The aim of this study was to define the incidence of left ventricular thrombus (LVT) and its predictors in the contemporary era of primary percutaneous intervention (pPCI) and contrast echocardiography. We retrospectively analyzed 1,059 patients presenting with ST-elevation myocardial infarction (STEMI) to our tertiary cardiac center and treated with pPCI. Preprocedural pharmacology and procedural technique (including access route, the use of drug-eluting stents, and thrombectomy) were at the operators’ discretion. Transthoracic echocardiography was performed before discharge; echo contrast agent was used when appropriate. LVT was detected in 42 subjects (4%). There were no significant differences in baseline demographics or pre-PCI clinical features between the 2 groups. Post-treatment, mean ejection fraction (EF) in patients with LVT was 35 ± 8.4% and in those without LVT was 47 ± 10%, p <0.001. Thirty-seven patients (88%) in the LVT group presented with an anterior STEMI versus 471 patients (42%) in the without LVT group (p <0.001). Apical akinesis was noted in all patients with LVT irrespective of the principal location of the MI. Multivariate analysis predictors of LVT were reduced EF, anterior site of MI, and the use of platelet glycoprotein IIb/IIIa inhibitors. After diagnosis of LVT, patients were treated with warfarin for 3 to 6 months. No significant difference in mortality was detectable at discharge between the 2 groups. In conclusion, in the contemporary era of pPCI, the incidence of LVT in patients with STEMI is significantly lower than that of the previous (thrombolysis) literature. The early presence of LVT is more likely in patients with anterior STEMI (involving the apex) and reduced EF.
In the last 2 decades, the treatment of ST-elevation myocardial infarction (STEMI) has dramatically changed with early reperfusion therapy increasing myocardial salvage and improving survival. However, despite this, left ventricular thrombus (LVT) is still reported as a relatively frequent complication. Recent studies have produced contradictory results with a variably reported incidence from 2.5% to 15%, up to 23.5% in anterior MI. Two-dimensional transthoracic echocardiography (TTE) is the most effective diagnostic technique in this setting and can be quickly performed at the bedside but can lead to misdiagnoses due to difficult echo windows, artifact, or the misinterpretation of LV trabeculations or chordae. TTE has been shown to have a sensitivity of only 33% but a specificity of 91%. However, the diagnostic capability of TTE has dramatically changed because contrast agents and contrast-specific imaging modalities have become available. Contrast fills the LV cavity enhancing the endocardial border, thereby facilitating the recognition of mural thrombus. The aim of this study was to define the current incidence of LVT and identify its predictors in the contemporary era of primary percutaneous intervention (pPCI) with modern contrast echocardiography.
Methods
We retrospectively analyzed 1,059 patients urgently transferred for pPCI to Kings College Hospital with a diagnosis of STEMI. Kings College Hospital is a tertiary referral center with a busy and mature (>10 years), 24 hours, 7 days per week pPCI service. Patients were included when they fulfilled the diagnostic criteria for STEMI: characteristic symptoms of myocardial ischemia in association with persistent electrocardiographic ST elevation and subsequent release of biomarkers of myocardial necrosis or imaging evidence of new loss of viable myocardium or new regional wall motion abnormality.
Patients were excluded when pPCI was not performed for clinical reasons. All patients included in the study received preprocedural pharmacology at the operators’ discretion but within current guidelines in all cases. The PCI technique (including the use of drug-eluting stents, procedural use of glycoprotein (GP) IIb/IIIa inhibitors, and aspiration thrombectomy) was also at the discretion of the interventional operator. In all cases, aspirin (300 mg loading dose followed by 75 mg daily) and Clopidogrel (300 or 600 mg loading dose followed by 75 mg daily) or Prasugrel (60 mg loading dose followed by 10 mg daily) were administered. Periprocedural anticoagulation comprised unfractionated heparin (50 U/kg), bivalirudin (0.75 mg/kg iv bolus and 1.75 mg/kg/h infusion), or abciximab (0.25 mg/kg iv bolus and 0.125 μg/kg/min). Heparin was used in all cases where bivalirudin was not. Coronary thrombus aspiration was performed with either Pronto (Vascular Solutions Inc., Minneapolis, Minnesota) or Export (Medtronic Inc., Minneapolis, Minnesota) aspiration catheters. All patients underwent TTE 24 to 48 hours after pPCI. TTE was performed with either iE33 (Philips Medical Systems, Andover, Massachusetts) or Vivid 7, Vivid 9 (GE Healthcare, Waukesha, Wisconsin) echocardiography machines. Low mechanical index, contrast-specific imaging modalities were available on all echo systems used. The images obtained were analyzed using dedicated image-viewing software (Xcelera R3.2L1 SP2 version, Phillips). Each study was performed and reported in accordance with the European Association of Echocardiography guidelines. The regional wall motion was assessed using the American Society of Echocardiography 16-segment model of the left ventricle.
Sonovue (Bracco, Altana Inc., Milan, Italy) or Optison (General Electric Fairfield, Connecticut) was used as ultrasound contrast agents to confirm or exclude the echocardiographic diagnosis of LVT in selected cases. These agents were delivered by bolus and were predominantly utilized when thrombus could not be confidently excluded or confirmed on TTE images due to the presence of suboptimal nonenhanced images or when 2 or more contiguous segments were not seen.
Once LVT was diagnosed, the images were reviewed by a senior imaging physician, and the findings were monitored by serial TTE with the use of contrast if necessary. On noncontrast images, LVT was defined as an echo-dense mass adjacent to an area of akinetic or aneurysmal myocardium with distinct margins from the LV wall and the LV cavity, visible in at least 2 or more transducer positions or modified views. On contrast images, thrombus appears as a dark linear or protruding structure, adjacent to akinetic myocardium, surrounded by opacified (bright) blood in the left ventricular cavity ( Figure 1 ).
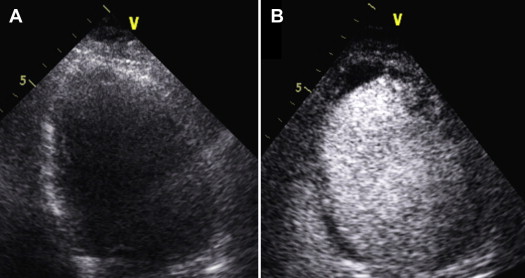
All statistical analyses were performed using STATA, version 12.1 (StataCorp LP, College Station, Texas). Continuous variables are presented as mean with SD, and frequency counts and percentages for categorical variables. Patients’ characteristics in LVT and in no LVT group were compared using chi-square test for qualitative variables and Student t test for quantitative variables. Multiple logistic regression analysis was used to identify independent predictors of thrombus formation. Statistical significance was assumed when p <0.05. Odds ratios (OR) and corresponding 95% confidence intervals (CIs) were reported. We used median and interquartile range for symptom-to-balloon time as these data were not normally distributed.
Results
A total of 1,059 subjects with STEMI underwent pPCI at Kings College Hospital in the study period. Patients were enroled from January 1, 2009 to August 31, 2012. The data were analyzed retrospectively. The mean age of the total population was 62 ± 13 years, and 26% of the patients were women.
All patients underwent 2-dimensional TTE. LVT was confidently confirmed in 25 cases (2%) and excluded in 885 cases (84%), and in the remaining 149 patients (14%), a contrast study was performed to clarify the diagnosis. The use of contrast ruled out LVT in 132 cases (OR 12%, 95% CI 10.5% to 14.6%) and confirmed it in 17 cases (OR 2%, 95% CI 0.9% to 2.5%). LVT was therefore finally diagnosed in a total of 42 patients (OR 4%, 95% CI 2.9% to 5.3%) by TTE with or without the use of echo contrast.
The baseline characteristics for patients with LVT and without LVT, listed in Table 1 , were not dissimilar between the 2 groups. No thromboembolic events were observed in either group during the index hospital admission.
| Continuous Variables | Thrombus | Total | p Value | ||||
|---|---|---|---|---|---|---|---|
| No | Yes | ||||||
| (n = 1,017) | (n = 42) | (n = 1,059) | |||||
| Mean | SD | Mean | SD | Mean | SD | ||
| Age (yrs) | 62 | 13 | 62 | 14 | 62 | 13 | 0.984 |
| Ejection fraction | 47 | 10 | 35 | 8 | 46 | 10 | <0.001 |
| Median | IQR | Median | IQR | Median | IQR | p Value | |
| Symptoms-to-balloon time | 198.5 | 144–290 | 200 | 145–290 | 198 | 144–290 | 0.110 |
| Discrete variables | n | n | n | p Value | |||
| Men | 749/1,017 | (74) | 35/42 | (83) | 784/1,058 | (74) | 0.208 |
| White | 826/1,017 | (84) | 38/42 | (90) | 864/1,025 | (84) | 0.384 |
| Hypercholesterolemia | 465/953 | (49) | 18/42 | (43) | 483/995 | (49) | 0.529 |
| Hypertension | 470/954 | (49) | 18/42 | (43) | 488/996 | (49) | 0.435 |
| Smoker | 402/963 | (42) | 17/39 | (44) | 419/1,002 | (42) | 0.869 |
| Diabetes mellitus | 152/978 | (16) | 3/42 | (7) | 155/1,020 | (15) | 0.186 |
| Previous angina pectoris | 209/982 | (21) | 11/41 | (27) | 220/1,023 | (22) | 0.437 |
| Previous myocardial infarction | 111/986 | (11) | 3/41 | (7) | 114/1,027 | (11) | 0.612 |
| Previous coronary artery by pass | 21/1,001 | (2) | 0/42 | (0) | 21/1,043 | (2) | 1.000 |
| Previous coronary angioplasty/stent | 97/1,002 | (10) | 1/41 | (2) | 98/1,043 | (9) | 0.169 |
| Heart failure | 15/968 | (2) | 2/42 | (5) | 17/1,010 | (2) | 0.155 |
| ACEI or ARB use | 188/928 | (20) | 6/40 | (15) | 194/968 | (20) | 0.546 |
| Statin use | 232/932 | (25) | 8/39 | (21) | 240/971 | (25) | 0.705 |
| β-blocker use | 101/938 | (11) | 3/40 | (8) | 104/978 | (11) | 0.792 |
Thirty-seven patients (88%) in the LVT group presented with an anterior MI versus 471 patients (42%) in the without LVT group (p <0.001). Mean ejection fraction (EF) was significantly lower in patients with LVT ( Table 1 ). Apical akinesis was noted on TTE in all patients with LVT irrespective of the main location of the MI.
The use of bivalirudin and coronary thrombus aspiration devices did not differentiate between the 2 groups. The use of GP IIb/IIIa inhibitors was significantly higher in the LVT group. Thrombolysis in myocardial infarction-3 flow postprocedure was achieved with higher frequency in patients without LVT although this was not statistically significant (p = 0.223; Table 2 ). Multivariate analysis was used to define the independent predictors of LVT formation. The variables with higher predictive value shown with the univariate analysis were introduced into the model. These consisted of gender, family history for cardiovascular disease, diabetes, previous PCI, EF, symptom-to-balloon time, anterior location of MI, periprocedural use of GP IIb/IIIa inhibitors, and presence of thrombolysis in myocardial infarction-3 flow in the culprit vessel post-PCI ( Table 3 ). Receiver operating characteristic curves plotted to predict the accuracy of the multivariate analysis model. The predictive value of the model is high (area under the receiver operating characteristic curve = 90%; Figure 2 ). The independent predictors of LV thrombus formation were therefore anterior MI, reduced EF, and the use of GP IIb/IIIa inhibitors. A prolonged symptom-to-balloon time was a borderline predictor.
| LV Thrombus | Total | p Value | |||||
|---|---|---|---|---|---|---|---|
| No | Yes | ||||||
| (n = 1,017) | (n = 42) | (n = 1,059) | |||||
| Cardiac arrest | 130/850 | (15) | 4/36 | (11) | 134/886 | (15) | 0.638 |
| Anterior site of myocardial infarction | 417/994 | (42) | 37/42 | (88) | 454/1,036 | (44) | <0.001 |
| Cardiogenic shock | 83/1,015 | (8) | 4/42 | (10) | 87/1,057 | (8) | 0.772 |
| Previous use of Clopidogrel | 41/964 | (4) | 2/41 | (5) | 43/1,005 | (4) | 0.693 |
| Bivalirudine | 415/968 | (43) | 18/42 | (43) | 433/1,010 | (43) | 1.000 |
| Glycoprotein IIb/IIIa inhibitors | 387/1,008 | (38) | 28/42 | (67) | 415/1,050 | (40) | <0.001 |
| Atherothrombus removal device | 722/1,017 | (71) | 30/42 | (71) | 752/1,059 | (71) | 1.000 |
| TIMI III flow postprocedure | 888/966 | (92) | 32/37 | (86) | 920/1,003 | (92) | 0.223 |
| Death at discharge | 56/1,017 | (6) | 1/42 | (2) | 57/1,059 | (5) | 0.722 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


