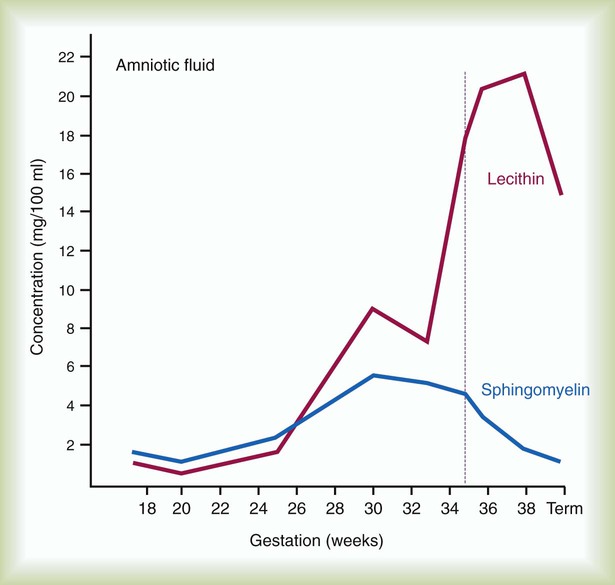
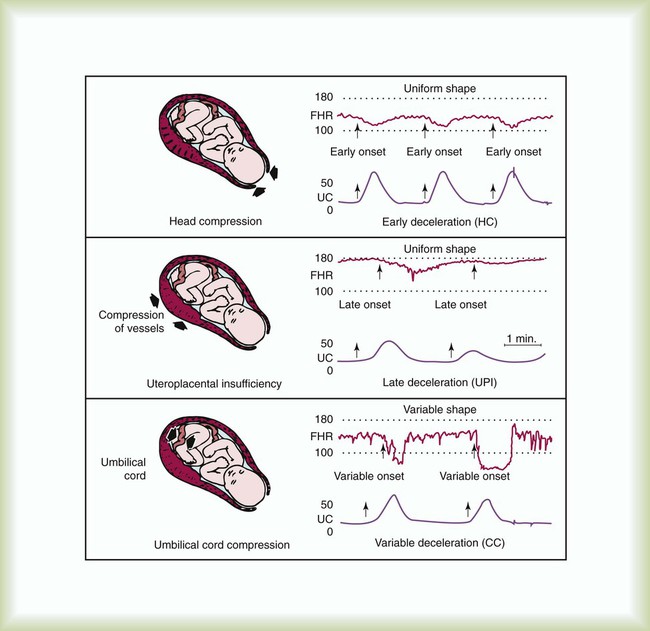
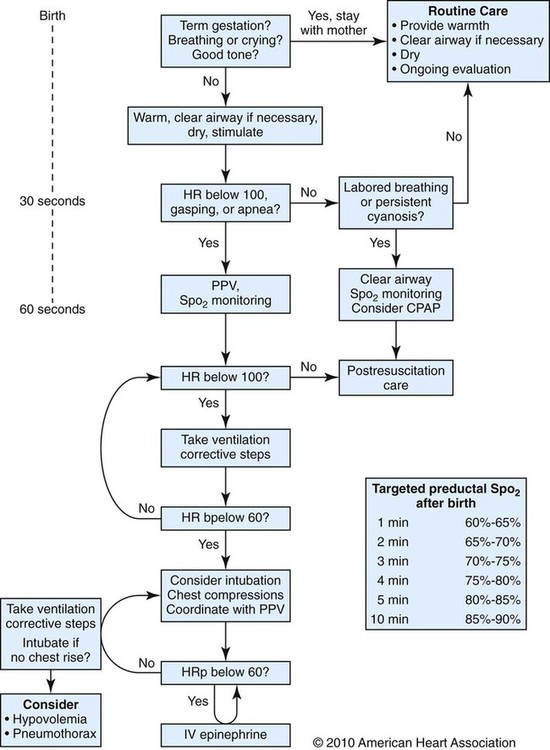
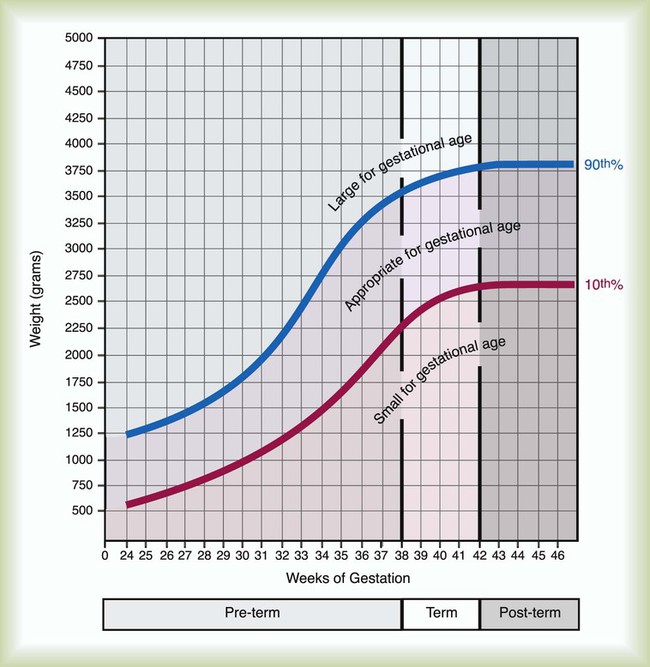
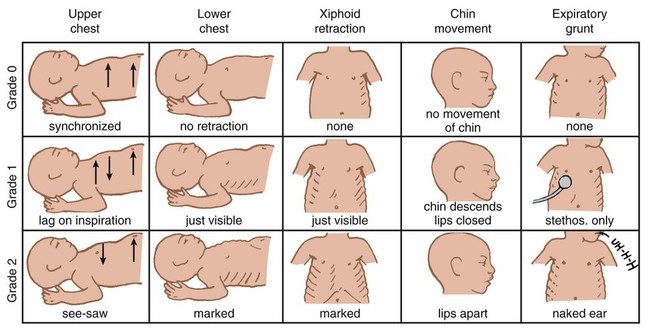
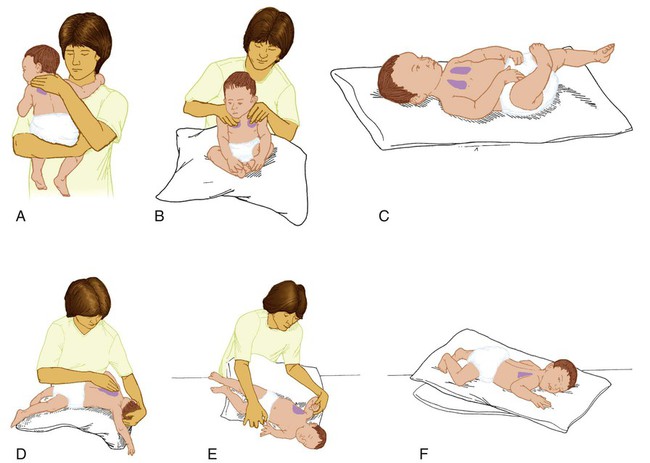
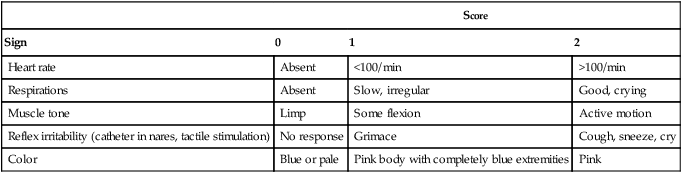
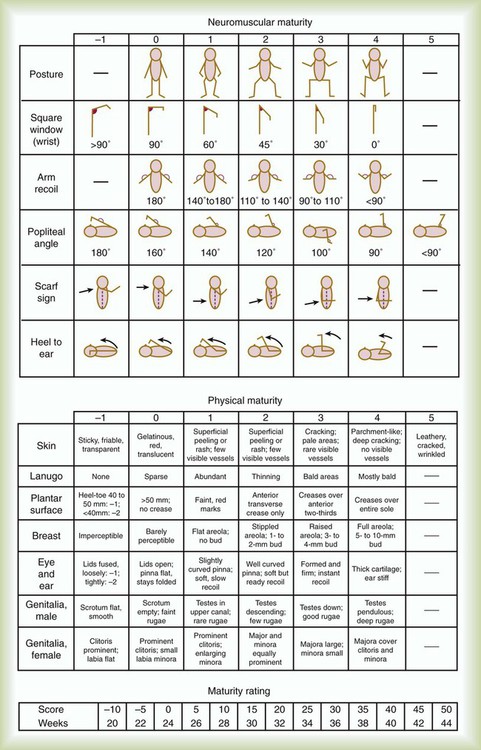
After reading this chapter you will be able to: Caring for infants and children is one of the most challenging and rewarding aspects of respiratory care. Competent clinical practice in this area requires knowledge of the many pathophysiologic differences among infants, children, and adults. Understanding the unique pathophysiology involved in neonatal and pediatric respiratory disorders (see Chapter 31) can assist the respiratory therapist (RT) in providing quality care to infants and children. A thorough understanding of how the respiratory system develops in the fetus is the first step toward acquiring the specialized knowledge needed to practice neonatal respiratory care (see Chapter 8). This chapter begins with an overview of neonatal and pediatric patient assessment and then describes respiratory care modalities used to treat these patients. Maternal risk factors include many medical, physical, and social conditions. Maternal health and individual physiology, pregnancy complications, and maternal behaviors affect the health of the fetus. Any condition that causes an interference with placental blood flow or the transfer of oxygen (O2) to the fetus can result in an adverse outcome. The clinician must be prepared for the possibility of resuscitation at delivery. This possibility is best anticipated by identifying risk factors that relate to neonatal compromise. Table 48-1 lists maternal risks and related outcomes of which the team preparing to receive the infant should be aware when the infant is delivered. TABLE 48-1 Maternal Condition and Neonatal Outcomes Amniocentesis involves direct sampling and quantitative assessment of amniotic fluid. Amniotic fluid may be inspected for meconium (fetal bowel contents) or blood. In addition, sloughed fetal cells can be analyzed for genetic normality. Lung maturation can be assessed with amniocentesis. The lecithin-to-sphingomyelin ratio (L : S ratio) involves measurement of two phospholipids, lethicin and sphingomyelin, synthesized by the fetus in utero. As shown in Figure 48-1, the L : S ratio increases with increasing gestational age. At approximately 34 to 35 weeks’ gestation, this ratio abruptly increases to greater than 2 : 1. An L : S ratio greater than 2 : 1 indicates stable surfactant production and mature lungs. Phosphatidylglycerol is another lipid found in the amniotic fluid that is used to assess fetal lung maturity. Phosphatidylglycerol first appears at approximately 35 to 36 weeks’ gestation. If phosphatidylglycerol is more than 1% of the total phospholipids, the risk of respiratory distress syndrome is less than 1%. Figure 48-2 illustrates the three common patterns of early decelerations, late decelerations, and variable decelerations. Early decelerations occur when the fetal heart rate decreases in the beginning of a contraction. This type of deceleration is benign and in most cases is caused by a vagal response related to compression of the fetal head in the birth canal. A late deceleration occurs when the heart rate decreases 10 to 30 seconds after the onset of contractions. A late deceleration pattern indicates impaired maternal-placental blood flow, or uteroplacental insufficiency. With variable decelerations, there is no clear relationship between contractions and heart rate. This pattern is the most common of the three and probably related to umbilical cord compression. Short periods of cord compression are generally benign, but prolonged periods of compression result in impaired umbilical blood flow and can lead to fetal distress. Fetal heart rate variability is the beat-to-beat variation in rate that occurs because of normal sympathetic or parasympathetic influences. A completely monotonous heart rate tracing may be indicative of fetal asphyxia. Fetal heart rate reactivity is the ability of the fetal heart rate to increase in response to movement or external stimuli. A healthy fetus has two accelerations within a 20-minute period. Meconium is the medical term for the infant’s first stools. It is a sticky green-black substance that if inhaled by the infant can cause significant respiratory problems. It is most likely present in a term or postterm newborn. Term infants delivered without evidence of meconium who are crying or breathing and have good tone should not routinely be separated from the mother. They should be dried, covered, and given to the mother and observed for breathing, activity, and color. If meconium is present and the infant is vigorous, pharyngeal suctioning with a bulb suction is appropriate. Simultaneously the infant should be dried and placed under a warmer and assessed for signs of respiratory distress. See later section on Respiratory Assessment of the Infant. If meconium is present in a nonvigorous infant, stimulation should be avoided. The most important and effective action in neonatal resuscitation is effective ventilation. Recommendations from the American Academy of Pediatrics are to attach a pulse oximeter to the infant, begin resuscitation efforts using room air, and assess carefully the amount of O2 needed.1 Effective PPV usually results in rapid improvement of heart rate. Initial ventilating pressures of 30 to 40 cm H2O may be necessary to achieve noticeable chest movement, particularly in a preterm newborn with surfactant deficiency. Continuous assessment of the lowest pressure needed to observe the chest rise is essential throughout the resuscitation. After application of PPV for 30 seconds, the heart rate is reassessed. If the heart rate is less than 60 beats/min, chest compressions are begun, and PPV is maintained. If the heart rate remains less than 60 beats/min after adequate ventilation with 100% O2 and chest compressions for 30 seconds, appropriate medications are given. As soon as the heart rate is noted to be greater than 100 beats/min, compressions are discontinued. If spontaneous breathing is present, PPV may be gradually reduced and then discontinued. If spontaneous breathing remains inadequate or if heart rate remains less than 100 beats/min, assisted ventilation is continued via bag-mask or endotracheal tube. Figure 48-3 outlines a newborn resuscitation algorithm and includes the targeted saturation levels for the first 10 minutes of life. An Apgar score is assigned at 1 minute and 5 minutes of life. The Apgar score is an objective scoring system used to evaluate a newborn rapidly. As shown in Table 48-2, the score has five components: heart rate, respiratory effort, muscle tone, reflex irritability, and skin color. Each component is rated according to standard definitions, resulting in a composite assessment score. Generally, infants scoring 7 or higher at 1 minute are responding normally. An infant with a score of 7 may require supportive care, such as O2 or stimulation to breathe. Infants with a 1-minute Apgar score of 6 or lower may require more aggressive support. TABLE 48-2 Apgar Scoring System for Newborn Assessment From Koff PB, Eitzman DV, Neu J: Neonatal and pediatric respiratory care, ed 2, St Louis, 1993, Mosby. Gestational age assessment and assessment of relationship of weight to gestational age are performed shortly after delivery. Determination of gestational age involves assessment of multiple physical characteristics and neurologic signs. Two common systems are used to determine gestational age: the Dubowitz scales and the Ballard scales. The Dubowitz scales involve assessment of 11 physical and 10 neurologic signs.2 Physical criteria include assessment of skin texture, skin color, and genitalia. Neurologic criteria include posture and arm and leg recoil. The Ballard scales are a simplified version of the Dubowitz scales and include six physical and six neurologic signs as illustrated in Figure 48-4. Soon after delivery, the newborn is stabilized and weighed, followed by determination of gestational age. Infants born between 38 weeks and 42 weeks are considered term gestation. Infants born before 38 weeks are preterm. Infants born after 42 weeks are postterm. All newborns weighing less than 2500 g are considered low birth weight. Newborns weighing less than 1500 g are considered very low birth weight (VLBW). Newborns weighing less than 1000 g are considered extremely low birth weight (ELBW). A newborn with a weight that is either too large or too small or who has been born preterm or postterm has a higher risk of morbidity and mortality. As shown in Figure 48-5, by plotting the infant’s gestational age against weight, the newborn’s relative developmental status can be classified. Infants whose weight falls between the 10th and 90th percentiles are appropriate for gestational age (AGA). Infants whose weight is above the 90th percentile are large for gestational age (LGA). Infants whose weight is below the 10th percentile are small for gestational age (SGA). Normal infant heart rates range from 100 to 160 beats/min. Heart rate can be assessed by auscultation of the apical pulse, normally located at the fifth intercostal space, midclavicular line. Alternatively, the brachial and femoral pulses may be used. Weak pulses indicate hypotension, shock, or vasoconstriction. Bounding peripheral pulses occur with major left-to-right shunting through a patent ductus arteriosus (PDA).3A strong brachial pulse in the presence of a weak femoral pulse suggests either PDA or coarctation of the aorta. Table 48-3 lists normal ranges of blood pressure for neonates of different sizes. TABLE 48-3 Normal Neonatal Blood Pressures From Whitaker K: Comprehensive perinatal and pediatric respiratory care, ed 3, Albany, NY, 2001, Delmar. Chest examination in an infant is more difficult to perform and interpret than in an adult because of the small chest size and the ease of sound transmission through the infant chest. Thorough observation of the infant greatly enhances the assessment data obtained. Infants in respiratory distress typically exhibit one or more key physical signs: nasal flaring, cyanosis, expiratory grunting, tachypnea, retractions, and paradoxical breathing. Nasal flaring is seen as dilation of the ala nasi on inspiration. The extent of flaring varies according to facial structure of the infant. Nasal flaring coincides with an increase in work of breathing. In concept, nasal flaring decreases the resistance to airflow. It also may help stabilize the upper airway by minimizing negative pharyngeal pressure during inspiration.4 Cyanosis may be absent in infants with anemia, even when arterial partial pressure of oxygen (PaO2) levels are decreased. In addition, infants with elevated fetal hemoglobin levels may not become cyanotic until PaO2 decreases to less than 30 mm Hg. Hyperbilirubinemia, common among newborns, may mask cyanosis. Grunting occurs when infants exhale against a partially closed glottis. By increasing airway pressure during expiration, grunting helps prevent airway closure and alveolar collapse. Grunting is most common in infants with respiratory distress syndrome, but it is also seen in other respiratory disorders associated with alveolar collapse. Figure 48-6 illustrates the Silverman score, which is a system of grading severity of lung disease. Surfactant production begins around the 24th week of gestation and continues through gestation. Surfactant contributes to the stability of the alveolar sacs by reducing the surface tension of the fluids that coat the alveoli. Surfactant deficiency places an infant at increased risk for respiratory distress. By about 34 weeks’ gestation, most infants have produced enough surfactant to keep the alveoli from collapsing. There are two specific approaches to preventing and treating surfactant deficiency. Surfactant deficiency is due to lung immaturity. When a premature delivery is anticipated, steroids are given to the mother to help promote lung maturation. In addition, infants born before 35 weeks’ gestation, especially infants born very prematurely (<30 weeks), should be assessed for the need to receive exogenous surfactant. The need for surfactant is determined by assessing the infant’s lung volume on chest x-ray, evaluating the inspired O2 concentration to maintain O2 saturations greater than approximately 88%, and clinically assessing the infant’s work of breathing. Once surfactant deficiency is determined, administering exogenous surfactant as soon as possible has been found to be most beneficial.5 Blood gas analysis is helpful in assessing respiratory distress in an infant. Many noninvasive techniques, such as transcutaneous partial pressure of oxygen (PtcO2), transcutaneous partial pressure of carbon dioxide (PtcCO2), end tidal carbon dioxide (CO2), and pulse oximetry (SpO2), are used to obtain comparable data, although blood gas analysis is more precise when results are critical. An infant blood gas sample can be obtained from an artery or capillary. Chapter 18 summarizes the advantages, disadvantages, and complications of these sampling methods. Care must be taken in assessing the results of capillary sampling. Capillary blood gases provide only information regarding ventilation and acid-base status, and accuracy is highly dependent on technique.6 Normal values for infant blood gases are listed in Table 48-4. TABLE 48-4 Age-Related Values Commonly Reported for Normal Blood Gases BE, Base excess; HCO3−, bicarbonate. Modified from Orzalesi MM, Mendicini M, Bucci G, et al: Arterial oxygen studies in premature newborns with and without mild respiratory disorders. Arch Dis Child 42:174, 1967. From Koff PB, Eitzman DV, Neu J: Neonatal and pediatric respiratory care, ed 2, St Louis, 1993, Mosby. Normal breathing in children is evidenced by quiet inspiration and passive expiration at an age-appropriate rate. Respiratory rates are rapid in neonates and decrease in toddlers and older children. Table 48-5 lists normal respiratory rates. The initial assessment of a pediatric patient starts with evaluating airway patency. Normal heart rates are higher in younger children and decrease with age. In assessing a pediatric patient, establishing if the airway is patent or has any obstructive component is essential. Signs that suggest upper airway obstruction include increased inspiratory effort with retractions or inspiratory efforts with no airway or breath sounds. TABLE 48-5 Normal Respiratory and Heart Rates by Age Lower saturation levels are targeted in infants less than 32 weeks’ gestation. There is evidence that exposure to supplemental O2 in a premature infant is a risk factor for the development of retinopathy of prematurity (ROP). ROP is caused by an abnormal vascularization of the retina, which in the most severe cases leads to retinal detachment. Preterm neonates weighing less than 1500 g are most susceptible. Hyperoxia is not the only factor associated with ROP, but close monitoring and adjusting of O2 therapy to avoid hyperoxia is crucial to decrease the risk of ROP. Specific saturation goals for this age group should be established, and O2 should be adjusted to maintain the intended target. Avoiding very high or very low saturation levels is critical. Adjusting the delivered O2 concentration by small increments avoids large swings in saturation levels.7–11 The effectiveness of O2 devices depends on the performance characteristics of the device (delivered FiO2, flow rate, relative humidity), the interface of the device, and the tolerance of the patient for using the device. Children are often frightened and combative, making it impractical to use some O2 administration devices. Selection of an O2 device must be based on the degree of hypoxemia and the emotional and physical needs of the child and family. O2 can be administered to infants and children by mask, cannula, high-flow nasal cannulas, or oxyhood. Table 48-6 compares the advantages and disadvantages of standard O2 delivery methods. TABLE 48-6 Secretion clearance techniques that can be applied to infants and children include chest physiotherapy, positive expiratory pressure therapy, autogenic drainage, flutter therapy, and mechanical insufflation-exsufflation.12,13 Secretion clearance techniques are considered when accumulated secretions impair pulmonary function and an infiltrate is visible on a chest radiograph. Secretion retention is common in children who have pneumonia, bronchopulmonary dysplasia, cystic fibrosis, bronchiectasis, and some neuromuscular diseases. Figure 48-7 shows postural drainage and percussion positions for infants and children. Infants and young children cannot cough on command. For this reason, secretions often must be removed by suctioning. For older children with excessive secretions, directed deep breathing and coughing may help improve pulmonary clearance. The use of mechanical insufflation-exsufflation in children with neuromuscular disease can be helpful in clearing secretions. Adjunctive therapy devices such as positive expiratory pressure, flutter, or intermittent percussive ventilation therapy have been effective in secretion clearance in patients with cystic fibrosis.14 In children with an intact upper airway, O2 therapy devices, such as low-flow nasal cannulas, do not routinely need to be humidified When the upper airway is bypassed by intubation, supplemental humidification must be provided using a heated humidifier. Humidification of inspired gases for infants and children receiving mechanical ventilation is commonly provided by a servo-controlled humidifier. Ideal features for these systems include the following: (1) low internal volume and constant water level to minimize compressed volume loss; (2) closed, continuous feed water supply to avoid contamination; (3) distal airway temperature sensor and high/low alarms. Common problems with humidifier systems include condensation in the tubing, inadequate humidification, and hazards associated with the heating coil.15,16 Using heated wire circuits can also reduce condensation in the circuit. Frequent evaluation of the humidification system is necessary to increase the potential of adequate humidity delivered to the airway. Inadequate humidification occurs in nonheated circuits when the humidifier temperature probe is placed too far upstream from the airway connector. Variable humidification problems occur when ventilator circuits pass through an environment and then into a warmed enclosure, such as an incubator or radiant warmer. Drug action in infants and children differs significantly from drug action in adults because of differences in physiology, which may include immature enzyme systems, immature receptors, and variable gastrointestinal absorption. Dosing may be imprecise, and systemic effects may be hard to predict. Table 48-7 lists aerosolized medications commonly used in children. TABLE 48-7 Commonly Used Aerosolized Medications PCP, Pneumocystis jiroveci pneumonia; RSV, respiratory syncytial virus. Small volume nebulizers (SVNs), MDIs, and dry powder inhalers (DPIs) can be used to deliver aerosolized drugs via mouthpiece or face mask to infants and children.17 Continuous aerosol drug therapy is also used for patients unresponsive to intermittent SVN treatments. Aerosol drug administration to intubated infants and children is challenging because of the decreased deposition from baffling of small endotracheal tubes in these patients, which prevents approximately 90% of the drug from entering the lungs, regardless of delivery system. In addition, careful adjustments must be made to the ventilator so that nebulizer flows do not alter delivered VT and inspiratory pressure and interfere with triggering efforts.18
Neonatal and Pediatric Respiratory Care
 Describe the correct approach to assessment of the fetus and newborn infant.
Describe the correct approach to assessment of the fetus and newborn infant.
 Discuss the use of oxygen therapy, bronchial hygiene therapy, aerosol drug therapy, airway management, and resuscitation approaches in the care of infants and children.
Discuss the use of oxygen therapy, bronchial hygiene therapy, aerosol drug therapy, airway management, and resuscitation approaches in the care of infants and children.
 Describe the correct approach to assessment of the pediatric patient.
Describe the correct approach to assessment of the pediatric patient.
 Discuss the use of continuous positive airway pressure and the basics of mechanical ventilation including high-frequency ventilation in the care of infants and children.
Discuss the use of continuous positive airway pressure and the basics of mechanical ventilation including high-frequency ventilation in the care of infants and children.
 List clinical situations where nitric oxide and extracorporeal life support are used, and discuss the basic application of each.
List clinical situations where nitric oxide and extracorporeal life support are used, and discuss the basic application of each.
Assessment of the Newborn
Maternal Factors
Maternal Condition
Fetal or Neonatal Outcome
Previous pregnancy complication
Same outcome as previous fetus
Diabetes mellitus
LGA, congenital malformations, RDS, hypoglycemia
Pregnancy-induced hypertension
Prematurity, SGA (preeclampsia)
Maternal age <17 years
Low birth weight, prematurity
Maternal age >35 years
Prematurity, chromosomal defects
Placenta previa
Prematurity, bleeding, SGA
Abruptio placentae
Fetal asphyxia, bleeding
Alcohol consumption
SGA, CNS dysfunction, mental retardation, facial dysmorphology
Smoking
SGA, prematurity, mental retardation, SIDS
Drug use
Placental abruption, IUGR, prematurity, CNS abnormalities, withdrawal disorders
Fetal Assessment
Evaluation of the Newborn
Apgar Score
Score
Sign
0
1
2
Heart rate
Absent
<100/min
>100/min
Respirations
Absent
Slow, irregular
Good, crying
Muscle tone
Limp
Some flexion
Active motion
Reflex irritability (catheter in nares, tactile stimulation)
No response
Grimace
Cough, sneeze, cry
Color
Blue or pale
Pink body with completely blue extremities
Pink
Assessment of Gestational Age
Respiratory Assessment of the Infant
Physical Assessment
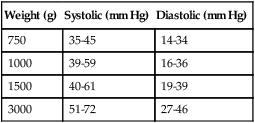
Weight (g)
Systolic (mm Hg)
Diastolic (mm Hg)
750
35-45
14-34
1000
39-59
16-36
1500
40-61
19-39
3000
51-72
27-46
Surfactant
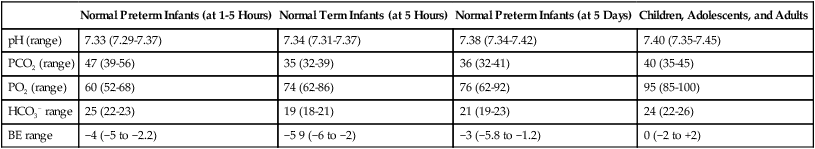
Blood Gas and Pulse Oximetry Analysis
Normal Preterm Infants (at 1-5 Hours)
Normal Term Infants (at 5 Hours)
Normal Preterm Infants (at 5 Days)
Children, Adolescents, and Adults
pH (range)
7.33 (7.29-7.37)
7.34 (7.31-7.37)
7.38 (7.34-7.42)
7.40 (7.35-7.45)
PCO2 (range)
47 (39-56)
35 (32-39)
36 (32-41)
40 (35-45)
PO2 (range)
60 (52-68)
74 (62-86)
76 (62-92)
95 (85-100)
HCO3− range
25 (22-23)
19 (18-21)
21 (19-23)
24 (22-26)
BE range
−4 (−5 to −2.2)
−5 9 (−6 to −2)
−3 (−5.8 to −1.2)
0 (−2 to +2)
Respiratory Assessment of the Pediatric Patient
Age
Breaths/Minute
Heart Rates
Infants (<1 yr)
30-60
90-120
Toddler (1-3 yr)
24-40
80-100
Preschooler (4-5 yr)
22-34
70-90
School age (6-12 yr)
18-30
70-90
Adolescent (13-18 yr)
16-22
60-80
Respiratory Care
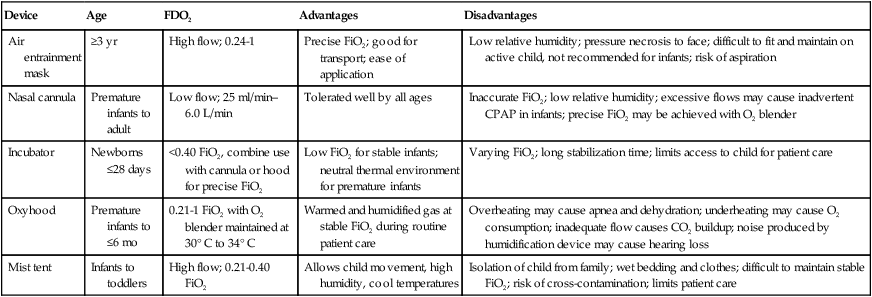
Oxygen Therapy
Goals and Indications
Methods of Administration
Device
Age
FDO2
Advantages
Disadvantages
Air entrainment mask
≥3 yr
High flow; 0.24-1
Precise FiO2; good for transport; ease of application
Low relative humidity; pressure necrosis to face; difficult to fit and maintain on active child, not recommended for infants; risk of aspiration
Nasal cannula
Premature infants to adult
Low flow; 25 ml/min–6.0 L/min
Tolerated well by all ages
Inaccurate FiO2; low relative humidity; excessive flows may cause inadvertent CPAP in infants; precise FiO2 may be achieved with O2 blender
Incubator
Newborns ≤28 days
<0.40 FiO2, combine use with cannula or hood for precise FiO2
Low FiO2 for stable infants; neutral thermal environment for premature infants
Varying FiO2; long stabilization time; limits access to child for patient care
Oxyhood
Premature infants to ≤6 mo
0.21-1 FiO2 with O2 blender maintained at 30° C to 34° C
Warmed and humidified gas at stable FiO2 during routine patient care
Overheating may cause apnea and dehydration; underheating may cause O2 consumption; inadequate flow causes CO2 buildup; noise produced by humidification device may cause hearing loss
Mist tent
Infants to toddlers
High flow; 0.21-0.40 FiO2
Allows child movement, high humidity, cool temperatures
Isolation of child from family; wet bedding and clothes; difficult to maintain stable FiO2; risk of cross-contamination; limits patient care
Secretion Clearance Techniques
Methods
Humidity and Aerosol Therapy
Humidity Therapy
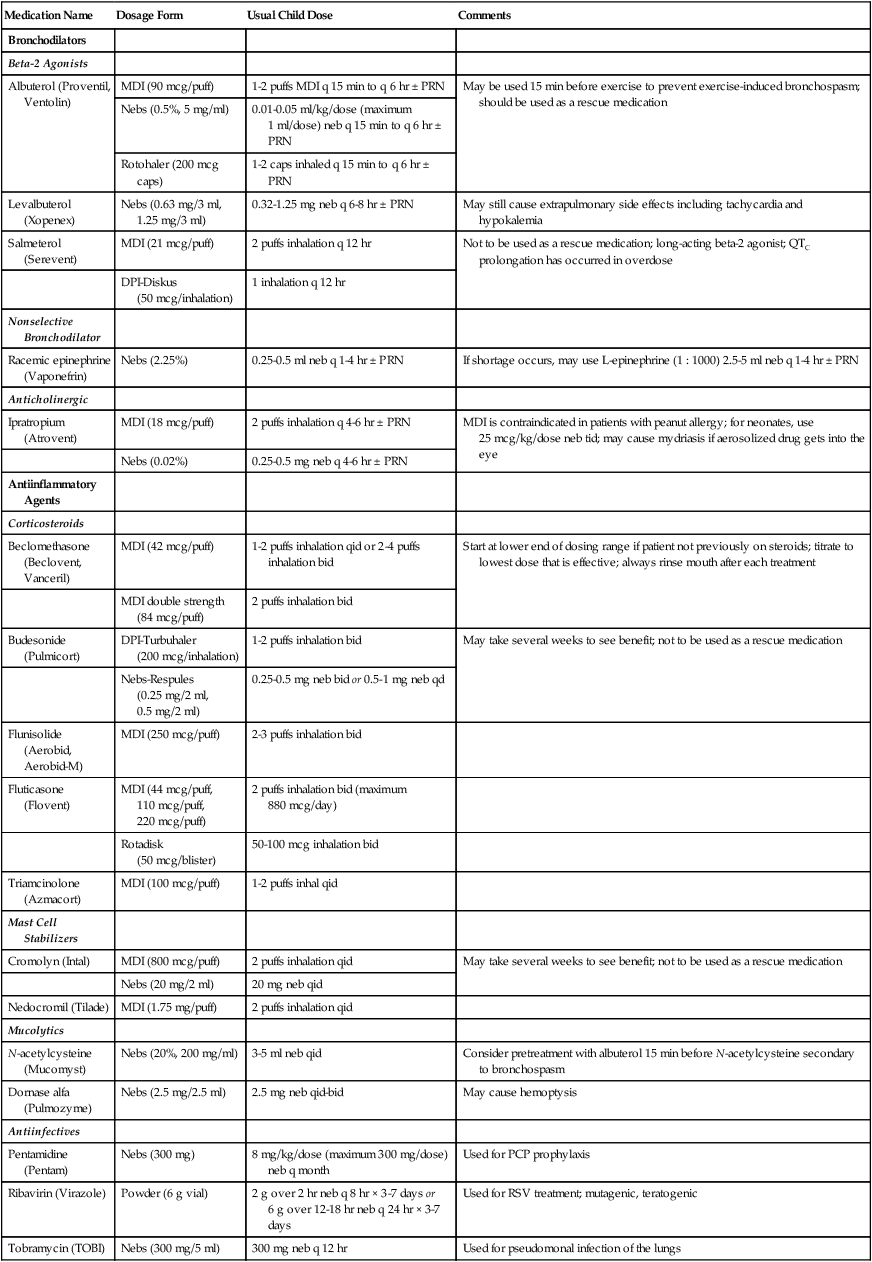
Aerosol Drug Therapy
Medication Name
Dosage Form
Usual Child Dose
Comments
Bronchodilators
Beta-2 Agonists
Albuterol (Proventil, Ventolin)
MDI (90 mcg/puff)
1-2 puffs MDI q 15 min to q 6 hr ± PRN
May be used 15 min before exercise to prevent exercise-induced bronchospasm; should be used as a rescue medication
Nebs (0.5%, 5 mg/ml)
0.01-0.05 ml/kg/dose (maximum 1 ml/dose) neb q 15 min to q 6 hr ± PRN
Rotohaler (200 mcg caps)
1-2 caps inhaled q 15 min to q 6 hr ± PRN
Levalbuterol (Xopenex)
Nebs (0.63 mg/3 ml, 1.25 mg/3 ml)
0.32-1.25 mg neb q 6-8 hr ± PRN
May still cause extrapulmonary side effects including tachycardia and hypokalemia
Salmeterol (Serevent)
MDI (21 mcg/puff)
2 puffs inhalation q 12 hr
Not to be used as a rescue medication; long-acting beta-2 agonist; QTC prolongation has occurred in overdose
DPI-Diskus (50 mcg/inhalation)
1 inhalation q 12 hr
Nonselective Bronchodilator
Racemic epinephrine (Vaponefrin)
Nebs (2.25%)
0.25-0.5 ml neb q 1-4 hr ± PRN
If shortage occurs, may use L-epinephrine (1 : 1000) 2.5-5 ml neb q 1-4 hr ± PRN
Anticholinergic
Ipratropium (Atrovent)
MDI (18 mcg/puff)
2 puffs inhalation q 4-6 hr ± PRN
MDI is contraindicated in patients with peanut allergy; for neonates, use 25 mcg/kg/dose neb tid; may cause mydriasis if aerosolized drug gets into the eye
Nebs (0.02%)
0.25-0.5 mg neb q 4-6 hr ± PRN
Antiinflammatory Agents
Corticosteroids
Beclomethasone (Beclovent, Vanceril)
MDI (42 mcg/puff)
1-2 puffs inhalation qid or 2-4 puffs inhalation bid
Start at lower end of dosing range if patient not previously on steroids; titrate to lowest dose that is effective; always rinse mouth after each treatment
MDI double strength (84 mcg/puff)
2 puffs inhalation bid
Budesonide (Pulmicort)
DPI-Turbuhaler (200 mcg/inhalation)
1-2 puffs inhalation bid
May take several weeks to see benefit; not to be used as a rescue medication
Nebs-Respules (0.25 mg/2 ml, 0.5 mg/2 ml)
0.25-0.5 mg neb bid or 0.5-1 mg neb qd
Flunisolide (Aerobid, Aerobid-M)
MDI (250 mcg/puff)
2-3 puffs inhalation bid
Fluticasone (Flovent)
MDI (44 mcg/puff, 110 mcg/puff, 220 mcg/puff)
2 puffs inhalation bid (maximum 880 mcg/day)
Rotadisk (50 mcg/blister)
50-100 mcg inhalation bid
Triamcinolone (Azmacort)
MDI (100 mcg/puff)
1-2 puffs inhal qid
Mast Cell Stabilizers
Cromolyn (Intal)
MDI (800 mcg/puff)
2 puffs inhalation qid
May take several weeks to see benefit; not to be used as a rescue medication
Nebs (20 mg/2 ml)
20 mg neb qid
Nedocromil (Tilade)
MDI (1.75 mg/puff)
2 puffs inhalation qid
Mucolytics
N-acetylcysteine (Mucomyst)
Nebs (20%, 200 mg/ml)
3-5 ml neb qid
Consider pretreatment with albuterol 15 min before N-acetylcysteine secondary to bronchospasm
Dornase alfa (Pulmozyme)
Nebs (2.5 mg/2.5 ml)
2.5 mg neb qid-bid
May cause hemoptysis
Antiinfectives
Pentamidine (Pentam)
Nebs (300 mg)
8 mg/kg/dose (maximum 300 mg/dose) neb q month
Used for PCP prophylaxis
Ribavirin (Virazole)
Powder (6 g vial)
2 g over 2 hr neb q 8 hr × 3-7 days or 6 g over 12-18 hr neb q 24 hr × 3-7 days
Used for RSV treatment; mutagenic, teratogenic
Tobramycin (TOBI)
Nebs (300 mg/5 ml)
300 mg neb q 12 hr
Used for pseudomonal infection of the lungs
Neonatal and Pediatric Respiratory Care