Estimating the incidence, prevalence, and mortality—and hence, prognosis—of HF-PSF has been complicated by heterogeneous diagnostic criteria used in previous epidemiologic studies. Whereas early published reports found a wide range of prevalence (13% to 74%) and annual mortality (1.3% to 17.5%), (
2) more recent studies found a prevalence ranging from one third to almost one half of community-based and hospitalized populations. (
3,
4,
5). Exacerbations of heart failure due to HF-PSF carry the same high hospital readmission rate as do those of heart failure due to systolic dysfunction (
6), and it has been estimated that HF-PSF is responsible for roughly 25% of the total health care cost of treating heart failure in the United States (
7). The prevalence of HF-PSF appears to have increased over the past 15 years, although survival has not improved, in contrast with trends in survival with systolic heart failure (
5). Although mortality with HF-PSF currently appears to be very similar to or slightly better than that with systolic heart failure, survival for both entities remains poor; one study observed 5-year mortality rates of 65% and 68% after heart-failure hospitalization for HF-PSF and systolic heart failure, respectively (
5). Compared with individuals with systolic HF, a greater proportion of female gender, obesity, hypertension, and atrial fibrillation is found among those with HF-PSF, and patients with HF-PSF also tend to be older (
4,
5).
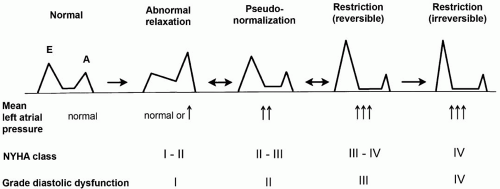
Many processes can cause the diastolicfilling abnormalities that lead to HF-PSF, although the final common pathway in pure diastolic heart failure involves impaired relaxation, decreased passive elasticity, or both. It is important to exclude other
conditions that may also present with heart failure and normal ejection fraction (
Table 15.2). In primary diastolic heart failure, as a result of these myocardial compliance abnormalities, the ventricle is stiffened and requires higher pressures for filling. Consequently, the heart is particularly sensitive to alterations in factors that affect ventricular filling, including volume status, preload, afterload, and diastolic filling time. Perturbations of any of these factors can precipitate acute pulmonary edema and decompensated heart failure. Although the ejection fraction is normal, patients with HF-PSF demonstrate effort intolerance because of impaired cardiac output at rest and diminished capacity to augment cardiac output with exercise (
8).