Percutaneous Approach, Including Transseptal and Apical Puncture
Claudia A. Martinez
Mauro Moscucci
Dr Donald S. Baim and Dr Daniel I. Simon authored this chapter in the prior edition.
In contrast to other cutdown techniques (see Chapter 8), the percutaneous approach to left and right heart catheterization uses a needle puncture to achieve vascular access1 and thus obviates the need for surgical isolation of the vessels during either the insertion or the subsequent removal of cardiac catheters. Once the needle has been positioned within the vessel lumen, a flexible guidewire is advanced through the needle and well into the vessel being accessed.2 This guidewire remains in an intravascular position as the needle is withdrawn to allow direct insertion of an appropriately sized sheath that is equipped with a back-bleed valve and a side-arm port. The desired catheters can then be placed through the sheath and into the vasculature.3,4 This modification reduces patient discomfort and eliminates repetitive local arterial trauma during catheter exchanges, although it does increase the size of the puncture slightly since the outer diameter of the sheath is 1F size (0.33 mm) larger than the corresponding bare catheter. At the termination of the percutaneous catheterization procedure, the catheters and introducing sheaths are withdrawn, and bleeding from the puncture sites is controlled by the application of direct pressure or the use of one of several vascular closure devices (see below).
Because of its speed and simplicity, percutaneous entry via the femoral approach continues to be the dominant approach to cardiac catheterization. More than 90% of the procedures contained in the 2011 CathPCI Registry of the National Cardiovascular Data Registry were performed via this route.5 With appropriate skills and knowledge of regional anatomy, moreover, the same percutaneous techniques used for femoral artery and vein cannulation can be adapted to allow catheter insertion from a variety of other entry sites. Venous catheterization can thus be performed via the internal jugular, subclavian, or median antecubital vein, whereas arterial catheterization can be performed via the brachial, axillary or radial arteries, or even via the lumbar aorta.
CATHETERIZATION VIA THE FEMORAL ARTERY AND VEIN
Patient Preparation
After palpation of the femoral arterial pulse within the inguinal area, a safety razor is used to shave an area approximately 10 cm in diameter surrounding this point. Although most catheterizations can be performed quickly and easily from a single groin, it is generally routine to have both groins prepared. The right groin is commonly used, since it is more easily accessed by the operator standing on that side of the table. If difficulties in catheter advancement force a switch to the other groin once the procedure has begun, however, having the left groin already prepared saves time and inconvenience. In the past, the shaved areas were traditionally scrubbed with a povidone-iodine/detergent mixture and then painted with povidone-iodine solution. More recently, most laboratories (including ours) have replaced povidone-iodine solution with chlorhexidine-alcohol-based antiseptic preparations, which have been shown to be more effective and less irritant.6 The patient is then draped from clavicles to below the feet, leaving only the sterile prepared groin areas exposed. Most laboratories now use disposable paper drapes with adhesive-bordered apertures for this purpose, frequently packaged together with other disposable supplies (syringes, needles, bowls, and so on) in a custom kit available from any of several vendors.
Selection of Puncture Site
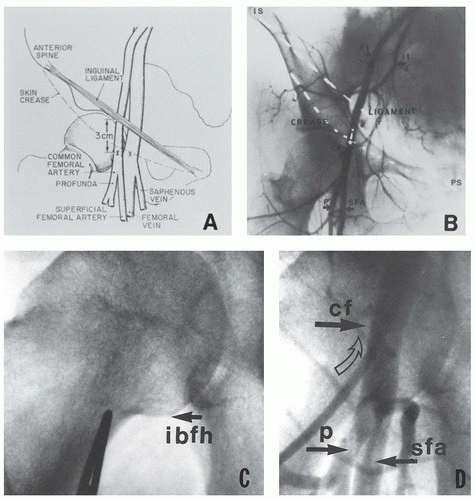
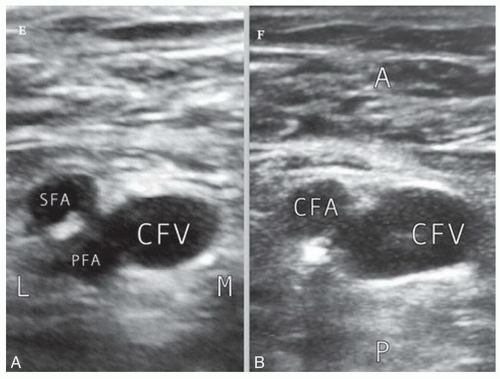
The adjacent common femoral artery and vein (Figure 6.1A and B) are the most commonly used vessels for percutaneous diagnostic cardiac catheterization.5 It is important to puncture these vessels at the correct level, that is, at the mid common femoral artery, above the arterial bifurcation into the profunda and superficial femoral artery and 1 or 2 cm below the inguinal ligament. Identification of the correct puncture site facilitates vessel entry and effective compression to minimize local vascular complications. The position of the skin crease itself is not a reliable marker for the puncture site and it can instead be misleading in obese patients. The appropriate localization of the skin puncture should be first identified by fluoroscopic landmarks; a radiopaque marker (i.e., mosquito forceps which routinely come within the sterile instrument package) should be placed on top of the inguinal area where the pulse appears stronger, and adjusted by fluoroscopy in the anteroposterior view so that it is positioned overlying the inferior quadrant of the femoral head (Figure 6.1C and D). Making the skin nicks at this level increases the chance that needle puncture will take place in the common femoral segment (overlying the middle of the femoral head) rather than either too high (above the inguinal ligament) or too low (in the superficial femoral or profunda branches of the common femoral artery). The femoral artery should be easily palpable over a several-centimeter span above and below the skin site. The femoral vein will lie approximately one fingerbreadth medial to the artery along a parallel course. It should be recognized that there are anatomical variations to this course. Thus, more recently the use of intraprocedure vascular ultrasound guidance has emerged as an alternative method to identify each vessel and select the puncture site. After initial fluoroscopic delineation of the landmarks, using a sterile plastic cover, the ultrasound vascular probe is positioned over the strongest pulse area to scan the femoral artery and vein (Figure 6.2A and B). The femoral artery appears as a pulsatile circle, usually with thicker and brighter delineation of the walls and on occasion with atherosclerotic and calcific plaques. Upon gentle pressure with the vascular probe, the artery is not compressible and the pulsations become more evident. The arterial bifurcation is identified by scanning north to south, in order to prevent puncture at this level (Figure 6.2A). A needle guide can be used to fix the angle of the needle and to intersect the ultrasound plane at the desire depth and avoid the bifurcation. By keeping the needle and the artery in the center of the image, one can reduce the number of puncture attempts and the incidence of unwanted venipunctures, reducing time to access and the risk of potential vascular complications.7,8 Medial to the femoral artery (or occasionally underneath) lays the femoral vein, which can be identified because of its thinner and smother walls as well as the compressibility upon gentle pressure with the vascular probe. Once the vessels are identified, an anterior wall stick can be visualized with the ultrasound as the needle enters the vessel.
Most difficulties in entering the femoral artery and vein—and most vascular complications—arise as the result of inadequate identification of the landmarks prior to attempted vessel puncture. Once the catheter is introduced, a femoral angiogram (15 to 30 degrees ipsilateral) can be performed to confirm the position of the sheaths. A high puncture of the artery at or above the inferior epigastric artery or above the inguinal ligament makes catheter advancement difficult and predisposes to inadequate compression, hematoma formation, and/or retroperitoneal bleeding following catheter removal (see Chapter 4). A low puncture of the artery (at or below the femoral bifurcation or >3 cm below the inguinal ligament) increases the chance that the puncture will be at the bifurcation of the profunda and superficial femoral branches and will fail to enter the arterial lumen. Puncture of either one of the branches increases the risk of false aneurysm formation or thrombotic occlusion owing to smaller vessel caliber. Because the superficial femoral artery frequently overlies the femoral vein, low venous punctures may pass inadvertently through the superficial femoral artery, leading to excessive bleeding and possible arteriovenous (A-V) fistula formation.9 (see Chapter 4).
Local Anesthesia
Adequate subcutaneous local anesthesia is essential for a successful catheterization. Inadequate anesthesia leads to poor patient cooperation and makes the time in the catheterization laboratory unpleasant for both patient and operator. Once the inguinal ligament and femoral artery have been identified, the femoral artery is palpated along its course using the three middle fingers of the left hand, with the uppermost finger positioned just below the inguinal ligament. Without moving the left hand, a linear intradermal wheal of 1% or 2% lidocaine is raised slowly by tangential insertion of a 25- or 27-gauge needle along a course overlying both the femoral artery and vein at the desired level of entry. The smaller needle is then replaced by a 22-gauge 1.5-inch needle, which is used to infiltrate the deeper tissues along the intended trajectory for arterial and venous entry. As this needle is advanced, small additional volumes of lidocaine are infiltrated by slow injection. Each incremental infiltration should be preceded by aspiration so that intravascular boluses can be avoided. To avoid unnecessary injury to the femoral artery, we sometimes intentionally infiltrate medially and laterally to the pulse. If the anesthetic track passes through the artery, infiltration should be suspended until the tip of the needle has passed out of the back wall of the vessel and then continued to the full length of the needle or to the point where the needle tip contacts the periosteum. Approximately 10 to 20 mL 1% or 2% Xylocaine administered in this fashion usually provides adequate local anesthesia. The patient should be warned that he or she may experience some burning as the anesthetic is injected, but that the medication will prevent any subsequent sharp sensations.
Once local anesthesia has been achieved, with the left hand remaining in place, transverse skin punctures can be made over the femoral artery and vein, using the tip of a #11 scalpel blade. This procedure can decrease the resistance that is encountered during subsequent advancement of the vascular sheath and increases the likelihood that any vascular bleeding will manifest as oozing through the puncture rather than being hidden in the formation of a deep hematoma, although some operators prefer inserting small diameter vascular sheaths (5F or 6F) without a skin nick.
Femoral Vein Puncture
If right heart catheterization is to be performed, or secure venous access is desired (for administration of fluids and medications or rapid placement of a temporary pacing catheter), the femoral venous puncture is usually performed prior to arterial puncture. With the left hand palpating the femoral artery along its course below the inguinal ligament, the needle is introduced through the more medial aspect. If vascular ultrasound is available, this makes venous access easier by rapidly identifying the vein and visualizing the anterior wall stick.
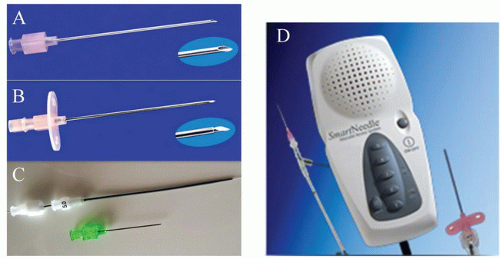
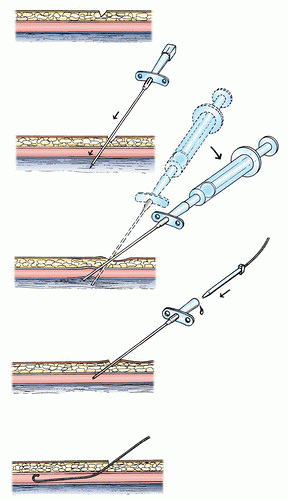
Classically, an 18-gauge thin-walled Seldinger needle was used in the past; this needle consists of a blunt, tapered external cannula through which a sharp solid obturator projects (Figure 6.3). The needle is to be grasped so that the index and middle fingers lie below the lateral flanges of the needle and the thumb rests on the top of the solid obturator as the needle is advanced along the sagittal plane angled approximately 45° cephalad (depending on the degree of obesity of the patient). Although this needle can occasionally be advanced up to its hub, the tip of the needle will usually stop more superficially as it encounters the periosteum of the underlying pelvic bones. The periosteum is well innervated and may be quite tender if the initial lidocaine infiltration fails to reach this level. Accordingly, forceful contact with the periosteum is neither necessary nor desirable. If the patient experiences significant discomfort, some operators would remove the obturator from the Seldinger needle and infiltrate additional lidocaine into the deep tissues through the outer cannula. At this point, the Seldinger needle should have transfixed the femoral vein. The obturator is removed, and a 10-mL syringe is attached to the hub of the cannula. The syringe and cannula are then depressed so that the syringe lies closer to the anterior surface of the thigh (Figure 6.4) and the needle is more parallel (rather than perpendicular) to the vein. Gentle suction is applied to the syringe, and the whole assembly is slowly withdrawn toward the skin surface. In doing so, it is helpful to control the needle with both the left hand (which also rests on the patient’s leg for support) and the right hand (which also controls the aspirating syringe). As the tip of the cannula is withdrawn into the lumen, venous blood will flow freely into the syringe.
However, nowadays the technique most widely used is a modified Seldinger, where instead an 18- to 21-gauge single-wall puncture needle with a sharpened tip and without the inner obturator is used (Figure 6.3). Placement of a fluid-filled syringe on the needle’s hub allows direct frontwall entry of the vein without the need to first exit the back wall and then pull back. Otherwise, the technique used is identical after entry of the venous lumen has been achieved.
With the left hand stabilizing the needle, the right hand is used to remove the syringe and to advance a 0.018 to 0.038-inch J guidewire into the hub of the needle under fluoroscopy or ultrasound guidance. The curved wire tip may be straightened by hyperextension of the wire shaft in the right hand or by leaving the tip of the wire within the plastic introducer supplied by the manufacturer; when using a 0.018 wire for the 21-gauge needle, the wire has a straight or slightly curved soft tip. The wire should slide through the needle and 30 cm into the vessel with no perceptible
resistance. Fluoroscopy should then show the tip of the guidewire just to the left (patient’s right) of the spine.
resistance. Fluoroscopy should then show the tip of the guidewire just to the left (patient’s right) of the spine.
If difficulty is encountered in advancing the guidewire, it should never be overcome by the application of force. Fluoroscopy may reveal that the tip of the wire has lodged in a small lumbar branch; it can be drawn back slightly and redirected or gently prolapsed up the iliac vein. When resistance to advancement is encountered at or just beyond the tip of the needle, however, even greater care is required. This resistance may be caused by apposition of the tip of the needle to the back wall of the vein, which can be corrected by further depression of the needle hub or slightly reorienting the hub to one side or the other, with or without slight withdrawal of the needle shaft. If this maneuver fails to allow free advancement of the wire, however, the wire should be removed, and the syringe should be reattached to the needle hub to ensure that free flow of venous blood is still present before additional wire manipulation is attempted—the wire should not be reintroduced unless free flow is obtained. If it is necessary to withdraw the wire, this should always be done gently, since it is possible for the wire to snag on the tip of the needle. To prevent this, the needle and wire should be removed as a unit. If the wire still cannot be advanced after these maneuvers, the needle should be withdrawn and the puncture site should be compressed for 1 to 3 minutes. The anatomic landmarks should be reconfirmed and puncture reattempted. In some cases, puncturing the vein during a Valsalva maneuver or after giving a bolus of intravenous fluids may help by distending the femoral vein and making clean puncture more likely.
After the wire has freely entered the vein, the needle is removed, leaving the wire well within the vein and secured at the skin entry site by the left hand. At this point, a small skin nick can be enlarged and deepened, using the tips of a curved mosquito forceps. The protruding wire is wiped with a moistened gauze pad, and its free end is threaded into the lumen of a sheath and dilator combination adequate to accept the intended right-sided heart catheter (Note: if a micropuncture kit is used, a 4F or 5F sheath and dilator are used over the 0.018-inch wire in order to exchange for an 0.035-0.038 inch wire, over which the intended sheath appropriate for the right heart catheter is then inserted). All current sheaths are equipped with a back-bleed valve and side-arm connector (Figure 6.5) to control bleeding around the catheter shaft and to provide a means of administering drugs or extra intravenous fluids during the right-sided heart catheterization. The operator must make sure that he or she has control of the proximal end of the guidewire and that it is held in a fixed position as the sheath and dilator are introduced through the skin. Insertion is eased if the sheath and dilator are rotated as a unit while they are advanced progressively through the soft tissues. If excessive resistance is encountered, it may be necessary to remove the dilator from the sheath and to
introduce the dilator alone before attempting to introduce the combination. If inspection shows that initial attempts have created significant burring at the end of the sheath, a new sheath should be obtained.
introduce the dilator alone before attempting to introduce the combination. If inspection shows that initial attempts have created significant burring at the end of the sheath, a new sheath should be obtained.
Once the sheath is in place, the wire and dilator are removed, and the sheath is flushed by withdrawal of blood and injection of heparinized saline solution. If needed, this sheath can be connected via a sterile length of intravenous extension tubing to provide a carrier for drug administration by the nurse. Although drug administration can also take place via a peripheral intravenous line, the side arm of the sheath avoids any concerns about how quickly volume can be administered or whether infiltration of the peripheral line might jeopardize drug delivery in an emergency. Even if right heart catheterization is not planned, the femoral sheath makes it easy to place a right heart catheter or a temporary transvenous pacemaker lead if hemodynamic instability or bradyarrhythmia ensue.
Catheterizing the Right Heart from the Femoral Vein
A right (as well as a left) heart catheterization is needed to obtain a full profile of the hemodynamic state. Right heart catheterization can provide data regarding mean left heart filling pressures (the pulmonary capillary wedge, rather than just the post-A wave left ventricular [LV] end diastolic pressure), detect pulmonary arterial hypertension, measure the cardiac output, and detect left-to-right intracardiac shunts. Leaving the right heart catheter in the pulmonary artery during a complex interventional procedure also gives an ongoing measure of changes in the hemodynamic state as fluid and contrast loading take place, various medications (nitrates, diuretics, and so on) are given, and as episodes of ischemia develop and are treated.
For these reasons, it was once common to perform a right heart catheterization in every patient who came to the cardiac catheterization laboratory. However, the 1990 SCA&I survey showed that the practice was to perform right heart catheterization in only 28% of procedures.10 The use of right heart catheterization has fallen further after several standard-setting and regulatory agencies ruled that a left heart catheterization alone is adequate for most patients undergoing evaluation for coronary artery disease. The time (<5 minutes), added expense (<$100), and added risk (<1/10,000) of right heart catheterization are small, but so is the added information. So right heart catheterization is no longer routinely done in patients with a primary diagnosis of coronary artery disease, unless they have symptoms of congestive heart failure, noninvasive evidence of depressed LV function, associated valvular disease, or history of pulmonary hypertension. In such patients, however, we still believe that the quantitation of overall hemodynamic function provided by right heart catheterization justifies performance of this low-risk adjunctive part of the overall catheterization evaluation.
If right heart catheterization is to be performed, the desired right heart catheter (Figure 6.6) is flushed, attached to the venous manifold, introduced into the sheath, and advanced up the inferior vena cava. Although conventional woven Dacron (Goodale-Lubin or Cournand) catheters provide excellent torque control, their inherent stiffness makes them poorly suited for routine use in a training laboratory. It is considerably safer to use 7F balloon flotation catheters to exploit their ease of passage, low risk of injury to the rightsided heart chambers, and (with a suitably equipped catheter) their ability to perform thermodilution measurements of cardiac output. Unfortunately, such soft catheters with smaller internal diameters tend to have poor frequency response (see Chapter 10), may not adequately transmit the torque required for easy catheterization of the right-sided heart from the femoral approach, and accept only <0.025-inch guidewires. To bridge this gap, stiffer, balloon-tipped catheters (PWP monitoring catheter, Medtronic) have been developed that combine the safety of the Swan-Ganz catheter with the catheter control and frequency response previously found only in the woven Dacron catheters. The larger lumen diameter also allows the passage of conventional 0.035- and 0.038-inch diameter guidewires when necessary.
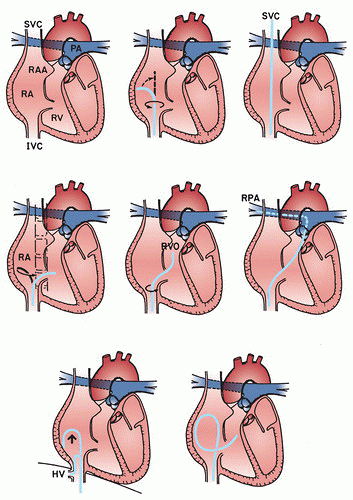
Deviation of the catheter tip from its paraspinous position during advancement from the leg suggests entry into a renal or hepatic vein, which can be corrected by slight withdrawal and rotation of the catheter. Once the catheter is above the diaphragm and within the right atrium, it is rotated counterclockwise to face the lateral wall of the right atrium (Figure 6.7). Additional counterclockwise rotation and gentle advancement allow passage of the catheter tip into the superior vena cava, which is contiguous with the posterolateral wall of the right atrium. In contrast, anterior orientation of the catheter tip at this point may result in its entrapment in the right atrial appendage and inability to
reach the superior vena cava. If passage to the superior vena cava is difficult, the tip of the catheter can be withdrawn to the inferior vena cava, where a 0.035-inch J guidewire can be introduced to traverse the straight-line path from the inferior to the superior vena cava along the back wall of the right atrium. Once in position, a baseline superior vena caval blood sample is obtained for measurement of oxygen saturation and comparison with the subsequently measured pulmonary arterial blood oxygen saturation to screen for unsuspected left-to-right shunts. The catheter is then flushed with heparinized saline solution and withdrawn to the right atrium for pressure measurement.
reach the superior vena cava. If passage to the superior vena cava is difficult, the tip of the catheter can be withdrawn to the inferior vena cava, where a 0.035-inch J guidewire can be introduced to traverse the straight-line path from the inferior to the superior vena cava along the back wall of the right atrium. Once in position, a baseline superior vena caval blood sample is obtained for measurement of oxygen saturation and comparison with the subsequently measured pulmonary arterial blood oxygen saturation to screen for unsuspected left-to-right shunts. The catheter is then flushed with heparinized saline solution and withdrawn to the right atrium for pressure measurement.
To advance a catheter from the femoral vein to the pulmonary artery, the tip of the catheter is positioned in the lower portion of the right atrium, directed toward its lateral border. If a balloon flotation catheter is being used, the balloon is inflated at this point. Clockwise rotation is applied, which causes the catheter tip to sweep the anterior and anteromedial wall of the right atrium, along the location of the tricuspid valve (Figure 6.7). As the catheter tip passes over the tricuspid orifice, a slight advancement causes it to enter the right ventricle, where pressure is again recorded. If the right atrium is enlarged, greater curvature of the catheter may be necessary, i.e., a large J loop. Such a loop may be formed by bending the tip of the catheter against the lateral right atrial wall or by engaging in the ostium of the hepatic vein (just below the diaphragm). This larger loop can then be rotated clockwise in the atrium as described above, causing the tip of the catheter to enter the right ventricle. Right ventricular pressure is then recorded.
Simple advancement of the catheter in the right ventricle causes the tip to move toward the apex of that chamber and usually does not result in catheterization of the pulmonary artery. To catheterize the pulmonary artery, the catheter must be withdrawn slightly so that its tip lies horizontally and just to the right (patient’s left) of the spine. In this position, clockwise rotation causes the tip of the catheter to point upward (and slightly posterior) in the direction of the right ventricular outflow tract (Figure 6.7). The catheter should be advanced only when it is in this orientation to minimize the risk of ventricular arrhythmias or injury to the right ventricle. Advancement may be facilitated if performed as the patient takes a deep breath.
If these maneuvers fail to achieve access to the pulmonary artery owing to enlargement of the right atrial and ventricular chambers, the catheter may be withdrawn to the right atrium and formed into a large reverse loop, which allows the tip of the catheter to cross the tricuspid valve in an upward orientation (similar to that when right heart catheterization is performed from above), which makes it more likely to enter the outflow tract (Figure 6.7, bottom right). When manipulated appropriately, the catheter tip should cross the pulmonic valve and advance to a wedge position without difficulty. Having the patient take a deep breath and cough during advancement often helps to achieve a wedge position. Alternatively, a small amount of air may be released from the balloon to decrease its size and facilitate wedging in a smaller, more distal branch of the pulmonary artery. Catheters advanced from the leg are more likely to seek the left pulmonary artery, whereas catheters advanced from above tend to seek the right pulmonary artery as they make a continuous counterclockwise curve through the right heart chambers. If needed, either pulmonary artery can be catheterized by appropriate manipulation or careful introduction of a curved J guidewire, but extending guidewires into the thin-walled pulmonary arteries should be avoided unless absolutely necessary.
Following measurement of the wedge pressure, the balloon (if a balloon-tip catheter is being used) is deflated and the catheter is withdrawn into the more proximal left or right pulmonary artery. There, pulmonary arterial pressure is measured and another blood sample for measurement of oxygen saturation is obtained. If a more simultaneous “snapshot” of the hemodynamic state is desired, these entry pressures can be rerecorded during a rightsided heart pullback. For practical reasons, we now tend to rerecord only the wedge pressure (simultaneous with the LV pressure) and pulmonary artery pressure, coincident with the measurement of the cardiac output. When baseline hemodynamics are abnormal, we commonly leave the right heart catheter in the proximal pulmonary artery for the duration of the case to allow continuous monitoring of the pulmonary artery diastolic pressure as an index of volume status and ischemic LV dysfunction.
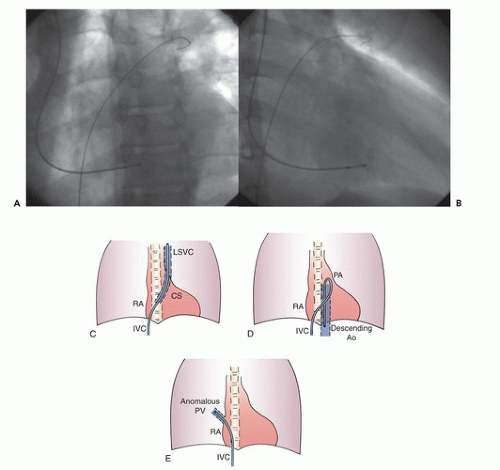
Attempts to perform right heart catheterization occasionally result in entry into other structures. If a woven Dacron catheter is advanced in the right atrium with a posteromedial orientation, it may cross a patent foramen ovale and enter the left atrium. This is sometimes hard to detect by catheter position alone because the catheter appearance in the left atrium or ventricle may be indistinguishable (in the anteroposterior view) from its course during usual right heart catheterization. It can, however, be recognized by a change to a left atrial pressure waveform, position of the catheter tip across the spine and frequently out into the left lung field (i.e., into a pulmonary vein, Figure 6.8A and B), and the ability to withdraw fully oxygenated blood from the catheter tip. Although more unusual, a woven Dacron catheter can also enter the ostium of the coronary sinus, located inferiorly and posteriorly to the tricuspid orifice. There will be continued presence of a right atrial waveform, but blood sampling will disclose far lower oxygen saturation (20% to 30%) than was present in the superior vena cava. In the right anterior oblique (RAO) projection, the catheter will be seen to remain in the atrioventricular groove rather than passing rightward into the ventricle. Anatomic abnormalities can also be suspected when the catheter takes an unusual position or course during attempted right heart catheterization. Figure 6.8C, D, and E depict the appearance of the right-sided heart catheter course in three such congenital abnormalities (persistent left superior vena cava, patent ductus arteriosus, and anomalous pulmonary venous return). The most important points about these side trips off the beaten path to the right ventricle are that the operator should recognize that the tip of the catheter is not in the right ventricle (i.e., one should not attempt to get to the pulmonary artery) and should decide where the catheter is (by pressure monitoring, saturation analysis, or hand
injection of a small amount of contrast agent) before withdrawing the catheter to the right atrium and proceeding with the right heart catheterization.
injection of a small amount of contrast agent) before withdrawing the catheter to the right atrium and proceeding with the right heart catheterization.
In patients with elevated right heart pressures or prior placement of an inferior vena caval filter or umbrella, those undergoing specialized procedures (endomyocardial biopsy, coronary sinus catheterization), or those in whom prolonged postprocedural monitoring with a balloon flotation catheter is desired, the right upper extremity (subclavian or median basilic vein) or the right internal jugular vein offers an excellent alternative to the femoral vein. The technique for jugular puncture is described in Chapter 26, and the method of advancing the
right-sided heart catheter to the pulmonary artery from the median basilic vein is described in Chapter 7.
right-sided heart catheter to the pulmonary artery from the median basilic vein is described in Chapter 7.
Femoral Artery Puncture
The original Seldinger technique for cannulation of the femoral artery consists of advancing the Seldinger needle completely through the artery until the periosteum is encountered. The obturator is then removed, and the hub of the needle is depressed slightly toward the anterior surface of the thigh. Arterial pressure makes it unnecessary to attach a syringe to the cannula, so that both hands can be used to stabilize the needle as it is slowly withdrawn. When the needle comes back into the lumen of the femoral artery, as evidenced by vigorous pulsatile flow of arterial blood, a 0.035- or 0.038-inch J guidewire is advanced carefully into the needle. This technique has largely been replaced by an anterior wall puncture or modified Seldinger technique described above. Once the fluoroscopic landmarks have been identified, vascular ultrasound can help visualize a single anterior wall stick above the bifurcation and in the common femoral artery as explained above. When the artery is accessed without ultrasound imaging guidance, the needle is inserted at approximately 45° along the axis of the femoral artery (depending on degree of obesity of patient) as palpated by the three middle fingers of the left hand. Occasionally, the “bounce” technique can be used. With this technique, the proximity to the artery can be confirmed based on the pulsation transmitted to the needle; if the needle is on top of the artery the movement is north to south, while when the needle is lateral to the artery the pulsation is transmitted laterally and the movement of the needle is from side to side. This technique is more obvious when using a 21-gauge needle.
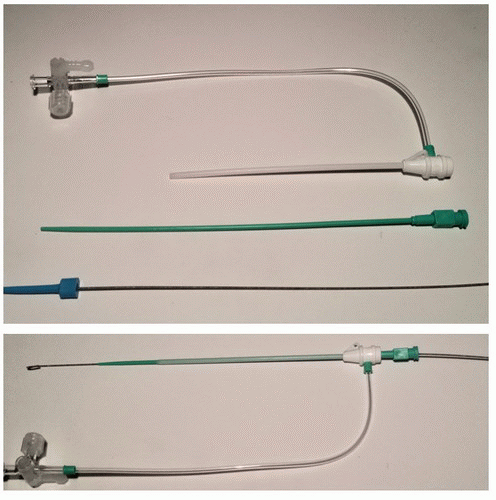
There are several needle options used for the modified Seldinger technique (Figure 6.3). The Potts-Cournand needle has an obturator with a small lumen that transmits a flashback of arterial blood as the vessel is entered, while the 18-gauge single-wall puncture needle described for venous entry does not include an obturator. When the femoral pulse is difficult to palpate or numerous needle insertions have been fruitless, it may be easiest to use the 18-gauge SmartNeedle (Vascular Solution, Maple Grove, MN; see Figure 6.3, bottom panel). The obturator of this device contains a Doppler crystal that picks up pulsatile arterial or more continuous venous flow, and thereby helps aim the needle tip toward the center of the desired vascular lumen. In addition, the use of ultrasound guidance in conjunction with a micropuncture kit can facilitate the identification of the optimal puncture site. It should be noted that when a 21-gauge needle is used, the amount of blood that exteriorizes has less pressure than when a larger bore needle is used. However, pulsatile bright blood will still be noted. The Femoral Micropuncture or Routine Introducer Study (FEMORIS), a randomized multicenter study, is assessing whether the use of the micropuncture technique provides a clinical benefit when compared with the standard 18-gauge needle front-wall technique.
Whichever needle is used to enter the arterial lumen, the guidewire introduced through the needle should move freely up the aorta (located to the right [patient’s left] side of the spine on fluoroscopy) up to the level of the diaphragm. When difficulty in advancing the guidewire is encountered at or just beyond the tip of the needle and is not corrected by slight depression or slight withdrawal of the needle, the guidewire should be withdrawn to ensure that brisk arterial flow is still present before any further wire manipulation is attempted. If flow is not brisk or if the wire still cannot be advanced, the needle should be removed and the groin should be compressed for 5 minutes. The operator should verify the correctness of the anatomic landmarks and attempt repuncture of the femoral artery. If the second attempt is also unsuccessful in allowing wire advancement, a third attempt on the same vessel is unwise, and an alternative access site should generally be selected.
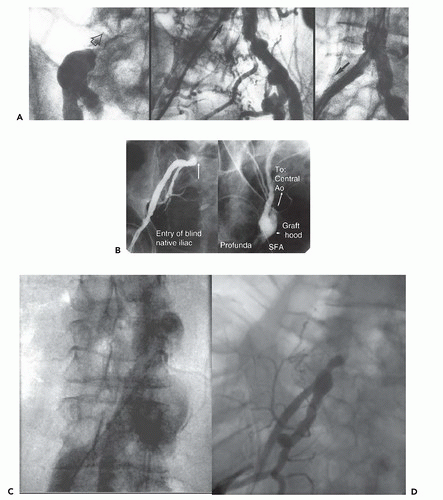
If wire motion is initially free, but resistance is encountered after several centimeters (particularly if the patient complains of any discomfort during wire advancement), extensive iliac disease or subintimal wire position are distinct possibilities. The wire should be pulled back slightly under fluoroscopic control, and the needle should be removed as the left hand is used to stabilize the wire and control arterial bleeding. After the wire is wiped with a moist gauze pad, a small (4F or 5F) dilator can be cautiously introduced to a point just below where wire movement became difficult. The wire is then withdrawn from the dilator, blood is aspirated to ensure free flow, and a small bolus of diluted low-osmolar contrast medium is then injected gently under fluoroscopic monitoring. This should disclose the anatomic reason for difficult wire advancement—generally iliac tortuosity, stenosis, or dissection. Problems advancing the wire above the aortic bifurcation may also suggest the presence of an abdominal aortic aneurysm.11 Either can usually be overcome by use of a floppy steerable (Wholey wire, Malinckrodt, Hazelwood, MO) or hydrophilic (Glidewire, Terumo) guidewire, carefully reintroduced through the dilator in an attempt to reach the descending aorta, using extreme care to avoid perforation, dissection, or dislodgment of atherothrombotic debris (Figure 6.9A). In an era when the obstructing lesion can be quickly and effectively treated by angioplasty or stent placement (see Chapter 34), iliac stenosis is no longer a firm indication to abandon transfemoral left heart catheterization.
If contrast injection through the small dilator reveals that subintimal wire passage has occurred or that the ipsilateral iliac artery is occluded, retrograde left heart catheterization should be relocated to the other femoral artery, the radial, or brachial artery. Patients with retrograde dissection should be observed for signs of progressive dissection or arterial compromise, both of which are fortunately rare with retrograde guidewire dissections.
In an aging population with diffuse atherosclerotic disease, the question of performing left heart catheterization via a prosthetic (e.g., aortobifemoral) graft arises frequently.12,13 This is not an ideal approach because the graft wall is tough (making sheath insertion difficult), such grafts may contain diffuse atherosclerotic or thrombotic debris, and graft closure or serious graft infection may occur. The graft should be identified as a separate structure from the adjacent native femoral artery and punctured using a front-wall approach, ideally under vascular ultrasound. Even if the graft hood is punctured correctly, the guidewire may pass through the anastomosis and into the native femoral artery rather than proximally up the graft.12 In that event, contrast injections through a small dilator in an RAO projection (ipsilateral) will disclose the problem. Partial withdrawal of the dilator and the use of special steerable guidewires may then allow the wire to be redirected into the graft lumen and thereby reach the descending aorta (Figure 6.9B). A vascular introducing sheath should always be used to avoid excessive friction during catheter movement or excessive traction on catheter tips during withdrawal, but serial dilators may be needed to facilitate sheath passage through the tough graft wall. Hydrophilic sheaths can also be used in order to facilitate sheath insertion. This approach via a vascular graft can thus be used with care, particularly when other alternatives (e.g., brachial, axillary, or radial artery) are less than desirable. Some operators choose to administer prophylactic antibiotics (Kefzol 1 g every 8 hours for 24 hours) when achieving vascular access via a prosthetic graft.
Catheterizing the Left Heart from the Femoral Artery
Once the guidewire has been advanced to the level of the diaphragm and the needle has been removed, the operator’s left hand is used to stabilize the wire and control arterial bleeding while the wire is wiped with a moistened gauze pad to remove any adherent blood. If the catheter is to be introduced directly into the artery, the soft tissues are predilated by brief introduction of a Teflon arterial dilator one F size smaller than the intended catheter before insertion into the left heart catheter itself. Essentially all left heart catheterizations from the femoral approach, however, are now performed using an appropriate-sized vascular sheath (e.g., a 6F sheath for a 6F catheter) that is equipped with a back-bleed valve and side-arm tubing as described above. The 15-cm-long sheath is commonly used for diagnostic catheterization, but can reach only the midiliac. In the presence of severe tortuosity, it may be preferable to use the 23-cm-long sheath designed for interventional procedures, which is sufficiently long to enter the distal aorta above the bifurcation. This helps improve the torque responsiveness of diagnostic catheters under those circumstances.
The chosen sheath is introduced over the guidewire (the proximal end of which is held in a straightened, fixed position) with a rotational motion, following which the guidewire and dilator are removed and the sheath is aspirated and flushed. The sheath can be connected to a pressurized flush system (Intraflo II [30 mL/h], Abbot Critical Care, North Chicago, IL) to avoid clot formation in the sheath. Alternatively, this side arm can be connected to a manifold for monitoring arterial pressure at a separate site (e.g., during passage of a pigtail catheter across a stenotic aortic valve). This sheath should be flushed before insertion and after removal of each catheter.
In the classic approach as described above, the guidewire was removed once the sheath had been inserted. This required that the desired left heart catheter be flushed and loaded with a 145-cm J guidewire before its nose was introduced into the back-bleed valve of the sheath. The soft end of the guidewire was then advanced carefully through the catheter, out the end of the sheath, and to the level of the diaphragm before the catheter itself was advanced. One concern, however, is that readvancement of the guidewire out the end of the sheath can cause vascular injury in the presence of severe iliac tortuosity or disease. We therefore adopted a modified technique in which a short exchange length (175 cm) Newton J (Cook, Inc.) is placed through the access needle, and its tip is left at the level of the diaphragm as the dilator is removed from the sheath, the sheath is flushed, and the left heart catheter is inserted over the wire and through the sheath lumen. This obviates the need to renegotiate complex iliofemoral anatomy with the guidewire.
Once the catheter has been advanced to the desired level (either above the diaphragm or into the ascending aorta), the guidewire is removed so that the catheter can be connected to the arterial manifold and double-flushed (withdrawal and discarding of 10 mL of blood, followed by injection of heparinized saline solution). All subsequent left heart catheters are then introduced by reinserting this wire to the level of the diaphragm (allowing one catheter to be removed and the second to be reintroduced safely), rather than withdrawing the first catheter completely and then inserting the second catheter and wire through the sheath de novo. Of course, if the left heart catheterization is being performed without the aid of a sheath, the operator must leave the tip of the wire in the abdominal aorta during the removal of the first catheter and the introduction of a second catheter to retain access to the vessel. These over-the-wire catheter exchanges are facilitated by extending the back end of the wire straight down the patient’s leg and holding it fixed there to ensure that the wire remains in constant position within the aorta as the newer catheter is advanced.
A Word About Heparin
As described in Chapters 4 and 8, early catheterizations from the femoral artery had a higher incidence of major complications than catheterization from the brachial artery. One difference was that brachial catheterization used systemic heparinization to avoid thrombosis in the smaller diameter brachial artery. When systemic heparinization was adopted in femoral procedures, the rates of complications became equivalent, and it became standard practice to achieve full intravenous heparinization (5,000 U) immediately after the left-sided sheath was inserted. Lesser amounts of heparin (2,500 to 3,000 U) were used,
particularly in smaller patients, and additional heparin (up to a total of 50 to 70 U/kg) was given if the procedure went on to a coronary intervention. This type of higher heparin dosing is routinely monitored by an activated clotting time (ACT) machine in the cardiac catheterization laboratory, and titrated to an ACT of roughly 300 seconds.14 If it is planned to use an intravenous IIb/IIIa receptor blocker, lower levels of heparin anticoagulation (target ACT >200 seconds) may be desirable to prevent excessive bleeding risk; on the other hand, if direct thrombin inhibitors are used (i.e., bivalirudin) the goal is to confirm that the ACT is above 200 seconds before introducing coronary wires to confirm that the drug has been administered, and from then the ACT is monitored to confirm that the patient is receiving the drug through the continuous intravenous infusion. Most of the times the ACT is >300 seconds while the infusion is maintained.
particularly in smaller patients, and additional heparin (up to a total of 50 to 70 U/kg) was given if the procedure went on to a coronary intervention. This type of higher heparin dosing is routinely monitored by an activated clotting time (ACT) machine in the cardiac catheterization laboratory, and titrated to an ACT of roughly 300 seconds.14 If it is planned to use an intravenous IIb/IIIa receptor blocker, lower levels of heparin anticoagulation (target ACT >200 seconds) may be desirable to prevent excessive bleeding risk; on the other hand, if direct thrombin inhibitors are used (i.e., bivalirudin) the goal is to confirm that the ACT is above 200 seconds before introducing coronary wires to confirm that the drug has been administered, and from then the ACT is monitored to confirm that the patient is receiving the drug through the continuous intravenous infusion. Most of the times the ACT is >300 seconds while the infusion is maintained.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree