Immune category
Age: <12 months
Age: 1–5 years
Age: 6–13 years
CD4/μl (%)
CD4/μl (%)
CD4/μl (%)
No immunosuppression
≥1500 (≥25)
≥1000 (≥25)
≥500 (≥25)
Moderate immunosuppression
750–1499 (14–24)
500–999 (14–24)
200–499 (14–24)
Severe immunosuppression
<750 (<15)
<500 (<15)
<200 (<15)
The virus also infects monocytes, macrophages, and dendritic cells, but the destructive effect on them is not as strong as on CD4+ T lymphocytes; therefore, these cells become a repository for the virus and enable it to be distributed throughout the rest of the body. When these cells are infected, phagocytosis, chemotaxis, and antigen presentation are affected. These anomalies increase susceptibility to infections.
HIV produces an overstimulation of B lymphocytes, which results in polyclonal hypergammaglobulinemia mainly based on IgG, which appears earlier than the selective loss of CD4+ T lymphocytes. This polyclonal hypergammaglobulinemia is inefficient and behaves as functional hypo- or agammaglobulinemia, leading to a high infection rate from encapsulated bacteria and an insufficient response to antibodies, which explains the recurrence of infections and the deficient response to immunization.
In lung tissue, as in the peripheral bloodstream, the virus also infects lymphocytes and causes a reduction in CD4+ T lymphocytes; in addition, there is an increase in CD8+ T lymphocytes that infiltrate interstitial and alveolar spaces.
Clinical Aspects
Respiratory system compromise
Infections not suggestive of AIDS |
Persistent or recurrent upper acute respiratory infections (otitis, sinusitis) |
Acute pneumonia, pleuropneumonia (1 episode in 1 year) |
Bronchial infections, lung infections from herpes simplex virus (in <1 month of life) |
Bronchial infections, lung infections from cytomegalovirus (in <1 month of life) |
Lung infection caused by Mycobacterium tuberculosis |
Infections suggestive of AIDS |
Bacterial pneumonia (2 or more episodes in 2 years) |
Pneumonia from Pneumocystis jirovecii |
Pneumonia from cytomegalovirus (in >1 month of age) |
Bronchitis, pneumonitis from herpes simplex virus (in >1 month of age) |
Pneumonia from Candida |
Disseminated or extrapulmonary infection caused by Mycobacterium tuberculosis |
Lung infection fromto non-tuberculous Mycobacterium (M. avium complex and others) |
Noninfectious causes |
Chronic lung disease |
Lymphoid interstitial disease/lymphoid interstitial pneumonitis |
Bronchiectasis |
Immune reconstitution syndrome |
Tumors |
The etiology of lung infections as well as their severity, development, and outcome depend on the patient’s immunosuppression level. In two thirds of the children infected perinatally, who are neither diagnosed nor treated with antiretroviral drugs in a timely fashion, viral load increases rapidly during the first months of life. Afterward, the absolute count of CD4+ T lymphocytes drops sharply, which leads to the development of severe opportunistic infections with a high mortality. These patients are known as rapid progressors. Another patient subgroup has a slower and more benign progression, with lower viral loads and relatively high CD4+ T-lymphocyte counts that prevent the development of opportunistic infections. They develop bacterial infections that are similar to normal children, but recurrent. These cases are referred to as slow progressors. In a third group, progression is intermediate.
Unfortunately, no reliable markers have been identified earlier than 24 months of age that would enable clinicians to identify the progression and behavior of the virus in each individual. Therefore, the current recommendation is to initiate antiretroviral treatment in all children under 1 year of age with a confirmed infection to prevent immunological deterioration.
Infectious Complications
Infections in HIV-positive children can have a bacterial, viral, or fungal etiology. In general, they present with rapid-onset respiratory symptoms, increased breathing effort, fever, usually with a clear chest X-ray; in contrast, noninfectious complications are characterized by progressive and slow starting respiratory symptoms with chest X-ray shadows.
Bacterial Infections
Upper airway bacterial infections (sinusitis and otitis media) are more common in children with HIV and can be classified as clinical stage A or mildly symptomatic. Recurrent respiratory bacterial infections must cause clinicians to suspect an HIV infection when they are associated with other unspecific manifestations such as adenopathy, chronic parotid swelling, hepatomegaly, or splenomegaly.
Among the severe bacterial infections, bacterial pneumonia is the most frequent infectious entity in all stages of the disease, even without immune deterioration, and especially during the first 12 months of life.
During the AIDS stage, there is a high risk of complications that is directly proportional to the viral load and inversely proportional to the absolute count and percentage of CD4+ T lymphocytes. Its incidence has been reduced from 11 to 2 per 100 children-year with HAART and immunizations (especially the conjugate Streptococcus pneumoniae and Haemophilus influenza type b vaccines), but it remains the most prevalent infection in this group. The etiology of bacterial pneumonia is diverse. Even though it is not easy to make an etiological diagnosis, authors have described infections caused by gram-positive, gram-negative, and mixed bacteria.
The main cause of bacterial infection is Streptococcus pneumoniae, although Staphylococcus aureus now is more important given the high levels of oxacillin (50%) and clindamycin resistance (22–30%). The most common gram-negative bacteria are Haemophilus influenzae (encapsulated and non-encapsulated), Escherichia coli, Salmonella non-typhi, and Pseudomonas aeruginosa. Other agents that cause lower respiratory infections include Mycoplasma pneumoniae and Chlamydophila pneumoniae.
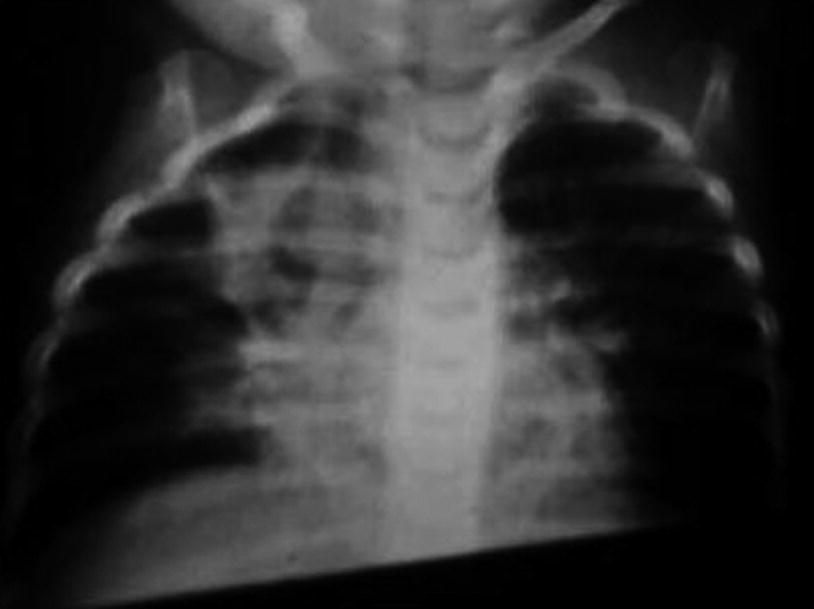
Necrotizing pneumonia. Chest X-ray of 2-year-old girl shows a bilateral condensation pattern with a right apical abscess. Streptococcus pneumoniae was isolated in the bronchoalveolar lavage and hemoculture
Any episode of pneumonia or confirmed bacterial pleuropneumonia indicates clinical stage B or moderate symptomatology, and two or more episodes over a 2-year period indicate clinical stage C or severe symptomatology.
Treatment for bacterial pneumonia is similar to that recommended for the general pediatric population. All guidelines propose using amoxicillin as an initial antibiotic therapy associated with macrolides when clinical or radiological signs suggest the presence of an Mycobacterium pneumoniae or Chlamydia pneumoniae infection. In the case of Haemophilus influenzae (encapsulated or non-encapsulated) and Moraxella catarrhalis, the use of clavulanic acid or sulbactam is recommended.
Primary prophylaxis involves immunizations and the administration of intravenous immunoglobulin for patients with absolute (IgG <400 mg/dl) or functional hypogammaglobulinemia (insufficient IgG production in response to specific immunizations).
Secondary prophylaxis is conducted with intravenous immunoglobulin or trimethoprim sulfamethoxazole (TMP-SMX) in patients with more than two confirmed episodes of severe bacterial infections.
The conjugate pneumococcal vaccine has caused a 60-fold reduction in invasive pneumococcal disease in this population versus a 15-fold reduction in normal children. Patients who do not receive HAART display a marked decrease in anti-pneumococcal titers over a period of 2 to 4 years; therefore, periodic reinforcement is recommended. It is useful to conduct an anti-pneumococcal antibody titer test in patients with a history of frequent or severe pneumonias to indicate the reinforcement of the 13-valent conjugate vaccine, followed by the administration of the 23-valent polysaccharide vaccine 6–8 months later in children with low titers. The conjugate H. influenzae type B vaccine, included in immunization programs, has had a positive impact on the incidence of invasive disease in these patients.
Mycobacterium Infections
The global reemergence of tuberculosis (TB) resulting from the AIDS epidemic, among other factors, is currently followed by WHO as a global emergency. TB/HIV coinfection, manifested as either latent tuberculous infection or as active tuberculous disease, is a major public health problem. Estimates suggest that, in 2009, 1.1 million new coinfections had occurred globally and 24,000 in the Americas. Because of its high prevalence, every time a TB case is diagnosed, it is necessary to rule out HIV infection, and vice versa. In HIV-positive children, it is generally a primary infection transmitted by an adult with an active infection. High risk results from the reduction in T1 lymphocytes, the subsequent limited production of interferon-gamma (IFN-γ), and the functional alteration of the macrophage systems, all of which constitute the basic defenses of the body against Mycobacterium tuberculosis. As cellular immunodeficiency affects patients with HIV, the infection is poorly controlled, which fosters its progression to TB disease, its dissemination, the development of extrapulmonary forms, its recurrence, and its high fatality rate (causing the death of up to one third of patients with AIDS). In addition, the reservoir that these patients constitute increases the risk of transmission to the community. TB bacillus induces the macrophage to produce tumor necrosis factor and interleukins 1 and 6, proinflammatory substances that foster the replication of HIV in infected areas, thus increasing viral load and accelerating the progression to AIDS and death. In this patient group, more treatment failures are observed because of the increasing multiresistance of the TB bacillus (resistance to rifampicin and isoniazid).
TB can appear at any point of the HIV infection in children, either in the non-AIDS stage (lung compromise) or in the AIDS stage (disseminated or extrapulmonary compromise), and either with or without immunological deterioration.
Latent TB infection occurs when the tuberculin test (PPD) or the interferon-γ test are reactive in an asymptomatic patient, with a negative bacillus smear and normal chest X-rays. Active TB disease occurs when the patient is symptomatic, sputum cultures are positive, and chest X-rays are abnormal. In addition, PPD can be positive.
Clinical presentation in HIV-positive children is atypical and severe, depending on the level of immunosuppression. Miliary compromise and extrapulmonary manifestations are more common than in HIV-negative children. In general, they appear in children with a history of contact with TB or diagnosed TB and symptoms such as fever, major weight loss, sweating, tachypnea, and unsolved condensing or interstitial pneumonia with persistent X-ray signs despite the treatment administered.
Diagnosing TB in children is hard, especially in HIV-positive children. If the treating physician suspects TB, it is essential to obtain samples to confirm its etiology through bacteriology.



