Fig. 14.1
Pleural lymphangitis in a chylothorax. The parietal pleura is covered with a network of ectatic lymphatic vessels (Courtesy C Boutin, Marseille, France)
The presence of lymphangitis (Figs. 14.2, 14.3, and 14.4), a disseminated pleural carcinomatosis (Fig. 14.5), and a performance status index lower than 70 have been associated with a poorer clinical course after the induction of pleurodesis (Vargas and Teixeira 1996). Finally, lung entrapment (Figs. 14.6, 14.7,and 14.8), either due to pleural loculations or to failure of pulmonary expansion, reduces the efficacy of pleurodesis, as well as increasing the risk of infections in the pleural space. Therefore, pleurodesis is not recommended under these conditions (Vargas et al. 2004).
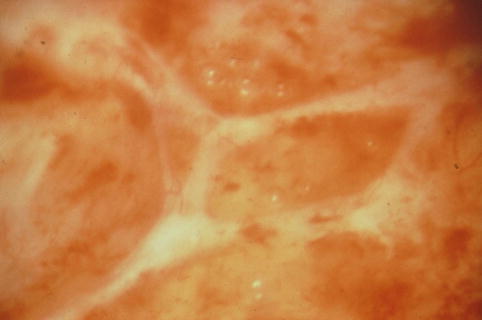
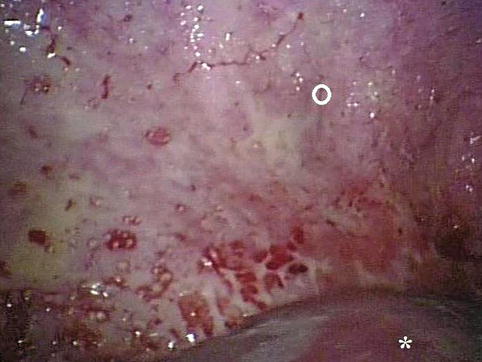
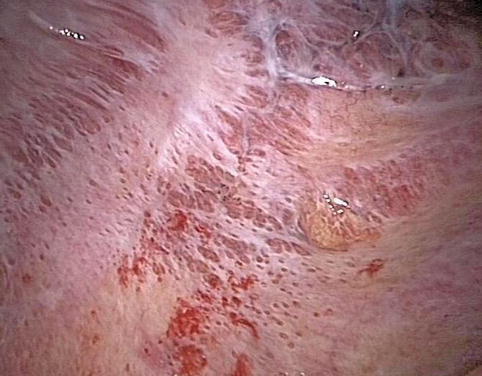
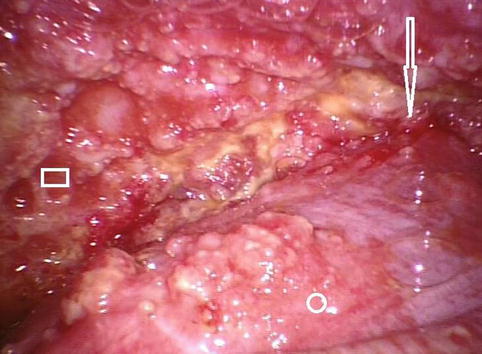
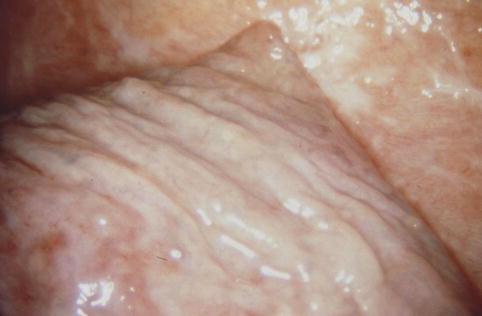
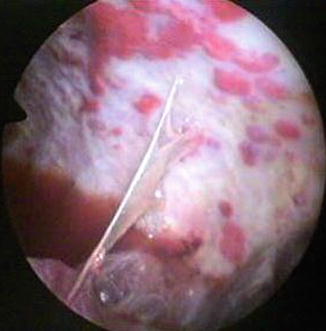
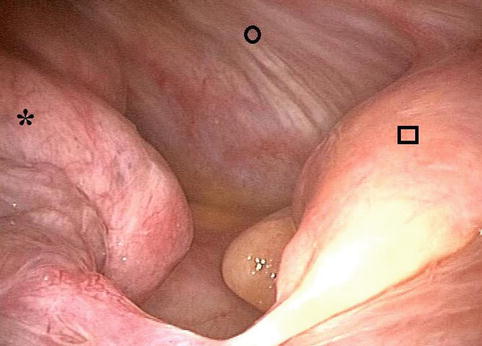

Fig. 14.2
Lymphangitis is frequently seen in cases of malignant pleural effusion. In some situations the lymphangitis can become thicker, resembling candle wax droppings (Courtesy C Boutin, Marseille, France)

Fig. 14.3
In this case of malignant pleural effusion due to a primary lung adenocarcinoma, an irregular white network can be seen over the posterior parietal pleura (○). Fibrin deposits are visible on the lung (star) (Courtesy Ph Astoul, Marseille, France)

Fig. 14.4
Malignant lymphangitis on the parietal pleura and costodiaphragmatic gutter (Courtesy Ph Astoul, Marseille, France)

Fig. 14.5
A view of the costophrenic angle (arrow). The lung (○) and the posterior parietal pleura (□) are covered by multiple nodules (Courtesy Ph Astoul, Marseille, France)

Fig. 14.6
Trapped lung (lower lobe) in a patient with a malignant pleural effusion (Courtesy C Boutin and Ph Astoul, Marseille, France)

Fig. 14.7
This picture shows pleural lymphangitis, nodules, and fibrin deposits. There is an adhesion between the posterior parietal pleural (lower part of the pleural cavity) and the lower lobe of the left lung (Courtesy C Boutin, Marseille, France)

Fig. 14.8
Even without a fibrotic inflammatory reaction of the visceral pleura, the lung can be trapped. This patient was diagnosed with a lung tumor of the lower lobe (star). This view shows the costodiaphragmatic angle with the lower part of the posterior parietal pleura (○) and the diaphragm (□) (Courtesy Ph Astoul, Marseille, France)
14.3 Types of Procedures
Pleurodesis can be achieved using a variety of stimuli: direct physical abrasion, instillation of caustic or irritating chemical substances (talc, doxycycline, silver nitrate, or bleomycin) into the pleural space, or immunological induction with Corynebacterium parvum, transforming growth factor beta (TGF-ß) or interferon-alpha 2 (IFN-a 2).
14.3.1 Mechanical Stimuli
Among the mechanical techniques employed, abrasion is the principal method. Abrasion is carried out during a surgical intervention, whether conventional or video-assisted, where the surgeon exfoliates the pleural mesothelium, creating friction with a rough-surfaced material (e.g., gauze). This irritation results in the desquamation of the mesothelium and activation of the inflammatory and coagulation pathways, with subsequent proliferation of fibroblasts and collagen deposition. This will result in pleural symphysis.
Pleural abrasion is not currently used in the control of recurrent neoplastic pleural effusions due to its lower efficacy, as well as to the high risk of bleeding in the regions involved by tumor and in addition, due to the theoretical possibility of malignant dissemination. Another inconvenience of pleural abrasion is that it requires surgical intervention.
14.3.2 Chemical Stimuli
Pleurodesis induced by chemical stimuli was first carried out at the beginning of the last century. There are references that, in 1901, Spengler injected silver nitrate into the pleural cavity for the control of recurrent pneumothorax (Vargas and Teixeira 1996).
Apparently, talc was first introduced into the pleural cavity, with the objective of collapsing the existing residual space after pulmonary TB by Bethune in 1935 (Vargas and Teixeira 1996). Since then, various substances have been used to induce pleurodesis. Chemical stimulation can be performed through various routes of access – pleurodesis can be achieved thoracosopically or through simple drainage.
Talc is classically considered the most efficacious sclerosant. When compared with other agents, it presents a relative risk of 1.34 for therapeutic success (95 % confidence interval: 1.16–1.55) with a success rate of over 90 % in most studies (Shaw and Agarwal 2004; Dresler et al. 2005). Talc has been considered the agent of choice, since it presents many of the characteristics cited in the definition of an ideal agent (low cost, wide distribution, easy administration, high efficacy, and low rate of side effects). Despite its low rate of complications, its use has been associated with acute respiratory distress syndrome. It is believed that this complication is related to the size of the talc particles. The smaller ones would be more easily absorbed from the pleural cavity and distributed throughout the circulation, resulting in a greater risk of remote complications and induction of cytokine-based systemic inflammatory reactions (Rehse et al. 1999; Maskell et al. 2004).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


