Fig. 19.1
CT scan (mediastinal window). Hypodense bilateral condensations in the lower lobes (arrows). The low density can be measured (not available measure for this section) or evaluated relative to muscular density (star)
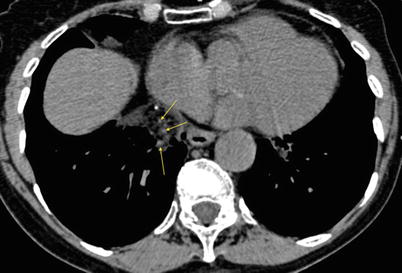
Fig. 19.2
CT scan (mediastinal window), without contrast injection. “Angiogram sign”: several cross-sectioned angiograms within the hypodense consolidation (arrows)
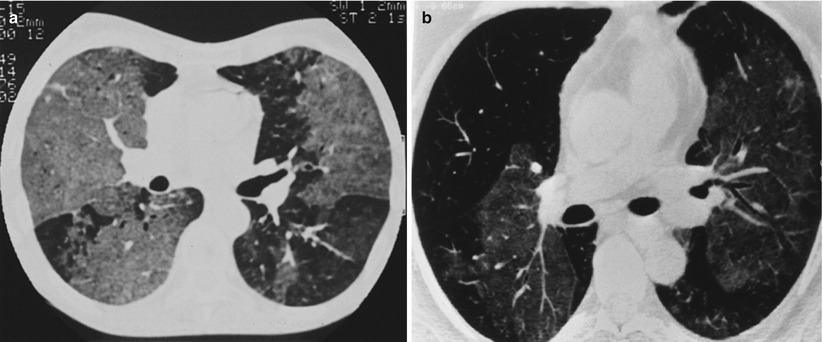
Fig. 19.3
(a, b) High-resolution CT scan (lung window). Bilateral, dense or less dense ground-glass opacities
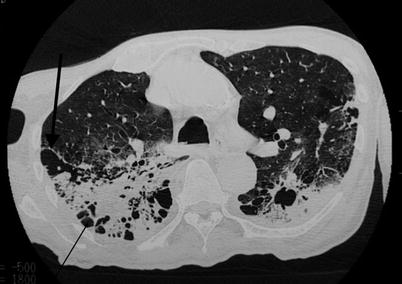
Fig. 19.4
High-resolution CT scan (lung window). Parenchymal condensation of the lower lobes, with signs of fibrosis: kystic images (thin arrow), architectural distorsion with scissural deformation (thick arrow)
One characteristic feature is the “paraffinoma”, which is a mass-like, irregular, spiculated lesion within areas of fat attenuation (Fig. 19.5a, b)
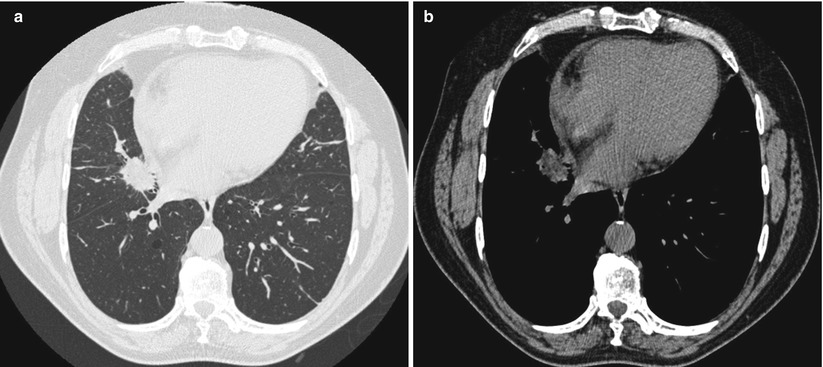

Fig. 19.5
(a) High-resolution CT scan (lung window). Paraffinoma: mass-like, irregular, speculated lesion. (b) CT scan (mediastinal window obtained at the same level). Areas of fat attenuation within the lesion
If the lung injury is declivous, images will have a “sandwich appearance” that some authors [27] consider as characteristic, with a subfissural zone of healthy lung between two pathological zones (Fig. 19.6a, b). The declivity of these lesions can sometimes be highlighted with procubitus sections.
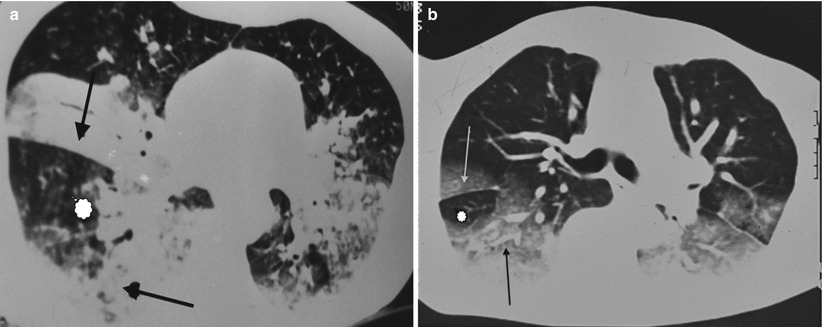

Fig. 19.6
(a, b) High-resolution CT scan (lung window). Declivity of the lesions, responsible for the “sandwich appearance”, with a subfissural zone of healthy lung (star) between two pathological zones (arrows)
Finally, the advent of tomodensitometry and its use in recent series have allowed to show ELP lesions to be far more diffuse than initially described, with frequent bilateral, plurilobar involvement.
Magnetic resonance imaging is seldom used. Findings are a high signal intensity on T1-weighted images and a slow decrease of signal on T2-weighted images [30].
PET scan shows lesions that can increase the uptake of 18F-FDG because of the inflammatory component and thus be misinterpreted as a malignancy [33].
Bronchoalveolar Lavage
The appearance of the fluid retrieved by bronchoalveolar lavage (BAL) suggests a diagnosis of ELP when it shows an oily component on the surface.
Cytological analysis reveals a normal or high mean cellularity with various cytological profiles: macrophage alveolitis, lymphocytic alveolitis or neutrophilic alveolitis; the latter can suggest a fibrosing course in the absence of infection. Alveolitis can also be mixed and no cytological profile is, therefore, more particularly suggestive of ELP [27].
Optical microscopy analysis after standard stains show lipid-laden macrophages that contain optically empty vacuoles with a negative Periodic acid-Schiff and negative Blue Alcian content. The fat stains using (Red Soudan, Black Soudan) allow to color these vacuoles or to show free oily droplets in BAL (Fig. 19.7).
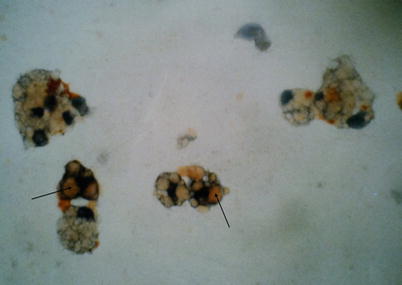

Fig. 19.7
Bronchoalveoloar lavage. Oil red O stain ×40. Intramacrophagic vacuoles (arrows) stained in orange
The fats can be characterized by chemical analysis – either chromatography or infrared spectroscopy – which will allow accurate identification of the oil and prove its exogenous origin. However, these techniques are not commonly used. They are particularly useful if either intoxication by oil or occupational origin cannot be proven.
Histopathology
The presence of oily substances can also be confirmed by histologic analysis, with specimens obtained by transbronchial biopsy, percutaneous fine-needle aspiration biopsy, thoracoscopic lung biopsy, open lung biopsy or operative specimen. The techniques used are the same as for BAL: chronic ELP is characterized by the presence of lipid-laden macrophages that fill and distend the alveoli and interstitium, where they may be associated with accumulation of lipid material, inflammatory cellular infiltration and a variable degree of fibrosis with architectural distorsion (Fig. 19.8).
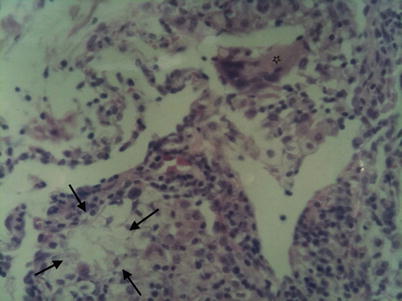
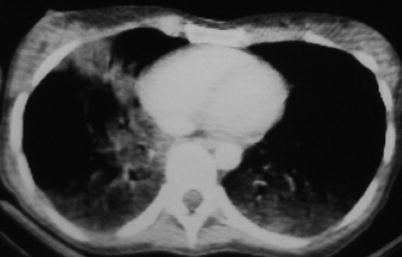

Fig. 19.8
Open lung biopsy. Hematoxylin and Eosin stain ×40. Lipohagic granuloma (arrows). Resorptive giant-cell (star)

Fig. 19.9
CT scan (mediastinal window). Bilateral alveolar condensations with hypodense aspect
Differential Diagnosis
Localized lesions can evoke a common-germ pneumonia or tuberculosis. Paraffinoma, with its radiological characteristics often mimes a malignant tumor.
The diffuse, bilateral forms should lead to investigating all interstitial lung diseases.
Tumors containing fats – hamartomas, lipomas, lipochondromas – must also be eliminated.
Finally, before concluding an exogenous origin, lipoid pneumonia must be examined for any condition that can be responsible for endogenous lipoid pneumonia.
Natural History and Complications
With treatment, acute ELP (fire-eater’s pneumonia, single accidental inhalation) can show clinical and radiological improvement.
The course of the other forms of ELP is chronic and often asymptomatic. As is often the case, they are discovered by chance and can then evolve towards chronic respiratory failure and death if the lesions are extensive and go undiagnosed.
Acute respiratory failure cases have been described, either in acute accidental forms or in the course of chronic forms.
They highlight various complications:
Superinfections are rarely proved and may be diagnosed in excess due to clinical presentation with low-grade fever, cough and sputum. However, superinfections by nontuberculous mycobacteria must be mentioned. First described in 1967 [12], several cases have been reported since then [34]. This condition is probably facilitated by lipids, which enhance the growth of these organisms.
Hemoptysis varies in amount, with a few cases of massive hemoptysis [35], and are either spontaneous or subsequent to bronchial biopsy in contact with the lesion.
Association with bronchial cancer, in which some authors incriminate the possible carcinogenic role of the oils, or “scar cancer”, while others consider this association fortuitous [36, 37].
Hypercalcemia, which can be explained by the production of calcitriol from inflammatory cells, as in other granulomatous diseases such as sarcoïdosis or tuberculosis [38].
ELP in children may be complicated by bronchectasis [39].
Finally, a case of hypertrophic osteoarthropathy has been described in children with chronic ghee oil intoxication [40].
Treatment
The basis of treatment is to recognize the oily substance responsible and to discontinue exposure to it. This will allow to stabilize or improve the lesions or even make them disappear. But most often, the imaging abnormalities last and remain unchanged for years even if the causative exposure has been terminated, contrasting with the usual outcome of conditions due to exogenous exposure (such as hypersensitivity pneumonitis or drug-induced lung diseases). Despite the elimination of intoxication, ELP may also deteriorate if the lesions are extensive at the time of diagnosis.
Predisposing factors such a gastro-oesophageal or abnormalities of upper aerodigestive tracts must also be treated.
Other medical measures should be considered:
Daily respiratory physiotherapy to eliminate the oily substance [41].
Oral corticosteroid therapy to slow down inflammatory reaction, although this treatment has shown mixed results and remains controversial.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


