Fig. 5.1
(a) Theodore Tuffier, (b) Alexis Carrel, and (c) Henry Souttar
A 26-year-old male was prepped for surgery on July 13, 1912 (Table 5.1). “I observed, in a young man, a grave and rapidly progressive aortic stenosis. On the repeated request of his physician I decided to explore it” [11]. After opening the chest of the patient, Tuffier set aside his scalpel, grasped the beating heart, and invaginated the aorta with his little finger. “The vibration was intense: I reached the stenosis and very easily carried out a gradual dilatation by slowly introducing the little finger into the strictured ring, the vibrations under the finger being intense; I abstained from trying to divide the stricture as I did not consider experimental enquiry sufficiently advanced. I did not expect to attain any result. The patient was well in a few days; he improved temporarily and is still alive. I saw him three months ago” (Fifth Congress of the International Society of Surgery, Brussels, July 1920). The last report we could find on this patient indicated that he was still alive in 1920 [12].
Table 5.1
Summary of operations for valvular disease (1913–1928)
Author/surgeon | Date | Diagnosis | Method/instrument | Results after operation |
|---|---|---|---|---|
Doyen | 1913 | Congenital PS; VSD | Tenotome | Died within hours |
Tuffier | 1914 | AS | Finger dilation | Recovery/improved |
Cutler, Levine | 5/20/1923 | MS | Tenotome | Died 4 1/2 years |
Allen, Graham | 8/7/1923 | MS | Cardioscope | Operative death |
Cutler, Levine, Beck | 10/7/1923 | MS | Tenotome | Died 30 h |
Cutler, Levine, Beck | 1/12/1924 | MS | Tenotome | Died 20 h |
Cutler, Levine, Beck | 2/25/1924 | MS | Cardiovalvulotome | Died 6 days |
Cutler, Levine, Beck | 6/11/1924 | MS | Cardiovalvulotome | Died 3 days |
Souttar | 5/6/1925 | MS; AI | Finger dilation | Recovery/improved |
Pribram | 11/14/1925 | MS; Endo | Cardiovalvulotome | Died 6 days |
Cutler, Beck | 12/8/1926 | MS | Cardiovalvulotome | Died 45 h |
Cutler, Beck | 6/15/1928 | MS | Cardiovalvulotome | Died 3 h |
5.3.2 The First Successful Closed Surgery: Mitral Stenosis
In 1925, the general surgeon Henry Souttar (1875–1964) was asked to consider a 15-year-old girl afflicted with rheumatic mitral valve disease at the London Hospital (Fig. 5.1c) [13, 14]. Lilian Hine was first admitted to the hospital in 1921 with chorea and mitral stenosis. Her subsequent history was one of many relapses. She was admitted once again in 1925 with heart failure. By this time, Elliot Cutler had already experienced one palliative success and four failures with his technique of incising/resecting portions of the valve through the left ventricle (Table 5.1). However, Souttar’s surgery is considered the first successful mitral repair because his approach would eventually prove to be superior to Cutler’s.
The auricular appendage was clamped and incised to allow the left forefinger to be inserted, “and the appendage was drawn over the finger like a glove by means of the sutures.” Upon entering this appendage, Souttar immediately felt profound regurgitation and concluded that the extent of stenosis was only moderate. To avoid worsening the insufficiency, Souttar dilated the orifice using his finger instead of employing the hernia bistoury which had been set aside to section the valve [13]. While digital dilation succeeded in breaking down the adhesions between the cusps, the patient still suffered from a considerable amount of insufficiency and fought another bout of rheumatic fever only a year later. The patient lived for 7 years total following the operation, eventually passing away from cerebral embolism [7].
Souttar’s background in engineering enabled him to make a famous argument against the stance that surgery could not heal medical illnesses. “I felt an appreciation of the mechanical reality of stenosis and regurgitation which I never before possessed… I could not help being impressed by the mechanical nature of these lesions and the practicability of their surgical relief” [13]. He noted that most discussions on valvular surgery focused only on the problems of surgery, and he instead chose to underscore the clinical work of Elliot Cutler and the experimental work of Duff Allen as evidence of the practicality of valvular surgery. Souttar was knighted in 1949 for his daring efforts in surgery, including the introduction of atraumatic sutures [4]. Unfortunately, physicians refused to ever allow this great surgeon to operate on valves again, despite his successes. Souttar later reflected on his experience with valvular surgery: “… it is no use to be ahead of one’s time” [7]. Despite Souttar’s success, Cutler’s approach would dominate and hinder both experimental and clinical studies until after World War II, when Bailey would repopularize finger fracture valvuloplasty.
5.4 Cutler’s Era (1923–1928): Exchanging Stenosis for Insufficiency
Elliot Cutler (1888–1947) was the first surgeon to repeatedly attempt mitral valve surgery, and his method and theory on the surgical treatment of mitral stenosis would prove extremely influential (Fig. 5.2a) [15, 16]. Of the ten reported surgeries during this time period (1923–1928), seven were completed by Cutler and his assistants Samuel Levin and Claude Beck. Cutler followed the advice of Brunton and Samways and created notches in the valve cusps, approached the heart through the left ventricle with a tenotome in his first three patients, and later created a valvulotome for use in the last four patients [17]. Cutler preceded his clinical work with canine experiments in the famous lab of Harvey Cushing (studying stenosis since 1905) (Fig. 5.2a). As a result of his experimentation on the valves of healthy animals, Cutler was confident that the only effective treatment would be valvotomy with subsequent production of a degree of insufficiency. In total, they performed operations on 30 dogs, 24 of which survived. Cutler and his associates were ready to take on their first clinical case.
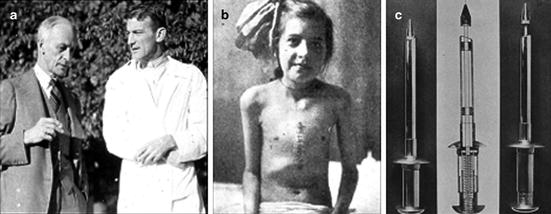

Fig. 5.2
(a) Harvey Cushing (left) and Elliot Cutler (right), (b) Cutler’s first and only successful mitral valve surgical patient, and (c) tenotome devised by Cutler and Beck to remove and retain large sections of valve tissue
On May 20, 1923 Cutler performed the first operation at the Peter Bent Brigham Hospital in Boston [16]. The 11-year-old girl had experienced dyspnea for 3 years and had been confined to her bed for 8 months, coughing up shocking amounts of blood. In each case (we operated on), the prognosis was distinctly bad (Fig. 5.2b). A curved tenotome knife was inserted between control sutures at the apex of the left ventricle, and each valve was incised. After closure, a serious tamponade developed; fortunately, the patient recovered, gained ten pounds, and was relieved of hemoptysis. Her life was prolonged more than 4 years. However, during that time, she had a restricted level of activity and persistent signs of mitral stenosis. Autopsy would later show a remarkably enlarged heart, particularly on the left side. It is likely that this patient lived because the valvular incision was relatively small; accordingly, the degree of regurgitation was tolerable for a short time.
As Cutler and associates continued with their clinical trials, they experienced difficulties with tamponade after closure, effectively punching through calcified and fibrosed valves, difficulty with orienting their instrument blindly, and keeping their decidedly ill patients alive through the significant trauma of sternotomy. Cutler studied each casualty and sought to improve his techniques in the next surgery; still, every patient died. These deaths may have been compounded by the valvulotome, a tool created to punch through the heavily calcified valve tissue and resect a large portion of the leaflet (Fig. 5.2c). In Cutler’s final report of all surgeries up to 1929 (Table 5.1) [16], the surgeons expressed their frustration over the lack of a proper animal model for mitral stenosis. Without an animal model and with only one patient surviving after the surgery, it was impossible to tell if valvotomy with a valvulotome produced satisfactory changes in the mechanical dynamics of the circulatory system. “It may be that the cardiovalvulotome with its actual removal of a piece of valve creates a too sudden change… all the changes created by nature are slow and gradual…” [16].
While disheartened, they called upon others to continue their work. “It is our conclusion that the mortality figures alone should not deter further investigation both clinical and experimental, since they are to be expected in the opening of any new field for surgical endeavor”[16]. It was also clear that thoracic surgical techniques needed desperate attention, as the results of Cutler’s study were blurred by death from complications following sternotomy, opening/closure of the pericardial sac, and incision into the heart itself. Respectable surgeons interested in valvular surgery turned to their laboratories to complete these essential experimental tasks.
5.5 Bailey, Harken, and Brock (1948–1957): Moving Away from Iatrogenic Insufficiency
The next phase of clinical valvular heart surgery would not begin for another 20 years, when a new generation of war-emboldened surgeons emerged onto the clinical field. Charles Bailey is remembered for performing what he called the first transatrial mitral commissurotomy on June 10, 1948 [18], and Dwight Harken (1910–1992) is noted for performing his mitral valvuloplasty on June 16, 1948 [19] (Fig. 5.3a, b). Russell Brock (1903–1980) performed one of the first successful valvulotomies for pulmonary stenosis on February 16, 1948, and later the first operation for the treatment of infundibular stenosis [20, 21] (Fig. 5.3c). These men, among others, began an era of reliable surgery on all four valves with a steady decrease in mortality and an attempt at objectively measuring patient improvement. Note that lesions of the tricuspid valve were comparatively rare; as such, this valve was never specifically targeted by any one surgeon during this time period.


Fig. 5.3
(a) Charles Bailey, (b) Dwight Harken, and (c) Russell Brock
5.5.1 A Race to Repair Mitral Stenosis
An exciting race to successfully repair mitral stenosis occurred between two surgical giants—the talented Dwight Harken in Boston and the colorful Charles Bailey in Philadelphia. While each experienced their first clinical success within days of each other, ultimately Bailey would be considered victorious by many, as he conducted his surgery first and his initial technique would eventually prove more successful.
Dwight Harken (1910–1992) was Elliot Cutler’s protégé, inheriting both the title of professor of surgery at the Peter Bent Brigham Hospital in Boston and his laboratory work on mitral stenosis [1]. Harken was well qualified to improve upon Cutler’s techniques, as he had been a surgeon at the 160th General Hospital during World War II and had reported a remarkable 139 surgeries in and around the heart without a single death [22, 23]. It was not long before Harken realized that Cutler’s technique merely traded one type of cardiac lesion for another. The conclusion was made that if an insufficiency had to be produced, then the posterior leaflet was better able to tolerate defects than the anterior leaflet. They observed that the posterior leaflet was pressed against the ventricle during systole, which would lessen any regurgitation from this portion of the valve. Harken began to advocate for a new technique called selective insufficiency, in which the commissures of the mitral valve were resected in thin wedges to treat stenosis and the anterior leaflet was always preserved [19]. This would restore movement to the stenosed valve in exchange for minimal insufficiency, which Harken believed was a necessary by-product of repair for mitral stenosis due to the influence of Cutler.
They said of their first clinical operation on March 12, 1947: “This case illustrates how completely even a carefully planned procedure can miscarry” [19]. The presence of tachycardia was later blamed for the man’s death, but in total six of Harken’s first ten patients died [23]. Only the encouragement of his colleague, Dr. Ellis, enabled him to continue operating. Harken then lost just one of the next 15 patients. Over time, Harken would learn that creation of any degree of insufficiency to treat stenosis was unnecessary and would choose to perform the technique presented by Bailey [24].
Charles P. Bailey (1910–1993) avoided Cutler’s regurgitant methods from the start [18]. His team was well prepared to begin work on mitral repair, having performed 60 cardiac operations in the canine model over 8 years. Bailey and colleagues demonstrated in the laboratory that (1) sudden creation of mitral regurgitation was tolerated poorly, (2) approach through the left auricular appendage produced fewer complications than a ventricular approach, and (3) several new instruments were useful in operations on stenotic valves.
Their method of commissurotomy was based on the rather simplified understanding of mitral valve anatomy of the day [25]. It was thought that the mitral valve consisted of two distinct leaflets separated by a linear oblique line. When a valve became stenotic, the tissue at the edges of this aperture would become fibrosed and rigid. However, if the stenosis had not progressed too far, a linear oblique incision could be made along the valve opening all the way through to the ring of soft healthy tissue which often surrounded the rigid stenotic portion. The valve would be freely hinged by this soft valvular tissue and would, in theory, return to physiological functioning without creating stenosis.
With this understanding of anatomy and plenty of experience in hand, Bailey operated on his first two patients at Hahnemann Hospital [18]. During the first surgery in 1945, they learned that the human auricular appendage in mitral stenosis can be extremely friable, after the purse string sutures tore through the wall and the bleeding could not be controlled. The second case took place in 1946, this time using digital dilation. The patient’s blood pressure at surgery was 60/50mmHg, and they died 48 h after the procedure. The valve had not been torn all the way through to healthy valvular tissue; consequently fibrin accumulated and sealed the orifice.
Unfortunately, these disastrous surgeries earned Bailey the nickname butcher of Hahnemann hospital [26]. As a result of the last surgery, his team conceived the clever idea of commissurotomy using overlapping gloves to slide a small knife safely into a sheath along the dorsum of the palpating finger [18]. Bailey had been threatened with loss of surgical privileges at Hahnemann after the first two surgeries, but was able to try his new instrument at Memorial Hospital in Delaware with his third case, Philadelphia General Hospital for a fourth case, and Episcopal Hospital in Philadelphia for the fifth case [18, 26]. The surgery for the fourth case took place on the morning of June 10 and ended in mortality due to an extremely irregular and irritable myocardium: “He had a patient with mitral stenosis at the Episcopal Hospital across Philadelphia, and he immediately scheduled that patient for surgery the same afternoon, knowing that when the morning death was publicized his cardiac surgery career was finished. He wanted one more chance; he took it” [27].
The next patient was 24-year-old Claire Ward, who had been afflicted with mild mitral stenosis for 2 years [4]. On June 10, 1948, they again employed a hooked knife between gloves to divide the commissures of the thickened noncalcified valve. The operation was completed in 80 min without problem [18]. Even the presystolic murmur disappeared, and a week later she was transported by train some 1,000 miles for the American College of Chest Physicians in Chicago. Her life was prolonged for 38 years with marked ballistocardiograph improvements during exercise and no need for digitalis [4]. Bailey suggested reoperation when recurrent stenosis first appeared 36 years later, a suggestion with which the cardiologist remarkably disagreed [4]. This method of splitting the commissures became very popular. The number of successes increased as surgeons developed a better understanding of valvular anatomy as Surgeons like Bailey eventually traded the hooked finger knife for the finger itself and coined the term finger fracture valvuloplasty.
5.5.2 Repairing Aortic Stenosis
The first surgeon to repeatedly attempt surgery for aortic stenosis was Horace Smithy. Smithy from the Medical College of South Carolina operated on seven patients to create insufficiency for the treatment of aortic stenosis (1914–1948) [28]. Smithy was the first to clinically focus on aortic stenosis since Tuffier, but he chose to follow Cutler’s techniques [2, 4]. The operations were carried out January 30, 1948, through June of 1948, with five patients surviving the procedure. Smithy ironically died from aortic stenosis before the long-term results could be determined on October 28, 1948 [28]. Smithy begged his colleague Alfred Blalock to operate on his valve, but Blalock firmly declined. He had some experimental experience with the procedure, but after his only clinical attempt died on the table before any cardiac manipulations. Two of Smithy’s five successes were known to be alive in 1955, likely due to advances in medical treatments for heart failure [4]. Smithy’s first patient, Betty Woolridge, died just 10 days after Smithy [29].
A reliable surgery for aortic stenosis would prove difficult even for Bailey and Harken to attempt, due to the proximity of the aortic valve to the coronary circulation [30]. Instruments were invented in an attempt to improve results when operating to relieve aortic stenosis. On April 3, 1952, Bailey would apply a new version of his triradiate dilator to correct aortic insufficiency. He was able to operate on a series of 29 patients with 20.7 % mortality [31]. The transventricular dilator he invented for the initial procedure was later slightly augmented by Andrew Logan in 1954 and again by Tubbs and Brock for commercial use [32]. Despite the use of these elaborate instruments, surgical treatments for aortic stenosis were largely palliative during this time period.
5.5.3 Repeated Repair Pulmonary Stenosis
Surgery of the pulmonary valve began with Baron Dr. Russell Brock (1903–1980) of Guy’s Hospital, who was the first to consistently attempt surgery on pulmonary stenosis since Doyen’s ill-fated attempt in 1913 [20] and Thomas Sellors’ single successful case in 1947 [33]. The Blalock–Taussig shunt or blue–baby operation was typically performed in cases of severe pulmonary stenosis, which resulted from a congenital defect [20]. Many were satisfied with this indirect vascular shunt approach because it allowed excellent palliation of complex congenital cardiac defects (e.g. Tetralogy of Fallot) without having to use cardiopulmonary bypass, which was still experiencing devastating problems. Why even attempt a direct repair of congenital pulmonary stenosis? In addition, it was thought that most cases of pulmonary stenosis were infundibular or subvalvular in nature and thus could not be treated with the new techniques recently developed by Harken and Bailey for mitral stenosis [20].
After a few disastrous clinical cases with the cardioscope invented by Allen and Graham in 1922 to enable direct vision within the closed heart [34], Brock developed his own version of a valvulotome and dilating forceps to incise the valve and enlarge the orifice in a blind procedure [20]. He was successful in correcting the valve of an 18-year-old girl on February 16, 1948; she had been cyanosed since birth, with a history of squatting and clubbing of the fingers characteristic of the morbus caeruleus ailment. The patient survived and was known to be doing well 2 months later, after some complications with peripheral emboli.
Even though Thomas Holmes Sellors technically performed the first successful clinical pulmonary valve operation, Brock is typically credited as he more thoroughly explored the subject and published first [33]. Brock’s other two cases were also considered to have excellent results; an 11-year-old girl was able to lead a completely normal life, and another patient was able to conceive after her operation [21]. Dr. Brock took treatment of pulmonary stenosis a step further by creating a surgery for resection of infundibular stenosis, by blindly removing the intruding cardiac wall with a punch in 1949 [21]. Of the eleven initial operations performed by Brock for this ailment, eight survivors were known to be alive and improved over a year later [35].
5.5.4 After the First 10 Years of Valve Repair
It was thought that the techniques of Bailey and Brock would essentially cure mitral, aortic, and pulmonary stenosis. However, an extensive summary by Bailey, Zimmerman, and Likoff in 1960 presented the long-term results of closed valvular repair in a less than favorable light [25]. The mortality rate of closed procedures for stenotic valves stabilized at an unsatisfactory 8.5 %. “Among surgeons with extensive experience in this field (10,000 cases in aggregate) 50 per cent of those who responded expressed dissatisfaction with existing surgical procedures” [25]. Additional problems included the following: (1) cardiologist continued to keep patients with mitral stenosis away from surgeons, (2) only valves with flexible cusps could be palliated for stenosis, and (3) iatrogenic insufficiency continued to be a problem. This was an unfortunate complication as there was no successful surgical treatment for insufficiency at the time. New approaches to palliate stenosis were attempted, including the creation of an atrial septal defect (ASD). Nonetheless, many surgeons sought the open field to improve accuracy via a direct curative approach. Henry Swan summarized this stance nicely: “That the blind but educated finger is capable of accomplishing much within the heart is to be freely admitted, and much admired; that it should be considered as the best method in the long run is absurd” [36].
5.6 Lewis, Gibbon, Lillehei, and Kirklin (1953–1955): Development of the Open Field
5.6.1 Cold Heart Logic
While many cardiac surgeons were scrambling to invent complicated pump oxygenator machines, Alfred Bigelow of the University of Toronto took a pure and simple physiological approach—hypothermia [37]. Bigelow had been confronted with the problem of the open field during his surgical training with Alfred Blalock of the University of Toronto, when he realized the inadequacy of the palliative Blalock–Taussig technique [38]. after shivering is inhibited Bigelow discovered a linear relationship between temperature and metabolism while at the University of Toronto. He believed this physiological phenomenon might be utilized to protect vital organs during prolonged periods of cardiac occlusion. Through careful experimentation on dogs and chimpanzees from 1950 to 1953, he estimated that open-cardiac surgery could safely occur for up to 15 min at 20°C. This happened to be enough time to close an ASD, a lesion which could not be treated surgically before this time.
It was F. John Lewis (1916–1993) at the University of Minnesota who would first successfully lay open the heart of a 5-year-old girl with an ASD on September 2, 1952, using hypothermia at 26°C and inflow occlusion for 6 min [39]. Lewis made numerous improvements to the methods of Bigelow to protect the myocardium and reduce the likelihood of ventricular fibrillation; as a result, the patient was known to be alive 33 years later with two children and a career as a carpenter [39]. Lewis’ hypothermic technique was adopted by numerous surgeons across the world to repair simple cardiac lesions including ASDs. From 1953 to 1960, Mustard of Toronto reported on 95 operations, Bigelow reported 50 operations, and Henry Swan of Denver reported 100 procedures, including the first operation on the aortic valve under direct vision on November 17, 1955 [39, 40]. This was an enormous first step. Progress in the open field was rapid, and surgeons continued to search for other techniques which could prolong the length of time the heart could be opened.
On October 21, 1952, the Dodrill–GMR, the first effective mechanical heart, was used to bypass the left side of the heart of a 16-year-old boy at Harper Hospital in Detroit [41]. The boy’s congenital pulmonary stenosis was successfully treated in the open field, and the patient was known to be alive and well nearly 50 years after this operation. Even though Forest D. Dodrill (1902–1997) went on to successfully treat numerous other cardiac anomalies with his pump (including the aortic and mitral valves), his success is often overshadowed [42]. The heart had been bypassed but not the lungs, which remained a challenge. Dr. Gibbon’s isolated success with the first heart–lung machine would occur just a year later and represented surgery’s greatest hope for more complex procedures such as valvular replacement, repair of insufficient valves, or repair of the more extensive congenital abnormalities.
5.6.2 The Mechanical Heart and Lungs
As the famous Dr. John H. Gibbon Jr. (1903–1973) recalls from the night of October 3, 1931 [42], during his research fellowship at Harvard, “My job that night was to take the patient’s blood pressure and pulse rate every 15 min and plot it on a chart. At 1:00 AM the patient’s condition became worse, manifested by stupor and lowered blood pressure. At 8:05 AM the blood pressure could not be measured. Churchill immediately opened the chest through an anterolateral incision on the left side… All this took place in the space of 6 min and 30 seconds. Despite the rapidity of the embolectomy, the patient died on the operating table” [42]. This experience lit a fire in Gibbon to prevent future deaths from pulmonary embolism; he imagined a machine which could sustain a patient through such a procedure by oxygenating and pumping the blood. He began to work on this machine in 1934 alongside his wife, Mary, in a surgical research lab at Massachusetts General Hospital (Fig. 5.4a). They used materials found in junk shops to build their small experimental oxygenator on a tiny budget [42]. “The animals used were cats and when our supply ran short, I can recall prowling around Beacon Hill at night with some tuna fish as bait and a gunny sack to catch any of the numerous stray alley cats which swarmed over Boston in those days” [43]. A big break came in 1946 when the Gibbons were introduced to the chairman of IBM, who supplied them with one of his best engineers as well as generous financial backing. The financing and expertise finally enabled the development of a parallel screen oxygenator large enough to support a human patient at the Jefferson Medical College.
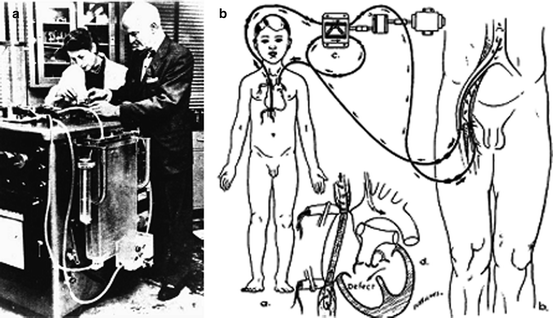

Fig. 5.4
(a) John and Mary Gibbon at the cardiopulmonary bypass machine. (b) Diagram of Lillehei’s cross circulation
Their first clinical patient died on the table, but on May 6, 1953, Gibbon placed an 18-year-old girl on bypass for 26 min as he mended an ASD [44]; she was known to be alive over 36 years later in 1986 [45]. But after nearly 20 years of working on the pump oxygenator, Gibbon was unable to replicate his success. Brilliant surgeons around the world took up his cross and continued to work on the pump oxygenator including Clarence Crafoord in Stockholm, Sweden (whom Gibbon called the night of his success), J. Jongbloed at the University of Utrecht in Holland, and Mario Dogliotti at the University of Torino in Italy (their heart–lung machine was used as early as 1951 to supplement the circulation of sick patients) [46]. Mustard of Toronto even attempted to use a freshly removed monkey lung to oxygenate his patient’s blood! Representing the Americans were Clarence Dennis (attempted first heart–lung bypass) from the University of Minnesota and Forest Dodrill (first successful heart bypass) to name a few [46].
5.6.3 Controlled Cross Circulation
C. Walton Lillehei (1918–1999) grew impatient with the progress of cardiopulmonary bypass and desired practice in open-heart repair before this technology was perfected. Lillehei was the second assistant on Lewis’ historic ASD procedure and would continue to be on the cutting edge of surgery for the rest of his career [47]. Lillehei set the bar for his academic surgical career at the University of Minnesota, when his team described a procedure called controlled cross circulation, in which the heart and lungs of a live donor (typically a parent or family member) were used to oxygenate the blood of a recipient in need of invasive cardiac surgery [48] (Fig. 5.4b). The superficial femoral artery and saphena magna vein of the donor were cannulated as well as the superior/inferior vena cava and aorta of the recipient. The cross circulation was controlled by a pump which kept the blood flow between the donor and recipient at a constant low rate. This was a radical idea indeed, although not necessarily original, having been used for end-stage uremia/toxemia [38]. The surgery put an otherwise healthy donor at risk of embolism among other surgical complications, inspiring some to dub this tactic immoral. Nevertheless, after extensive experimentation with dogs, the proposed clinical surgeries received the go-ahead from the adventurous chairman of surgery at the time Owen Wangensteen [49].
On March 26, 1954, Lillehei and his team successfully closed a ventricular septal defect in a 1-year-old boy [50]. This was no isolated success; Lillehei operated on a series of 45 patients from March 1954 to July 1955 using cross circulation (and eventually using other oxygenation techniques developed in the lab) [45]. Twenty-two of these patients were known to be alive and well more than 30 years later. Denton Cooley later said, “C. Walton Lillehei… provided the can opener for the largest picnic thoracic surgeons will ever know.” These procedures rocked the surgical world, and countless surgeons flocked to Minnesota to observe the clinical service and laboratory of Walt Lillehei. In the following years, he trained no less than 138 cardiac surgeons from 41 countries, including Christian Barnard (performed first heart transplant). While many were satisfied with controlled cross circulation at the time, others such as Dodrill and John Kirklin considered the risk to the donor unnecessary and were determined to succeed in creating a simple and safe mechanical substitute for the heart and lungs.
5.6.4 The First Reliable Success with the Pump Oxygenator
John W. Kirklin (1917–2004) was hard at work at the Mayo Clinic in Rochester, Minnesota, just hours from Lillehei’s bustling laboratory. Kirklin began assembling a team for advanced cardiac surgery in 1952 and modifying Gibbon’s blueprints in 1953 soon after Gibbon himself had thrown in the towel [51]. The physiologists, cardiologists, and engineers from IBM, along with other skilled people on his team, developed the Mayo-Gibbon pump oxygenator, which featured complex safety mechanisms and a screen oxygenator [52]. They tested their oxygenator in the canine model, of which 9 out of 10 survived 10–60 min of bypass. By this time, the success of Lillehei was well known, and many were questioning the need for a mechanical pump oxygenator. Nevertheless, on March 22, 1955, Kirklin successfully closed a ventricular septal defect in a 5.5-year-old child with direct suture and reported on his success in four of eight patients only 2 months later [53]. Kirklin and his team reportedly went on to perform cardiopulmonary bypass in a series of 245 patients by 1958 [39], as Lillehei continued to perform cardiac surgery using controlled cross circulation. Kirklin said of his relationship with Lillehei, “…our careers were parallel but intertwined, and it is probably fair to say that although we were 90 miles apart, over about a 12-year period, we constantly scouted each other’s programs as intently as does anyone today in the national Football League” [51]. Their friendly rivalry kicked up a notch when in July of 1955 Lillehei switched from human oxygenators to the wonderfully simple and efficient DeWall–Lillehei disposable bubble oxygenator [54], which would take over the market for a number of decades before the disposable membrane oxygenators commonly used today were introduced in the early 1980s [55]. But for a while, it was just Kirklin and Lillehei performing open-heart surgery only 90 miles apart.
One can only imagine the thoughts of the cardiac surgeon during this time period. Suffice it to say, imaginations ran wild and hands itched to begin treatment of cardiac defects which had previously been impossible to approach. The cardiac surgery specialty had certainly existed before this time, but it was like a fish out of water. It struggled for breath and progressed towards the water’s edge in desperate leaps and spurts of hopelessness. The development of a reliable pump oxygenator was the push cardiac surgeons needed to finally delve into a vast ocean of surgical possibilities. Naturally inspired and invigorated, the surgeon sought to tackle the most difficult challenge imaginable and attempted to create artificially a versions of the heart and valves.
5.7 Attempts to Repair Insufficiency (1956–1965): Before Carpentier
The development of the valvular prosthesis became an obsession in the field, and to many, the mechanical valve represented the perfect solution to all valvular lesions. Although there were many attempts to repair insufficient valves before replacement was possible, they all proved to be palliative including techniques to occlude the insufficient orifice with foreign material or indirectly/directly altering the size of the valve annulus. As a result, from the advent of the pump oxygenator in the mid-1950s into the 1970s, the main focus of valvular literature was on mechanical and biological prosthesis. However, there were those who doubted that any mechanical or preserved entity could mimic the elegant movements of the innate valve. As the long-term complications of valve replacement began to unfold, surgeons such as Carpentier were hard-pressed for a reparative procedure. Carpentier drew ideas and techniques from many early contributors for the treatment of insufficiency and finally created a set of techniques which enabled the treatment of regurgitation due to nearly any kind of pathology.
5.7.1 Earliest Attempts: Before the Open Field
The primary strategy in the beginning was focused on the creation of biological flaps or slings to be placed below the mitral valve or within the aorta, which in theory would occlude the insufficient areas and could be easily implanted using a closed operation [56]. These slings were explored in the experimental laboratory independently by reputable surgeons such as Murray (1938), Templeton and Gibbon (1949), and used by Bailey (1951) and Logan (1952) in the clinical setting [2]. Despite the efforts of these surgeons and others, all attempts failed in the long term due to shrinkage/calcification of the tissue or occlusion of the valve orifice. A breakthrough was made in the treatment of insufficiency in 1952 by Charles Hufnagel (1916–1989) of Georgetown University in Washington, D.C., with the first successful ball valve heterotopically placed in the descending aorta [57]. However, this procedure was purely palliative, required great dexterity, and did not correct the original lesion.
Researchers soon realized that many cases of insufficiency could only be corrected if the annulus fibrosus could be prevented from dilating further, which would inevitably occur if any strain on the heart remained. The idea for an indirect procedure to address dilation of the valvular orifice was first introduced by Bernheim in 1909 while attempting to produce a model for stenosis of the mitral valve in dogs using constricting ligatures or circumferential sutures [58]. Clinical attempts by Glover and Davila (1955) and McCallister (1954) failed in the long term as these ligatures often cut through the heart (Fig. 5.5a) [59, 60]. Attempts to place a constricting ligature around the aortic valve by Bailey in 1955 and Taylor in 1958 also failed, as these often slipped out of place [61, 62]. Despite the failure associated with constricting ligatures, these researchers contributed significantly to the literature by recognizing the problem of progressive annular dilation secondary to insufficiency.
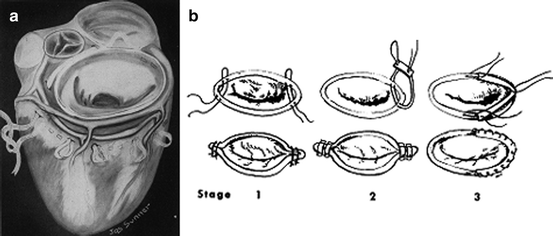

Fig. 5.5
(a) Glover’s circumferential purse string suture and (b) Kay’s review of annuloplasty evolution up to 1960
The next step towards reliable annuloplasty was to directly alter the annulus fibrosus itself. Earle B. Kay (1911–2000) was one of the first to clinically alter the valve annulus of a 26-year-old woman on May 28, 1954, using sutures placed to reduce the annulus at the posteromedial commissure, under guidance of a palpating finger within the valve [63]. Nichols performed a similar technique, in which a portion of the annulus was pinched shut using sutures in a closed technique [64]. This reduced the size of the annulus without altering the leaflets, and it was thought that the annulus was strong enough to prevent the sutures from tearing out. These procedures showed promise, but continued to be inconsistent in results and difficult to complete blind. “Subsequent clinical application of the previously described technical principle for reducing the size of the dilated mitral annulus at the posteromedial commissure has revealed that it is difficult or impossible in some cases…” [63]. Some reliable direct curative technique in the open field was deemed necessary for acquired, congenital, and iatrogenic regurgitation.
5.7.2 First Successful Repair of Mitral Insufficiency: Open Heart
Fortunately, C.W. Lillehei devoted his brilliant mind towards addressing regurgitation of the mitral valve. In an issue of the Lancet in 1957, Lillehei et al. described their extraordinary initial success for treating mitral insufficiency in the open field in four patients with (1) incompetence along the posterior commissure of the valve and/or (2) an enlarged mitral annulus [65]. On August 29, 1956, the initial version of open annuloplasty was performed at the University of Minnesota on a 15-year-old boy, using heavy silk sutures to close the insufficient portion of the patient’s valve. These sutures were anchored in the annulus and tied over pillows of polyvinyl sponge to add support to the leaflet and prevent the sutures from cutting through. All four patients were gravely ill, and, despite the creation of a slight stenosis, all four were remarkably improved. Continued improvement was seen in those four patients with an enlarged mitral annulus over the next 14 months [66]. Meredino et al. independently made a similar contribution, with correction of insufficiency via annuloplasty in the open field in October of 1956 [67].
Kay followed Lillehei into the open field in 1958 and would continue to expand upon and pioneer techniques to correct insufficiency of various origins [68]. In one review by Kay and H.A. Zimmerman, the technique for correction of mitral insufficiency in 82 patients under direct vision was summarized up to 1960. Modifications to their technique were necessary during this time period, as sutures continued to tear out in some patients in the long term (Fig. 5.5b). Kay eventually implemented a multipoint fixation technique, likely inspired by Hufnagel’s fixation method for his aortic prosthesis. Kay fashioned a semicircle of Teflon felt to the mitral annulus with a running suture; the sutures were placed farther apart in the annulus than in the felt to reduce the size of the annulus without undue tension on the sutures in any one segment of the valve. Use of the Teflon felt in this way was likely a precursor to the annuloplasty ring that would later be produced by Carpentier and is still in use today. George Reed would also contribute further to this general technique with his asymmetric annuloplasty (asymmetric with respect to the commissures), which reduced the orifice to a precise calculated size [69]; this kept the anterior leaflet full and mobile while ensuring a large enough annulus.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


