Chapter 30 Noninfectious Conditions in Patients with Human Immunodeficiency Virus Infection
Although most complications affecting the lungs during the course of HIV infection arise from the viral infection itself, especially as reported early in the epidemic, HIV-infected patients may present with or subsequently develop complications related to alterations in immune regulation. A marked CD8+ T cell infiltrate in the lung may be caused by HIV in both symptomatic and nonsymptomatic patients. The effects of the virus on the pulmonary microenvironment include a progressive decline in local immunocompetence that results in failure to mount a protective immune response against opportunistic infections. The spectrum of noninfectious complications associated with HIV infection encompasses other idiopathic conditions and pulmonary malignancies, which include Kaposi sarcoma, Hodgkin and non-Hodgkin lymphomas, and solid tumors (Table 30-1 and 30-2).
Table 30-1 Noninfectious Pulmonary Conditions Associated With Human Immunodeficiency Virus Type 1 Infection
| Disease Category | Specific Disease(s) |
|---|---|
| Neoplastic disease | |
| Inflammatory disease | |
| Airway disease | |
| Pulmonary vascular disease | Pulmonary hypertension |
| Miscellaneous | Antiretroviral treatment–induced respiratory disease |
Table 30-2 Association of Noninfectious Pulmonary Conditions with Degree of Immunodeficiency
| Degree of Immunodeficiency | Noninfectious Pulmonary Condition |
|---|---|
| Severe (CD4+ cell count less than 50 cells/µL) | |
| Moderate to severe (CD4+ cell count 50-200 cells/µL) | |
| Moderate (CD4+ cell count 200-350 cells/µL) | |
| Normal (CD4+ cell count greater than 350 cells/µL) |
Neoplasia
Kaposi Sarcoma
Clinical Features
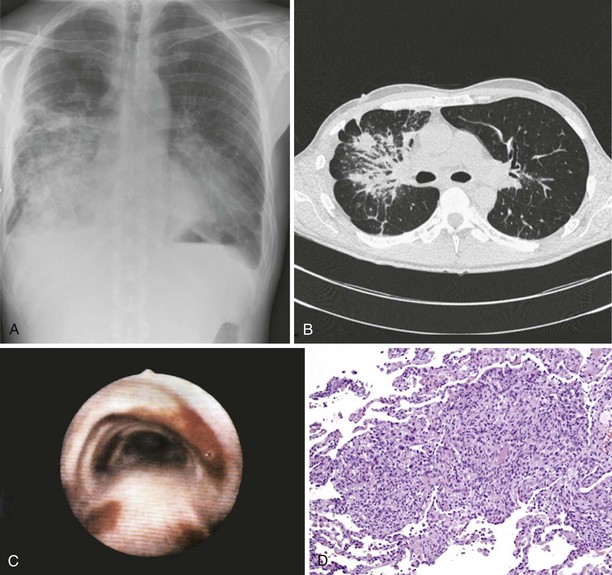
Pulmonary Kaposi sarcoma may cause nonproductive cough, hemoptysis, shortness of breath, chest pain, and fever. In rare instances, involvement of the larynx or trachea may cause airway obstruction. Extrapulmonary involvement is frequent, but 15% of cases of Kaposi sarcoma occur without skin lesions. CD4+ cell counts are low (0 to 100/µL) at the time of diagnosis. Examination of the lungs usually shows no abnormalities. Chest radiograph may be normal in appearance but commonly shows singular or multiple peribronchovascular nodules (Figure 30-1, A and B). Diffuse infiltration and air space consolidation also may be present. Pleural effusion is common. Hilar and mediastinal lymphadenopathy may be visible on chest films but are better visualized with computed tomography (CT) scanning. With pulmonary disease, CT scans typically reveal peribronchovascular nodules that are larger than 1 cm in diameter. The role of magnetic resonance imaging (MRI) or positron emission tomography (PET) in the diagnosis and management of pulmonary Kaposi sarcoma is not clear.
Diagnosis
Pulmonary Kaposi sarcoma is highly likely in a HIV-1–infected person with a CD4+ cell count of less than 100/µL, the characteristic cutaneous or mucocutaneous lesions, and pulmonary symptoms. Bronchoscopy is useful to visualize typical endobronchial lesions (see Figure 30-1, C). These are flat or slightly raised and occur throughout the tracheobronchial tree but are seen most frequently at airway bifurcations. Pleural effusions usually are exudative and may be serous or serosanguineous. Serum lactate dehydrogenase may be elevated. Histologic verification is usually not required for a diagnosis of Kaposi sarcoma. Endobronchial biopsy may confer a substantial risk of severe bleeding, but biopsy may be necessary in cases in which bronchial involvement is lacking. In such cases, transbronchial or percutaneous needle biopsy is useful. In cases of pleural Kaposi sarcoma, video-assisted thoracoscopy can be used to visualize pleural lesions and to perform biopsy. Open lung biopsy in this setting is obsolete because of the associated complications. Lung biopsy specimens show typical features of Kaposi sarcoma, which include a tumor-like infiltrate with a peribronchovascular distribution of spindle cells. Slitlike spaces without endothelium contain extravasated erythrocytes (see Figure 30-1, D). In situ hybridization and immunostaining usually are positive for HHV-8. HHV-8 DNA is detectable in bronchoalveolar lavage fluid (BAL) cells.
Treatment
Investigational agents include thalidomide, imatinib, mTOR inhibitors, IL-12, and fumagilin.