hypoxemia caused by a diffusion defect or shunt. Hypoxemia caused by hypoventilation responds well to an increase in FiO2, but alveolar ventilation can be restored only by improved ventilation. Hypoxemia caused by diffusion defect and shunt generally respond better to an increase in PEEP than to an increase in FiO2. The fact that PaO2 responds well to increased FiO2 generally indicates that a low 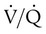 ratio is the cause of hypoxemia. If the patient is receiving mechanical ventilation and has adequate alveolar ventilation, failure of the PaO2 to respond to increased FiO2 likely means that hypoxemia is due to a diffusion defect or shunt.
ratio is the cause of hypoxemia. If the patient is receiving mechanical ventilation and has adequate alveolar ventilation, failure of the PaO2 to respond to increased FiO2 likely means that hypoxemia is due to a diffusion defect or shunt.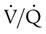 ratio. In the event that PaO2 is not restored by an increase in FiO2, hypoxemia is probably due to a diffusion defect or shunt.
ratio. In the event that PaO2 is not restored by an increase in FiO2, hypoxemia is probably due to a diffusion defect or shunt.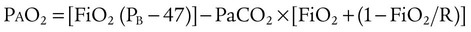
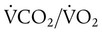 ), normally 0.8.
), normally 0.8.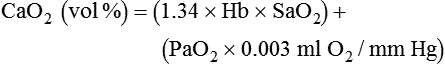
 matching and arterial oxygenation.
matching and arterial oxygenation.