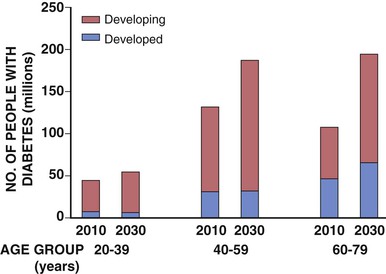
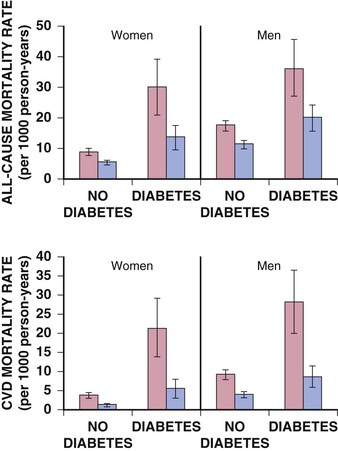
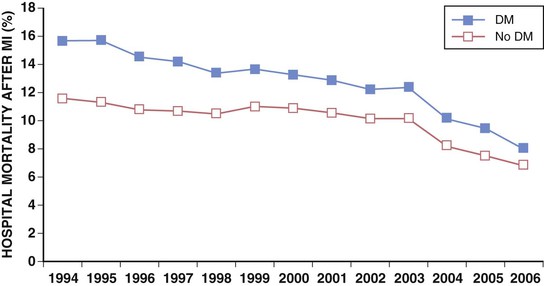
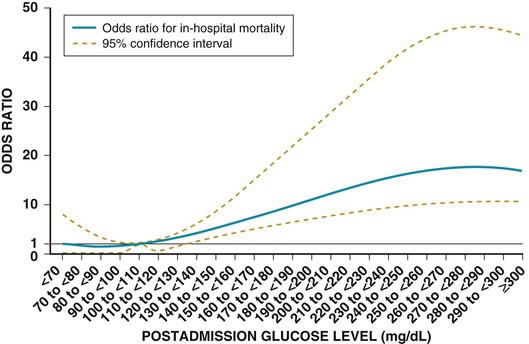
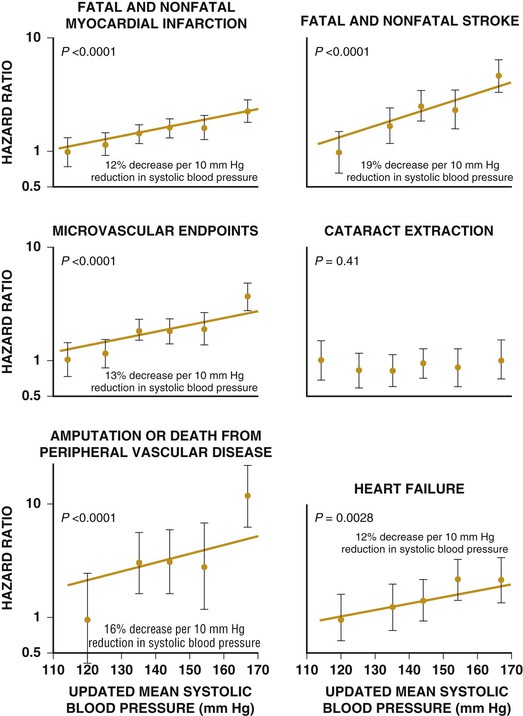
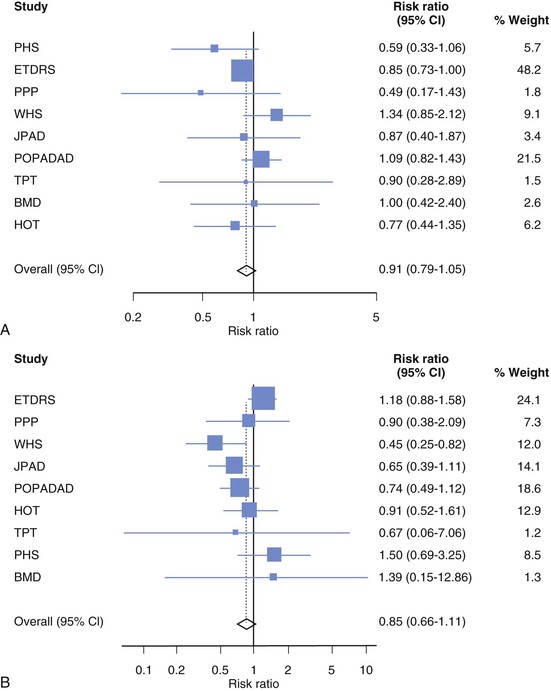
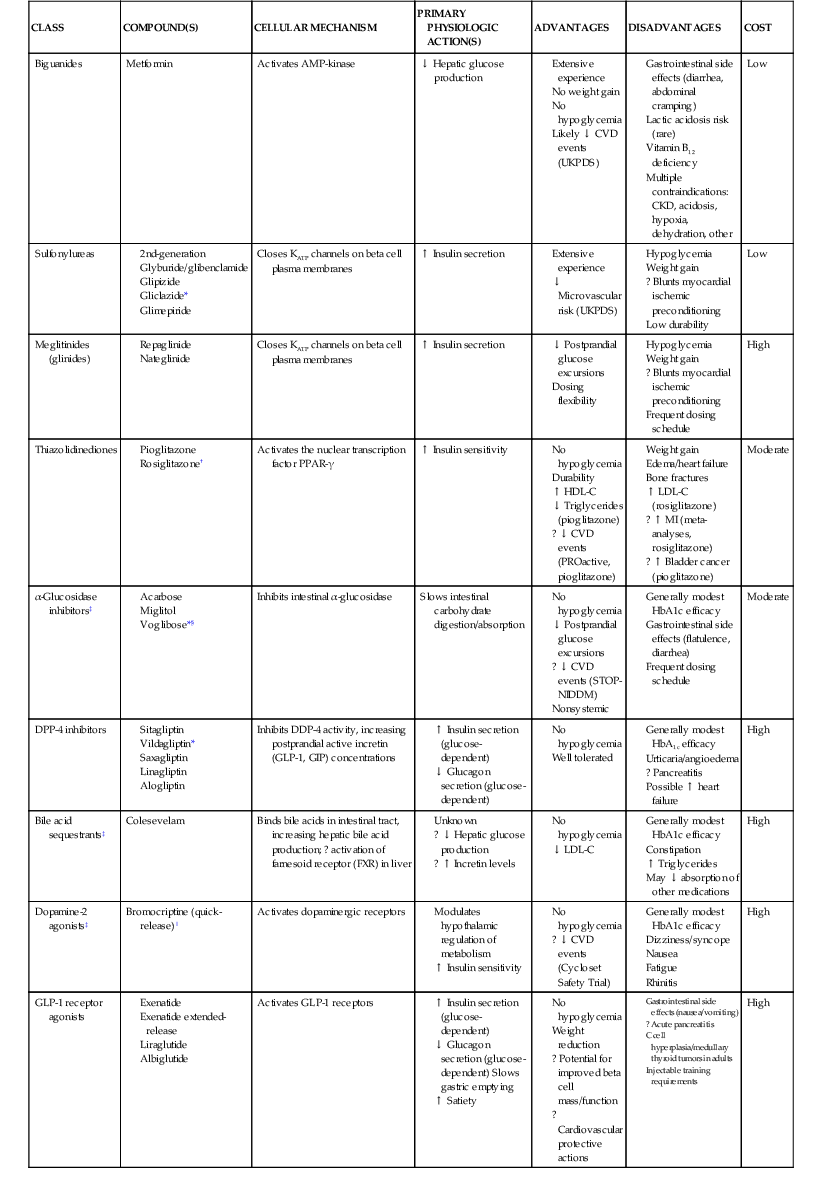
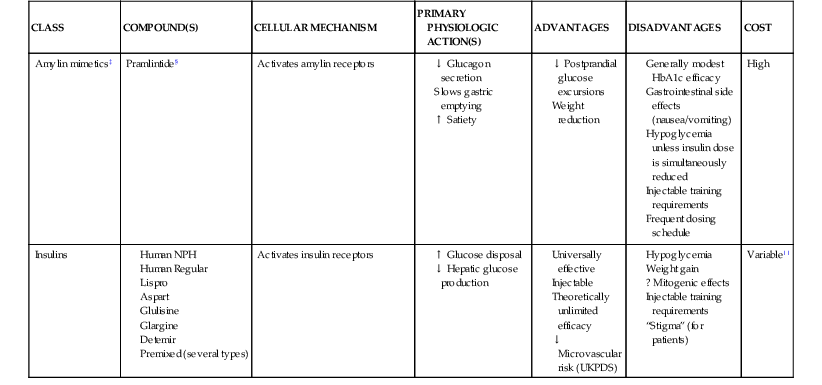
Darren K. McGuire Diabetes mellitus is a group of diseases characterized by insufficient production of insulin or by the failure to respond appropriately to insulin, resulting in hyperglycemia. The diagnostic criteria are summarized in Table 61-1.1 Diabetes typically is classified as either type 2 diabetes, characterized by insulin resistance and relative insulin deficiency, representing greater than 90% of all diabetes cases, or type 1 diabetes, characterized by absolute insulin deficiency. In view of the excess and increasing prevalence of type 2 diabetes and its incremental cardiovascular risk compared with type 1 diabetes, the focus of this chapter is on type 2 diabetes, except when specifically indicated otherwise. Diabetes is among the most common chronic diseases in the world, affecting an estimated 285 million adults in 2010 (6.4% of the global adult population).2 The mounting incidence and prevalence of type 2 diabetes, driven by increasing population age, obesity, and physical inactivity, compound this high global burden (see Chapters 1 and 42), as does the increasing longevity of patients with diabetes. Estimates project that diabetes will affect more than 430 million persons (7.7% of the global adult population) by 20302 (Fig. 61-1). Although much attention historically has focused on the prevention and treatment of microvascular disease complications of diabetes (i.e., retinopathy, nephropathy, and neuropathy), cardiovascular disease (CVD) remains the principal comorbid condition and primary contributor to mortality in the setting of diabetes—most commonly in the form of coronary heart disease (CHD), but also in the incremental risk associated with diabetes for cerebrovascular disease, peripheral vascular disease, and heart failure (HF). For these reasons, continuing efforts toward mitigating the risk of CVD in diabetes remain a global public health imperative. Compared with nondiabetic persons, patients with diabetes have a two- to fourfold increased risk for development of and death from CHD3 (Fig. 61-2). Although older studies have suggested a diabetes-associated CVD risk similar to that for nondiabetic patients with a previous myocardial infarction (MI), more recent observations from clinical trials including patients with diabetes suggest a substantially lower CHD risk, most likely reflecting the effectiveness of contemporary therapeutic interventions.4–6 Diabetes is associated with an increased risk for MI. Across the spectrum of acute coronary syndrome (ACS) events, in which diabetes may affect more than one in three patients,7,8 those with diabetes have worse CVD outcomes after ACS events9 (see Chapters 51 to 53). Despite overall improvements in outcomes during the past several decades for patients with and without diabetes, the gradient of risk associated with diabetes has persisted9 (Fig. 61-3), although the magnitude of incremental in-hospital mortality risk associated with diabetes after an ACS event has declined over the past decade8 (Fig. 61-4). Furthermore, the graded association of increased risk observed with diabetes in the setting of ACS extends to glucose values in the range well below the diabetes threshold, reflected by either glucose values at the time of presentation or those observed throughout hospitalization10 (Fig. 61-5). In addition to CHD, diabetes increases the risks of stroke and of cerebrovascular and peripheral arterial disease. The diagnosis of diabetes portends a twofold increased stroke risk compared with nondiabetic persons (see Chapter 59). Hyperglycemia affects approximately one in three patients with acute stroke, and is associated with a two- to sixfold increased risk for adverse clinical outcomes after stroke.11 Among patients with symptomatic peripheral arterial disease, diabetes prevalence ranges from 20% to 30% and accounts for approximately 50% of all lower-extremity amputations (see Chapter 58).12 In the ambulatory setting, diabetes is independently associated with a two- to fivefold increased risk of HF over that in persons without diabetes, comprising both systolic and diastolic HF, and patients with diabetes have worse outcomes once HF has developed. In addition, diabetes associates with an increased HF risk in the setting of ACS events. The multiple factors that increase HF risk in diabetes include ischemic, metabolic, and functional myocardial perturbations.13 Traditional CHD risk factors such as hypertension, dyslipidemia, and adiposity cluster in patients with diabetes (see Chapters 42 to 45)—but this clustering does not completely account for the increased risk observed among patients with diabetes, with numerous other implicated mechanisms14 (Table 61-2). TABLE 61-2 Examples of Mechanisms Implicated in Diabetic Vascular Disease AGEs = advanced glycation end products; ICAMs = intracellular adhesion molecules; IL = interleukin; MCP = monocyte chemoattractant protein; NF = nuclear factor; RAGE = receptor for advanced glycation end products; VCAMs = vascular cell adhesion molecules. Modified from Orasanu G, Plutzky J: The pathologic continuum of diabetic vascular disease. J Am Coll Cardiol 53:S35, 2009. The mechanisms by which hyperglycemia increases CVD risk remain poorly understood, but given the clear associations between the severity of hyperglycemia and CVD risk in both type 1 and type 2 diabetes, it probably directly influences atherosclerosis development, progression, and instability.14 The principal vascular perturbations linked to hyperglycemia include endothelial vasomotor dysfunction, vascular effects of advanced glycation end products, adverse effects of circulating free fatty acids, and increased systemic inflammation. In addition, the pernicious effects of hypoglycemia complicating diabetes therapy, the sympathovagal imbalance due to diabetic autonomic neuropathy, and the vascular effects of constitutive exposure to excess insulin may further contribute to atherosclerotic risk. Endothelial vasomotor dysfunction, a hallmark of diabetic vascular disease, is associated with increased hypertension and adverse CVD outcomes. The myriad mechanisms contributing to endothelial dysfunction include abnormal nitric oxide biology, increased endothelin and angiotensin II, and reduced prostacyclin (i.e., prostaglandin I2) activity, all of which contribute to abnormal control of blood flow. In the setting of ACS events, the no-reflow phenomenon after percutaneous intervention reflecting acute endothelial dysfunction occurs more commonly in the presence of hyperglycemia independent of diabetes status and may contribute to increased myocardial jeopardy, resulting in larger infarcts, increased arrhythmia, and worse systolic function. Abnormalities in lipid metabolism also contribute to the increased atherosclerotic risk associated with diabetes (see Chapters 42 and 45). High triglyceride levels, low high-density lipoprotein (HDL) concentration, and increased atherogenic small dense low-density lipoprotein (LDL) particles characterize diabetic dyslipidemia, and each may contribute to the accelerated development and progression of atherosclerosis. Perturbations in the coagulation and fibrinolytic pathways and in platelet biology add to the vascular risk of diabetes, yielding a constitutive prothrombotic milieu.15 These abnormalities include increased circulating tissue factor, factor VII, von Willebrand factor, and plasminogen activating inhibitor 1, with decreased levels of antithrombin III and protein C. In addition, disturbances of platelet activation, aggregation, morphology, and lifespan further contribute to increased thrombotic potential, as well as to the acceleration of atherosclerosis. Increased systemic inflammation portends an increased risk for diabetes and diabetic atherosclerotic disease, and diabetes is associated with increased oxidative stress and the accumulation of advanced glycation end products. For example, diabetes is associated with lipid-rich atherosclerotic plaque and increased inflammatory cell content, increased expression of tissue factor, and increased expression of the receptor for advanced glycation end products, yielding plaques with characteristics of higher risk in both coronary and carotid arteries.14 Therapeutic lifestyle interventions remain the cornerstone of prevention of the atherosclerotic complications associated with diabetes. As recommended by the American Diabetes Association (ADA) and the American Heart Association (AHA), overarching therapeutic lifestyle targets include smoking abstinence, at least 150 minutes of moderate-intensity aerobic activity weekly, and nutrition recommendations for weight control and dietary composition.16,17 Although lifestyle modification may not affect cardiovascular outcomes when instituted in patients with established diabetes, such intervention reduces risk factors for vascular disease and yields many health benefits.18,19 Beyond lifestyle, pharmacologic strategies effectively reduce CVD risk in diabetes.16,17,20 Such interventions include assiduous blood pressure and lipid management for all patients, and for patients at highest risk, angiotensin-converting enzyme (ACE) inhibitors independent of blood pressure and also daily aspirin.16,17 In the context of these evidence-based CVD interventions, the accumulated data regarding the effects of glucose control on CVD risk mitigation remain less robust.16,21,22 Type 2 diabetes is associated with a characteristic pattern of dyslipidemia, reviewed in detail in Chapter 45. Each component of the diabetic dyslipidemia profile is independently associated with CVD risk, including increased small, dense LDL particles, increased apolipoprotein B concentration, increased triglycerides, and decreased HDL cholesterol. Despite extensive research in modifying triglyceride and HDL cholesterol levels with a variety of pharmacologic agents, however, the net influence on CVD risk of these strategies remains uncertain, and statin treatment remains the cornerstone of therapeutic lipid intervention in patients with diabetes (see also Chapter 42). Contemporary guidelines for the management of diabetic dyslipidemia focus on the use of statin medications,16,17,23 with meta-analyses of randomized clinical trials enrolling large numbers of patients with diabetes yielding estimates of numbers needed to treat to prevent one major adverse CVD complication over 5 years in the setting of diabetes: 39 for primary prevention and 19 among patients with prevalent CVD.24 Recommendations from many contemporary professional societies do not require elevation of LDL cholesterol as a requisite for the initiation of a statin, advising such therapy in all patients with diabetes older than 40 years of age with one or more CVD factors, or younger in the setting of prevalent CVD, endorsing a target of LDL less than 100 mg/dL or 35% to 40% reduction from baseline.16 An optional, more intensive target has been endorsed for patients with diabetes of LDL cholesterol below 70 mg/dL and non-HDL cholesterol less than 100 mg/dL.17 The 2013 AHA/ACC Lipid Guideline advises statin treatment for people with diabetes between the ages of 40 and 75 years with LDL cholesterol levels between 70 and 189 mg/dL even if they have not had a prior cardiovascular event. This guideline does not recommend LDL target levels (see the Guideline section following Chapter 42).23 The 2013 guideline suggests that in statin-intolerant patients a secondary option is to add another lipid-modifying agent such as ezetimibe, bile acid binders, fibric acid derivatives, fish oil, or niacin. The net CVD efficacy and overall safety associated with such add-on therapy, however, remain unsubstantiated. The most recent guidelines from the AHA and the American College of Cardiology (ACC) endorse the prescription of statin drug and intensity predicated on estimated 10-year risk for atherosclerotic vascular disease,23 independent of LDL cholesterol concentrations or specified LDL cholesterol or non-HDL cholesterol therapeutic targets. For patients with type 2 diabetes, at least moderate-intensity statin therapy is endorsed for all patients with diabetes exceeding 7.5% estimated 10-year risk for atherosclerotic cardiovascular complications. These guidelines also discourage add-on lipid-modifying therapies, reserving their use for patients intolerant of statins. Intensive-dose versus moderate-dose statin therapy reduces cardiovascular risk further,24 but a large meta-analysis and analyses of data from several randomized clinical trials have identified an increased risk of new-onset diabetes associated with intensive-dose statin therapies compared with placebo or standard care (reviewed in detail in Chapter 42).25 These observations suggest a deleterious effect of intensive-dose statin therapy on glycemia, due to unknown mechanisms. Whether intensive-dose statin therapy adversely affects glucose control among patients with prevalent diabetes remains uncertain. In view of the potent favorable influence of statin therapy at reducing CVD risk, however, the observed incremental risk of incident diabetes should not discourage aggressive use of intensive-dose statin therapy in eligible patients with diabetes. Inasmuch as omega-3 fatty acids (see Chapters 45 and 46) can reduce circulating triglycerides by up to 40%, such preparations—predominantly fish oil preparations in clinical use—hold particular promise in the treatment of diabetic dyslipidemia. In addition, in the absence of reported interactions with statins, fish oil is particularly attractive as an add-on therapy to statins if incremental triglyceride reduction is desired once LDL cholesterol targets have been achieved with statin therapy. A series of randomized clinical trials have demonstrated beneficial effects on CVD outcomes with fish oil, such as data from a subanalysis of 4565 patients with impaired fasting glucose or diabetes participating in the Japan EPA Lipid Intervention Study (JELIS), a randomized trial comparing treatment with 1800 mg of eicosapentaenoic acid (EPA) plus simvastatin daily versus simvastatin alone.26 In this subset, EPA treatment conferred a 22% risk reduction (P = 0.048) for major adverse CVD events compared with simvastatin alone. But subsequent results from the Outcome Reduction with an Initial Glargine Intervention (ORIGIN) randomized trial have challenged these findings. In ORIGIN, 12,536 patients with impaired fasting glucose, impaired glucose tolerance, or diabetes were randomly assigned to receive either a 1-g capsule containing at least 900 mg (90% or more) of ethyl esters of n-3 fatty acids or a capsule containing 1 g of olive oil daily.27 The primary outcome was cardiovascular mortality. Over a median follow-up period of 6.2 years, no significant reduction in the primary outcome occurred with n-3 fatty acid treatment versus control (9.1% versus 9.3%, respectively; P = 0.72), with a total of 1155 total cardiovascular death events to analyze—yielding substantial statistical power. Of note, in contrast with the JELIS trial, the total dose of n-3 fatty acids used in the ORIGIN trial was significantly lower (1800 mg versus 900 mg, respectively); the fish oil formulation in ORIGIN contained a mixture of docosahexaenoic acid (DHA) (approximately 45%) and EPA, whereas the JELIS trial used a formulation of only EPA, and the comparator in ORIGIN was not a true placebo but a capsule containing 1 g of olive oil taken daily. The degree to which these contrasts contributed to the different observed results remains unclear. As indicated by the accumulated data, fish oil remains a reasonable adjunct to statin therapy in patients with diabetes or who have extremely high levels of triglycerides (>500 mg/dL), to mitigate hyperviscosity complications. Fibrates are agonists of the nuclear transcriptional regulator peroxisome proliferator–activated receptor alpha (PPAR-α) that lower triglycerides and modestly increase HDL cholesterol. Although they favorably affect two of the fundamental abnormalities of diabetic dyslipidemia, the net CVD effects of this class of drugs remain uncertain. Randomized trials of gemfibrozil versus placebo have demonstrated efficacy in subjects with diabetes, but these trials were executed before the use of statins became prevalent in the populations studied, and an increased risk of myopathy limits the use of gemfibrozil with statins. Subsequently, the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) trial evaluated the CVD effect of fenofibrate versus placebo in a population of 9795 patients with type 2 diabetes and showed no statistically significant reduction in the primary endpoint of coronary death or nonfatal MI, despite accumulating 544 primary outcome events for evaluation (5.2% versus 5.9%; hazard ratio [HR], 0.89 [95% confidence interval {CI}, 0.75 to 1.05]).28 The prevalent drop-in of statin use in the placebo arm may have contributed to this negative result, but in the present era, when statins are indicated for all patients eligible for the FIELD trial, the trial results have limited generalizability and challenge the utility of fenofibrate add-on therapy for CVD risk mitigation. In the more recently completed ACCORD (Action to Control Cardiovascular Risk in Diabetes) lipid trial that included 5518 patients with type 2 diabetes at high cardiovascular risk, fenofibrate compared with placebo, each added to simvastatin background therapy, likewise failed to yield significant improvements in major adverse cardiovascular outcomes, despite the accumulation of 601 primary endpoint events of cardiovascular death, MI, and stroke.29 Fibrates remain an option for patients with intolerance to statin medications, for isolated hypertriglyceridemia in diabetic patients at otherwise low CVD risk, and as add-on therapy to maximally tolerated statin monotherapy when patients do not achieve therapeutic targets (with the recognition of some increased myopathy risk). Gemfibrozil should never be added to statin therapy because of an unacceptable risk of muscle complications. Although individual trials have failed to demonstrate conclusively a benefit of fibrates on CVD risk, a meta-analysis of the published data suggests modest but significant improvements in composite and component cardiovascular outcomes, with or without background statin therapy.30 In addition, subanalyses of the reported trials suggest that patients with diabetes who have baseline high triglycerides concomitant with low HDL cholesterol may benefit from incremental CVD risk reduction with fibrates added to background therapy—a hypothesis pending confirmation in a dedicated randomized trial. Niacin is a potent modulator of lipid metabolism (although the mechanism of action remains poorly understood) and has the greatest effect among currently available drugs on increasing HDL-cholesterol while also lowering triglycerides. But the net CVD effects and safety of niacin, especially in the context of background statin therapy, remain undetermined. As with fibrates, the accumulated data set regarding the net CVD effects of niacin remains limited primarily by relatively small studies with substantial dropout rates; a recent meta-analysis estimated a 27% relative risk reduction associated with niacin in the absence of statin background therapy. Two large-scale randomized trials recently evaluated the incremental CVD efficacy and safety of niacin added to statin therapy by comparing niacin formulations versus placebo, each added to background simvastatin therapy. In the National Institutes of Health (NIH)-sponsored Atherothrombosis Intervention in Metabolic Syndrome with Low HDL/High Triglycerides: Impact on Global Health Outcomes (AIM-HIGH) trial, 3414 patients with established atherosclerotic cardiovascular disease were randomly assigned to blinded treatment with extended-release niacin, 1500 to 2000 mg/day, or matching placebo, with a primary trial outcome of the first event of the composite of death from CHD, nonfatal MI, ischemic stroke, hospitalization for an ACS, or symptom-driven coronary or cerebral revascularization.31 All patients received simvastatin, 40 to 80 mg/day, plus ezetimibe 10 mg/day if needed, to maintain a LDL cholesterol level of 40 to 80 mg/dL. This trial was halted prematurely with a mean follow-up of 3 years due to futility of the intervention on the primary outcome for niacin versus placebo (16.4% versus 16.2%, respectively; P = 0.79), in the setting of a nonsignificant numeric excess for ischemic stroke in the niacin versus placebo arm (29 patients versus 18 patients; P = 0.11). Observations among the subset of 1158 patients enrolled with prevalent diabetes were similar to the overall trial results, with no statistical interaction observed. The Heart Protection Study 2-Treatment of HDL to Reduce the Incidence of Vascular Events (HPS2-THRIVE) trial enrolled 25,673 patients with prevalent atherosclerotic vascular disease randomly assigned to blinded treatment with extended-release (ER) niacin/laropiprant combination tablet or placebo, both added to background simvastatin therapy with or without ezitimibe.32 The primary trial endpoint was time to the first event of a composite of coronary death, nonfatal MI, stroke, or coronary revascularization, with no significant difference observed between the groups randomized to ER niacin/laropiprant versus placebo (14.5% versus 15.0%; P = 0.29), although for the component endpoint of coronary revascularization, a statistically significant 11% reduction occurred with ER niacin/laropiprant versus placebo (4.6% vs. 5.2%; P = 0.04). The comparative efficacy in the subset of patients with prevalent diabetes at study entry (n = 8299 [32%]) conformed to the overall trial results, with an interaction P value of 0.98. Of considerable concern, the risk of adverse events increased significantly with ER niacin/laropiprant, including a statistically significant 3.7% absolute increased risk for hyperglycemic and hypoglycemic events among patients with diabetes, and a 1.8% absolute (27% relative) increased risk for incident diabetes with niacin/laropiprant (9.1% versus 7.3%) (HR, 1.27 [95% CI, 1.14 to 1.41]). Like fibrates, niacin remains an option for patients with intolerance to statins, for isolated hypertriglyceridemia in diabetic patients with an otherwise low CVD risk, and as add-on therapy to maximally tolerated statin monotherapy, although the AIM-HIGH and HPS2-THRIVE trial results challenge the latter application. Current data do not support niacin use as an add-on to statin therapy in patients achieving target LDL cholesterol levels. Hypertension (see Chapter 44) affects approximately 70% of diabetic patients (twice the rate observed in nondiabetic subjects), with a steep graded association between blood pressure and adverse cardiovascular outcomes33 (Fig. 61-6). In this context, numerous classes of antihypertensive medications reduce diabetic CVD risk,34 and given the potent benefits for both macrovascular and microvascular disease complications, blood pressure management has great importance in this high-risk population. Furthermore, blood pressure targets for patients with diabetes are more aggressive than for the overall population, with a goal of less than 130/80 mm Hg for patients with diabetes who can tolerate such aggressive management without undue clinical burden, and a target of below 140/80 for all others.16,17 ACE inhibitors and angiotensin II receptor blockers (ARBs) have become cornerstones of therapy for hypertension in diabetes because of their broadly demonstrated favorable effects on diabetic nephropathy and CVD outcomes, as well as their modest favorable effects on measures of glucose metabolism.16,17,34,35 These drugs are antagonists of the renin-angiotensin-aldosterone system (RAAS). Data from randomized trials of patients with and without hypertension support the recommendation for ACE inhibitors as first-line agents for treatment of hypertension in the setting of diabetes. For example, the Heart Outcomes Prevention Evaluation (HOPE),36 which compared ramipril (10 mg daily) with placebo in patients at increased risk for CVD, ramipril was superior to placebo in the diabetes subset of 3577 HOPE patients for the primary outcome of cardiovascular death, MI, and stroke (25% relative risk reduction [RRR]; P = 0.004), and for overt nephropathy (24% RRR; P = 0.027). Similar observations derive from the diabetes subanalysis of the EUROPA (EURopean trial On reduction of cardiac events with Perindopril in stable coronary Artery disease) trial,37 which tested perindopril versus placebo; the point estimate of treatment effect with perindopril compared with placebo, a 19% relative risk reduction among the 1502 participants with diabetes, resembled the 20% risk reduction observed in the overall trial. On the basis of these results and support from a meta-analysis of data from reported trials,34 ACE inhibitors are recognized as first-line agents for the treatment of hypertension in the setting of diabetes and should be considered for all diabetic patients with prevalent CVD or a clustering of CVD risk factors.16,17 Cardiovascular outcomes data for ARBs are much less robust than for ACE inhibitors, particularly in patients with diabetes. The Telmisartan Randomized AssessmeNt Study in ACE iNtolerant subjects with cardiovascular Disease (TRANSCEND) trial enrolled 5926 patients with intolerance to ACE inhibitors, randomly assigned to receive telmisartan (80 mg daily) or placebo, including 2118 patients with diabetes.38 In the overall trial, telmisartan failed to achieve statistical superiority over placebo in reducing the primary composite of CVD death, MI, stroke, and HF hospitalization (HR, 0.92 [95% CI, 0.81 to 1.05]), but it significantly reduced the secondary composite of cardiovascular death, MI, and stroke (HR, 0.87 [95% CI, 0.76 to 1.00]). Although subgroup analyses lacked statistical power, the point estimates of effect for both the primary and key secondary outcomes were markedly attenuated in the subset of patients with diabetes. Guidelines from the ADA and AHA have endorsed ARBs and ACE inhibitors with similar levels of recommendation,16,17 but with little evidence available regarding the effects of ARBs on CVD outcomes, ACE inhibitors should remain first-line agents, with ARBs reserved for patients who cannot tolerate ACE inhibitors because of cough, angioedema, or rash. Dihydropyridine calcium channel blockers generally are well tolerated and effectively lower blood pressure. Analyses of data for diabetes subsets in randomized clinical trials suggest a magnitude of CVD clinical benefit similar to or greater than that observed in nondiabetic cohorts, including evaluations of nitrendipine, nisoldipine, and amlodipine.34 In active-controlled comparisons, amlodipine has shown superiority to hydrochlorothiazide when added to background benazepril therapy,39 but in randomized trials directly comparing the efficacy of calcium channel blockers versus ACE inhibitors, ACE inhibitors provide superior outcomes. Antagonists of beta-adrenergic receptors (beta blockers) are another key component of effective CVD risk reduction in diabetes. Early in the course of clinical use, beta blockers were judged to be relatively contraindicated in the setting of diabetes because of concerns about masking hypoglycemia symptoms and adverse effects on glucose and lipid metabolism. The results of CVD outcomes trials have allayed these concerns and support the benefit of beta blockers for patients with diabetes in the chronic ambulatory setting40 and in the post-ACS population.41 In addition, the metabolic effects of various beta blockers may differ: With noncardioselective beta blockers that also have alpha receptor–blocking properties, metabolic markers may improve, although the clinical relevance of these differential effects remains unproved. A meta-analysis of randomized clinical trials supports the utility of beta blockers in the treatment of patients with diabetes.34 Concern about the adverse glycemic and triglyceridemic effects of the thiazide diuretic class of medications—including hydrochlorothiazide, chlorthalidone, indapamide, and bendroflumethiazide—has resulted in some degree of hesitancy regarding use of these medications in diabetic patients. Of note, however, randomized trials of thiazide diuretics that included substantial numbers of patients with diabetes have consistently demonstrated CVD benefits. In a subanalysis of the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT), the CVD effects of chlorthalidone compared with both lisinopril and amlodipine were similar in patients with diabetes or impaired fasting glucose, despite modest but statistically significant increases in incident diabetes associated with chlorthalidone use.42 A meta-analysis of randomized trials further supported the benefits of thiazide diuretics in the treatment of patients with diabetes.34 In addition to demonstrating efficacy of individual drugs, studies also have proved the benefits of combination antihypertensive therapy in patients with diabetes. In the Action in Diabetes and Vascular disease: preterAx and diamicroN-MR Controlled Evaluation (ADVANCE) trial, which compared combination therapy with perindopril plus indapamide versus placebo in 11,140 patients with type 2 diabetes,43 the combination therapy was associated with a 9% relative reduction in a composite primary outcome combining microvascular and macrovascular disease endpoints, compared with placebo. In the Anglo-Scandinavian Cardiac Outcomes Trial–Blood Pressure Lowering Arm (ASCOT-BPLA),44 which randomly assigned patients to treatment to amlodipine, with perindopril added as needed, or atenolol with bendroflumethiazide added as needed, the amlodipine-perindopril combination yielded a significant 13% relative risk reduction (P = 0.028) in major CVD outcomes in the 923 patients with diabetes, compared with atenolol-bendroflumethiazide. Finally, in the Avoiding Cardiovascular Events through Combination Therapy in Patients Living with Systolic Hypertension (ACCOMPLISH) trial,39 in which all patients were treated with benazepril, with randomization to add-on amlodipine versus add-on hydrochlorothiazide, treatment with benazepril-amlodipine versus benazepril-hydrochlorothiazide was associated with a 21% reduction in CVD outcomes among the 6946 patients with diabetes (60.4% of the study cohort; P = 0.003). Therefore, in combination with thiazide diuretics and with amlodipine, ACE inhibitors are associated with improved CVD outcomes, with the combination with amlodipine proving superior in head-to-head comparison. In view of the increased CVD risk associated with hypertension in diabetes mellitus and the clearly demonstrated graded association between blood pressure reduction and CVD risk, observational data suggest that patients with diabetes mellitus constitute a special population requiring more aggressive blood pressure control (see also Chapter 44 and the Guidelines section at the end of this chapter). Recent randomized clinical trials generally have supported this concept, including the National Heart, Lung and Blood Institute (NHLBI)-sponsored ACCORD trial, in which 4733 patients with type 2 diabetes at high cardiovascular risk were randomly assigned to treatment to achieve systolic pressure goals of either less than 120 mm Hg or less than 140 mm Hg.45 In a comparison of the more intensive and less intensive arms, the point estimate of a 12% relative risk reduction in the primary composite endpoint of cardiovascular death, MI, and stroke failed to achieve statistical significance (HR, 0.88 [95% CI, 0.73 to 1.06]); more intensive control was associated with a significant 41% reduction in stroke (HR, 0.59 [95% CI, 0.39 to 0.89]). Of note, at the time of randomization, when clinical guidelines endorsed systolic blood pressure targets of less than 130/80 mm Hg, the average blood pressure at study entry was 139/76 mm Hg in this high-risk cohort. During the trial, the average systolic pressure achieved in the below 120 mm Hg arm was 119.3 mm Hg, contrasted with an average of 133.5 mm Hg in the patients randomly assigned to a target of below 140 mm Hg, requiring an average of 3.4 and 2.3 medications, respectively. Therefore, although the trial failed to prove the benefit of more intensive blood pressure control over contemporary targets, the pressures achieved in the less intensive group fell quite close to such targets, and in the context of favorable secondary outcomes with no prohibitive safety signals observed, the current target of below 130/80 mm Hg seems prudent for most patients with diabetes, with no clear imperative to achieve more aggressive control. Bangalore and colleagues analyzed 13 randomized trials with 37,736 participants to evaluate target blood pressure goals for patients with type 2 diabetes, impaired fasting glucose (IPG), or glucose intolerance (IGT).46 Intensive blood pressure control (with targeted values of ≤130 mm Hg systolic) was associated with a 10% reduction in all-cause mortality (odds ratio [OR], 0.90; P < 0.05), and a 17% reduction in stroke, compared with standard blood pressure control levels (≤140 mm Hg). It also was associated, however, with a 20% increase in serious adverse events, including a 17-fold increase in hypotension and a 10-fold increase in hyperkalemia. Intensive blood pressure control did not improve cardiac, renal, or retinal outcomes over those achieved with standard blood pressure control. Meta-regression analysis showed continued risk reduction for stroke with a systolic pressure of less than 120 mm Hg, but at levels below 130 mm Hg, serious adverse events increased by 40%, with no benefit seen for renal, retinal, or cardiac outcomes. The investigators concluded that based on the available body of evidence, a systolic pressure treatment goal of 130 to 135 mm Hg is acceptable. Although the risk of stroke continued to fall with more aggressive goals, the risk of treatment-related adverse events rose substantially, with no benefit regarding the risk of other macrovascular or microvascular events. To explore further the epidemiologic relationships between intensity of blood pressure control achieved and cardiovascular outcomes, Redon and colleagues performed post hoc analyses of the Ongoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial (ONTARGET).47 In the trial, 25,584 patients, including 9603 with diabetes, were randomly assigned to treatment with ramipril, telmisartan, or both and observed for a mean follow-up period of 4.8 years. The primary endpoint, a composite of cardiovascular death, nonfatal MI, nonfatal stroke, and HF hospitalization, occurred in 20.2% of patients with diabetes and 14.2% of patients without diabetes. The patients with diabetes had a statistically significant 48% increased risk for the primary endpoint, as well as statistically significantly increased risks of the component endpoint of cardiovascular death, MI, stroke, and congestive HF hospitalization. The relationship between blood pressure achieved and overall cardiovascular risk showed similar patterns in participants with and without diabetes, although diabetes shifted the relationship of cardiovascular events upward at any level of blood pressure achieved. In patients with and without diabetes, a progressively larger reduction in systolic pressure accompanied a reduction in risk of the primary outcome, but only if baseline pressure ranged from 143 to 155 mm Hg (except from stroke). In terms of fatal and nonfatal outcomes except for stroke events, reducing systolic pressure to less than 130 mm Hg in patients with diabetes had no further benefit. The incidence of stroke events was reduced down to on-treatment blood pressure levels of 115 mm Hg, with no evidence of an upward “J curve” inflection. A J curve relationship was observed with other endpoints. For the primary outcome, the nadir of the J curve was 129 mm Hg systolic blood pressure in both patients with and without diabetes. These observations parallel those from the ACCORD trial, and the most recent ADA guidelines for the management of patients with diabetes now incorporate these data, and endorse a blood pressure target of less than 140/80 mm Hg for all patients with diabetes and hypertension, with a more intensive blood pressure target of less than 130/80 mm Hg for those patients who can achieve it without “undue treatment burden.”16 (See also the Guideline section following Chapter 44.) In summary, five classes of antihypertensive medications reduce CVD risk in patients with diabetes: ACE inhibitors, calcium channel blockers, beta blockers, thiazide diuretics, and ARBs. In addition, evidence supports a blood pressure target of less than 140/80 mm Hg for all patients with diabetes and hypertension, with a more intensive systolic pressure target of less than 130 mm Hg for those patients who can achieve that target without excessive adverse effects. As discussed earlier, patients with diabetes have aberrations of platelet structure, function, and activity, yielding in aggregate a prothrombotic milieu. In addition, absolute or relative aspirin resistance may occur in up to 40% of patients with diabetes, with increasing prevalence associated with poor metabolic control. On the basis of these observations, much interest and investigation have focused on optimizing antiplatelet therapy for this high-risk cohort of patients. At present. the ADA and AHA recommend daily aspirin (75 to 162 mg/day) for all patients with diabetes who have established CVD, or for primary prevention in patients with diabetes older than 50 years of age for men or 60 years of age for women, with additional CVD risk factors (or younger in the presence of prevalent CVD risk).16,20 A substantial evidence base in the setting of secondary CVD risk modification supports these recommendations, but a recent meta-analysis of primary prevention with aspirin in patients with diabetes has not shown statistically significant benefit (Fig. 61-7).20 Two randomized clinical trials currently are under way to explore further the role of aspirin in the setting of primary CVD risk prevention in type 2 diabetes. A Study of Cardiovascular Events In Diabetes (ASCEND) plans to enroll 10,000 patients with type 1 or type 2 diabetes without CVD, randomly assigned to treatment with 100 mg acetylsalicylic acid (ASA) daily or placebo, with a primary endpoint of major adverse cardiovascular events (http://www.ctsu.ox.ac.uk/ascend/). The Aspirin and Simvastatin Combination for Cardiovascular Events Prevention Trial in Diabetes (ACCEPT-D) plans to enroll 4700 patients with type 1 or type 2 diabetes to receive 100 mg ASA plus simvastatin, versus simvastatin alone, in a Prospective, Open-Label, Blinded Endpoint Evaluation (PROBE) design trial to assess the cardiovascular efficacy of ASA in primary prevention for patients with diabetes treated with statins.48 In summary, until further evidence becomes available, daily aspirin (75 to 162 mg) is recommended for all patients with diabetes who have CVD, or with increased CVD risk assessed by age older than 50 years for men and 60 years for women with additional CVD risk factors.16,20 Accumulated data suggest that patients with type 2 diabetes require more aggressive antiplatelet treatment to alter CVD risk. Thienopyridines and nonthienopyridine antagonists of the platelet receptor P2Y12 inhibit adenosine diphosphate (ADP)-induced activation of glycoprotein (GP) IIb/IIIa, a mediator of platelet aggregation, yielding more potent antiplatelet effects than those achieved with aspirin alone. Observations from trials of the thienopyridines clopidogrel and prasugrel, and the nonthienopyridine antagonist of the P2Y12 receptor ticagrelor, support their efficacy in patients with diabetes in the chronic ambulatory setting—and as reviewed later in this chapter, in the setting of ACS.15,49 The CAPRIE (Clopidogrel versus Aspirin in Patients at Risk of Ischaemic Events) trial, which compared outcomes in patients with previous non–ST-segment elevation MI (NSTEMI), ischemic stroke, or established peripheral artery disease randomized to treatment with aspirin versus clopidogrel, enrolled 3866 patients with diabetes. In the subset of patients with diabetes, the 12.5% reduction in major adverse CVD events with clopidogrel versus aspirin paralleled the effect observed in the overall study cohort. In view of the incremental expense of clopidogrel and its associated increment in bleeding risk, however, this strategy is not routinely recommended over the use of aspirin alone for most patients, but clopidogrel remains an option for patients with aspirin indication but intolerance or allergy to aspirin. Diabetes is associated with an increased prevalence of resistance to clopidogrel, a prodrug requiring metabolic conversion that tends to be impaired in diabetes, resulting in decreased circulating active metabolite.49 These observations have led some investigators to explore the effects of increased dosing of clopidogrel in patients with diabetes, with preliminary data suggesting increased antiplatelet effects with such a strategy. The net clinical safety and efficacy of increased dosing of clopidogrel for chronic secondary prevention treatment, however, requires further evaluation before application in clinical practice. Numerous drug classes are approved for clinical use for the treatment of hyperglycemia associated with diabetes (Table 61-3). These drugs work by stimulating endogenous insulin release, impairing hepatic glucose production, improving the body’s response to insulin, delaying intestinal carbohydrate absorption, providing exogenous insulin, or increasing glucose urinary losses. TABLE 61-3 Glucose-Lowering Medications for Type 2 Diabetes Mellitus * Not licensed in the United States. † Prescribing highly restricted in the United States; withdrawn in Europe. ‡ Limited use in the United States/Europe. ‖ Not licensed in Europe. I| Depends on type (analogues > human insulins) and dosage. AMP = adenosine monophosphate; CKD = chronic kidney disease; DPP-4 = dipeptidyl peptidase 4; GIP = glucose-dependent insulinotropic peptide; GLP-1 = glucagon-like protein 1; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; PROactive = Prospective Pioglitazone Clinical Trial in Microvascular Events; STOP-NIDDM = Study to Prevent Non-Insulin-Dependent Diabetes Mellitus; UKPDS = United Kingdom Prospective Diabetes Study. From Inzucchi SE, Bergenstal RM, Buse JB, et al: Management of hyperglycemia in type 2 diabetes: A patient-centered approach: Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 35:1364, 2012. To date, the approval of drugs for diabetes depended almost exclusively on proof of glucose lowering without the requirement of demonstration of efficacy on clinical outcomes. The regulatory landscape for diabetes drugs has recently undergone major changes, such that all future diabetes drugs (and probably those currently on the market) must demonstrate designated margins of CVD safety to achieve or to maintain regulatory approval, leading to a rapid proliferation of clinical trials under way or in planning to assess CVD outcomes with these therapies50 (Table 61-4). In this context, the available data on the net CVD safety and efficacy of such medications are limited, and current management strategies and guidelines remain grounded on the proven microvascular disease benefits demonstrated with glucose control.16,22 TABLE 61-4 Summary of Ongoing Cardiovascular Outcomes Trials of Drugs for Type 2 Diabetes
Diabetes and the Cardiovascular System
Scope of the Problem
Diabetes Mellitus
Atherosclerosis
Heart Failure
Coronary Heart Disease in the Patient with Diabetes
Mechanistic Considerations Linking Diabetes and Atherosclerosis
Endothelium
Vascular smooth muscle cells and vascular matrix
Inflammation

Prevention of Coronary Heart Disease and Its Complications in the Setting of Diabetes
Lipid-Lowering Therapy
Statins
Omega-3 Fatty Acids
Fibric Acid Derivatives (Fibrates)
Niacin
Hypertension Management
Renin-Angiotensin-Aldosterone System Antagonists
Angiotensin-Converting Enzyme Inhibitors.
Angiotensin II Receptor Blockers.
Calcium Channel Blockers
Beta Blockers
Thiazide Diuretics
Combination Therapy for Hypertension
Blood Pressure Targets for Patients with Diabetes
Antihypertensive Therapy Summary
Antiplatelet Therapy
Daily Aspirin
Thienopyridines
Glucose Management
CLASS
COMPOUND(S)
CELLULAR MECHANISM
PRIMARY PHYSIOLOGIC ACTION(S)
ADVANTAGES
DISADVANTAGES
COST
Biguanides
Metformin
Activates AMP-kinase
↓ Hepatic glucose production
Low
Sulfonylureas
Closes KATP channels on beta cell plasma membranes
↑ Insulin secretion
Low
Meglitinides (glinides)
Closes KATP channels on beta cell plasma membranes
↑ Insulin secretion
High
Thiazolidinediones
Activates the nuclear transcription factor PPAR-γ
↑ Insulin sensitivity
Moderate
α-Glucosidase inhibitors‡
Inhibits intestinal α-glucosidase
Slows intestinal carbohydrate digestion/absorption
Moderate
DPP-4 inhibitors
Inhibits DDP-4 activity, increasing postprandial active incretin (GLP-1, GIP) concentrations
High
Bile acid sequestrants‡
Colesevelam
Binds bile acids in intestinal tract, increasing hepatic bile acid production; ? activation of farnesoid receptor (FXR) in liver
High
Dopamine-2 agonists‡
Bromocriptine (quick-release)‖
Activates dopaminergic receptors
High
GLP-1 receptor agonists
Activates GLP-1 receptors
High
Amylin mimetics‡
Pramlintide§
Activates amylin receptors
High
Insulins
Activates insulin receptors
Variable||
Cardiovascular Effects of Selected Drugs for Diabetes
TRIAL
DRUG
NO. OF PATIENTS
STAGE
NCT
TOSCAIT
Pio vs SU
3371
9/2008
NCT00700856
TECOS
Sitagliptin
14,000
Started 12/2008
NCT00790205
ACE
Acarbose
7500
Started 2/2009
NCT0829660
CANVAS
Canagliflozin
4500
Started 11/2009
NCT01032629
ELIXA
Lixisenatide
6000
Started 6/2010
NCT01147250
EXSCEL
Exenatide LAR
12,000
Started 6/2010
NCT01144338
EMPA REG Outcome
Empagliflozin
12,500
Started 7/2010
NCT01131676
CAROLINA
Linagliptin
6000
Started 10/2010
NCT01243424
LEADER
Liraglutide
8723
Started 8/2010
NCT01179048
REWIND
Dulaglutide
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree

 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

Diabetes and the Cardiovascular System
61
FIGURE 61-3 Adverse clinical outcomes after acute coronary syndromes during more than 1 year of follow-up, according to diabetes status, among patients participating in the TRITON–TIMI 38 randomized trial.8 CVA = cerebrovascular accident; CVD = cardiovascular death; D = death; DM = diabetes mellitus. (Modified from Wiviott SD, Braunwald E, Angiolillo DJ, et al: Greater clinical benefit of more intensive oral antiplatelet therapy with prasugrel in patients with diabetes mellitus in the Trial to Assess Improvement in Therapeutic Outcomes by Optimizing Platelet Inhibition with prasugrel-Thrombolysis in Myocardial Infarction 38. Circulation 118:1626, 2008.)
FIGURE 61-7 Meta-analysis of trials examining the effects of aspirin on risk of cardiovascular disease events in patients with diabetes. A, Effect of aspirin on coronary heart disease events. Tests for heterogeneity: χ2 = 8.71, P = 0.367, I2 = 8.2%. B, Effect of aspirin on risk of stroke in patients with diabetes. Tests for heterogeneity: χ2 = 12.48, P = 0.131, I2 = 35.9%. BMD = British Medical Doctors; ETDRS = Early Treatment of Diabetic Retinopathy Study; HOT = Hypertension Optimal Treatment; JPAD = Japanese Primary Prevention of Atherosclerosis with Aspirin for Diabetes; PHS = Physicians’ Health Study; POPADAD = Prevention of Progression of Arterial Disease and Diabetes; PPP = Primary Prevention Project; TPT = Thrombosis Prevention Trial; WHS = Women’s Health Study. (From Pignone M, Alberts MJ, Colwell JA, et al: Aspirin for primary prevention of cardiovascular events in people with diabetes: A position statement of the American Diabetes Association, a scientific statement of the American Heart Association, and an expert consensus document of the American College of Cardiology Foundation. Circulation 121:2694, 2010.)