To evaluate myocardial infarction and describe the early to mid-term changes induced by percutaneous ventricular septal ablation (PVSA) in symptomatic patients with hypertrophic cardiomyopathy using cardiac magnetic resonance imaging. Cardiac magnetic resonance imaging was performed before and 1 week and 1 year after PVSA in 52 patients. The relation between the infarction size and other factors was determined. At 1 week after PVSA, regional hyperenhancement was visualized in the basal interventricular septum in all patients. The mean infarction size was 29.5 ± 15.9 g. The infarction size correlated well with the ethanol volume. The left ventricular myocardial mass decreased significantly from 196.1 ± 65.9 g at baseline to 183.4 ± 63.6 g 1 week after PVSA (p <0.001) and 164.1 ± 60.9 g within the 1-year follow-up period (p <0.001). In conclusion, cardiac magnetic resonance imaging allowed a detailed evaluation of the size and location of septal myocardial infarction induced by PVSA. The left ventricular myocardial mass decreased significantly during the follow-up period.
Cardiac magnetic resonance imaging (CMRI) has a high resolution. It has been considered the reference standard for in vivo determination of the left ventricular (LV) mass and volume and enables precise quantification of the LV wall thickness and dimensions. Also, CMRI with delayed contrast enhancement can be used to detect foci of collagen deposition in the myocardium of patients with hypertrophic cardiomyopathy (HC). Contrast enhancement can provide important feedback by accurately delineating the ultimate size and location of the infarction. Although contrast enhancement can be used to visualize percutaneous ventricular septal ablation (PVSA)-related infarction, its clinical value still needs to be established. The purpose of the present study was to evaluate the myocardial infarction size and describe the early to mid-term changes induced by PVSA in symptomatic patients with HC using CMRI.
Methods
The study population included 57 patients with symptomatic HC despite optimal medical treatment. The study period was January 2005 to September 2008. All subjects demonstrated a dynamic LV outflow tract (LVOT) pressure gradient of ≥50 mm Hg at rest or 100 mm Hg on provocation. Patients with symptoms limiting daily activities (New York Heart Association or Canadian Cardiovascular Society functional class II or exercise-induced syncope), together with a substantial degree of outflow obstruction, were considered candidates for intervention. Before PVSA, a diagnostic left heart catheterization and coronary angiogram were performed.
All patients with HC who were scheduled for PVSA were eligible for CMRI. The indication for PVSA was determined by a significant LVOT gradient as documented by echocardiography and New York Heart Association functional class II to IV despite medical treatment. The exclusion criteria included any absolute or relative contraindication to CMRI (e.g., pacemaker and claustrophobia), atrial fibrillation, or failure to give informed consent. A total of 57 patients were initially enrolled. However, 5 patients were excluded from the final analysis: 1 required pacemaker implantation after PVSA and 4 failed to return for the follow-up examination. The baseline characteristics of the remaining 52 patients are listed in Table 1 .
| Characteristic | Patients (n = 52) |
|---|---|
| Age (years) | 46.5 ± 11.1 |
| Gender | |
| Men | 40 |
| Women | 12 |
| Family history | |
| Hypertrophic cardiomyopathy | 17 |
| Sudden death | 2 |
| Syncope | 25 |
| Dyspnea | 31 |
| Angina pectoris | 25 |
| β Blocker | 33 |
| Verapamil | 15 |
| Diltazem | 9 |
| Amiodarone | 2 |
We used a S5-1 transducer with an iE33 echocardiographic scanner (Philips Medical Systems, Bothell, Washington) to image our subjects. The echocardiographic protocol was composed of obtaining apical 4-chamber, 2-chamber, and long-axis views in all patients. Transthoracic echocardiography was routinely performed to evaluate the site of hypertrophy, mitral valve anatomy, and LVOT gradient. The follow-up measurements included the septal thickness at the site of treatment, LVOT gradient, mitral regurgitation grade, and severity of systolic anterior motion. The peak LVOT gradient was calculated from the color-guided, continuous-wave Doppler velocity using the modified Bernoulli equation.
All patients underwent PVSA at Fuwai Hospital, using a standard Judkins technique. All patients received a 6F transfemoral pacemaker lead. A 5F pigtail or multipurpose catheter was positioned into the left ventricle, and a 6F Judkins guiding catheter was positioned in the ascending aorta. The LVOT pressure gradient was continuously monitored throughout the whole procedure. After initial angiography to localize the origin of the septal perforating arteries, a 1.5 to 2.5 × 9-mm over-the-wire balloon catheter was introduced over a 0.014-in. guidewire into the septal branch perforator artery and inflated. Contrast (Sonovue, BraccoImaging BV, Geneva, Switzerland) was then injected through the balloon catheter shaft during simultaneous registration of transthoracic 2-dimensional echocardiography to determine the part of the myocardium supplied by the targeted septal artery. If balloon placement was correct, 1 to 4 ml of ethanol was slowly (1 ml/min) injected. At 10 minutes after ethanol injection, the balloon was deflated and coronary angiography repeated. The intervention terminated with an angiogram to visualize occlusion of the target vessel and exclude damage to the left coronary tree. A successful procedure was defined as a reduction in the LVOT pressure gradient of 50% of baseline. If the results were not satisfactory, the whole procedure was repeated in another septal branch. The total volume of ethanol injected during the procedure was documented. All patients were monitored in the intensive or coronary care unit for 24 to 48 hours. The plasma creatine phosphokinase, creatine kinase-MB, and lactate dehydrogenase fraction levels were determined before and every 6 hours after the procedure for a 36-hour period.
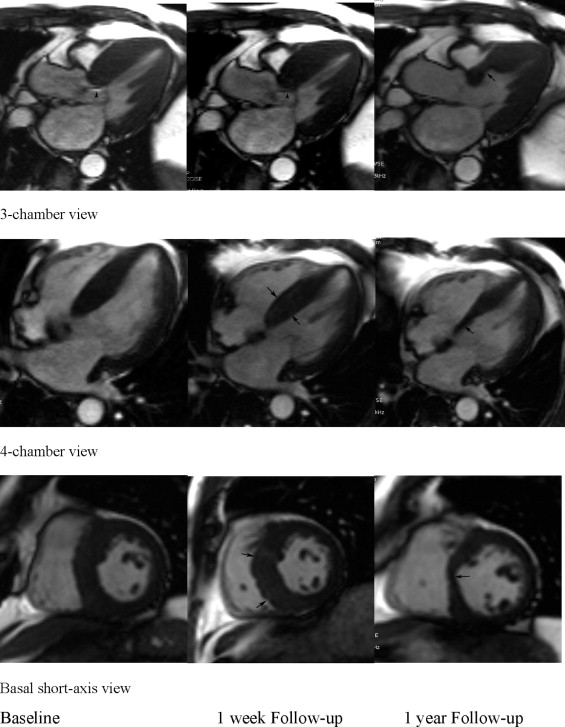
CRMI was performed before ablation and 1 week and 1 year after the procedure in all patients. All scans were performed using a 1.5-T speed clinical scanner (Avanto, Siemens, Erlangen, Germany), using a 4- or 8-element cardiac phased-array receiver coil. The images were acquired using electrocardiographic gating during multiple short breath holds (8 to 15 seconds). Functional performance of the ventricles was assessed using cine steady-state free-precession images. A initial 4-chamber long-axis views (2-, 3-, and 4-chamber view) was obtained, followed by multiple parallel short-axis slices (8-mm slice thickness, 2-mm gap) every 10 mm, covering the entire ventricle from the base to the apex. Contrast-enhanced images were acquired 15 to 20 minutes after intravenous administration of 0.1 mmol/kg gadolinium dimeglumine (Magnevist, Bayer HealthCare, Wayne, Germany) in the same views used in cine CMRI, using a 2-dimensional segmented inversion-recovery prepared gradient-echocardiographic sequence (excitation time 4.4 ms, repetition time 9.8 ms, inversion time 250 to 300 ms, typical voxel size 1.3 × 1.6 × 5 mm 3 ). Contrast-enhanced images were acquired in all patients at follow-up CMRI.
Short-axis cine images were used to calculate the maximum LV wall thickness and LV mass using endocardial and epicardial contours traced at end-diastole. The papillary muscles and prominent trabeculations were included in the mass calculations. The LV ejection fraction was calculated from the endocardial contours traced using computer-assisted planimetry at end-diastole and end-systole.
The myocardial infarction size after PVSA was measured by tracing the hyperenhanced areas using the Argus function software package (Siemens Medical, Erlangen, Germany). The hyperenhanced area was defined as the area within the septal myocardium with pixel signal intensity values ±4 SD of the remote, nonenhanced myocardium. Central dark zones within the area of hyperenhancement were included. The LV ejection fraction and myocardial mass were quantified using the MASS software package (Medis Medical Imaging Systems, Leiden, The Netherlands).
The results are expressed as the mean ± SD. Paired t tests were used to evaluate the changes in LV mass and LV volume after PVSA. Linear regression analysis was used to analyze the relation between myocardial infarction size (outcome variable) and cardiac enzymes, the volume of ethanol administered during the ablation procedure, total LV and septal mass reduction, and LVOT gradient reduction. The statistical analysis was performed using Statistical Package for Social Sciences software, version 11.0 (SPSS, Chicago, Illinois).
Results
The baseline characteristics of our patients are listed in Table 1 . During the ablation procedure, ethanol was injected in 1 septal artery in 40 patients, 2 septal arteries were ablated in 8 patients, and 3 septal arteries in 3 patients. The mean amount of alcohol injected was 2.6 ± 1.3 ml. The creatine phosphokinase peak was 1,545.4 ± 685.3 U/L, the creatine kinase-MB fraction was 177.9 ± 82.5 U/L, and the lactate dehydrogenase peak was 682.3 ± 246.9 U/L. Two patients (3.8%) developed ventricular fibrillation, but they both recovered well after the procedure. Right bundle branch block occurred at a rate of 48.1% (25 patients). Transitory trifascicular blocks occurred at a rate of 42.3% (22 patients). Only 1 patient (1.9%) underwent permanent pacemaker implantation. The peri-interventional mortality during the period of observation was zero. Balloon-induced dissections of the left anterior descending artery did not occur. Also, acute mitral regurgitation did not occur.
The outflow gradient had obviously decreased. The interventricular septal wall thickness measured at the infarct site had also decreased ( Figure 1 ). The anterior, lateral, inferior, and posterior wall thickness at the mid-LV level had not progressively decreased within 1 week or 1 year after PVSA. The LV end-diastolic dimensions and left atrial end-systolic dimensions had also decreased ( Table 2 ).
| Characteristic | Baseline | 1-wk Follow-Up | 1-yr Follow-Up |
|---|---|---|---|
| Left ventricular thickness (mm) | |||
| Interventricular septal wall thickness (mm) | 22.8 ± 4.6 | 21.0 ± 4.2 ⁎ | 17.8 ± 4.3 † |
| Posterior wall | 12.2 ± 3.2 | 11.8 ± 2.6 | 11.7 ± 2.8 |
| Anterior wall | 10.9 ± 2.1 | 10.8 ± 1.9 | 10.7 ± 1.8 |
| Lateral wall | 8.3 ± 1.2 | 8.3 ± 1.0 | 8.2 ± 1.1 |
| Inferior wall | 8.1 ± 1.1 | 8.0 ± 0.9 | 8.0 ± 1.0 |
| Left ventricular end-diastolic diameter (mm) | 43.1 ± 5.7 | 40.9 ± 5.5 ⁎ | 40.5 ± 6.1 ⁎ |
| Left ventricular outflow tract peak systolic gradient (mm Hg) | 96.1 ± 35.9 | 46.5 ± 28.6 † | 29.2 ± 18.9 † |
| Left ventricular ejection fraction (%) | 72.8 ± 7.8 | 69.2 ± 9.5 ⁎ | 68.3 ± 8.8 ⁎ |
| Left ventricular mass (g) | 196.1 ± 65.9 | 183.4 ± 63.6 † | 164.1 ± 60.9 † |
| Left atrial diameter (mm) | 42.1 ± 6.2 | 39.9 ± 7.0 † | 36.2 ± 6.5 † |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


