16 Defibrillation Testing, Implant Testing, and Relation to Empiric ICD Programming
Defibrillation testing also provides important information that could be used to program a device. Such testing would also verify that the sensing thresholds are adequate to detect VF reliably. A low DFT can allow the programming of a low-output initial shock that would reduce postshock ventricular dysfunction.1 Treatment with antiarrhythmic medication such as amiodarone may raise DFTs such that defibrillation testing may be indicated.2 When a patient is known to have a sustained monomorphic VT, testing of antitachycardia pacing efficacy may allow optimal programming of such parameters to terminate clinical events more expeditiously and to lessen acceleration of VT or conversion to VF.
 Strength-Duration Nature of Defibrillating Shocks
Strength-Duration Nature of Defibrillating Shocks
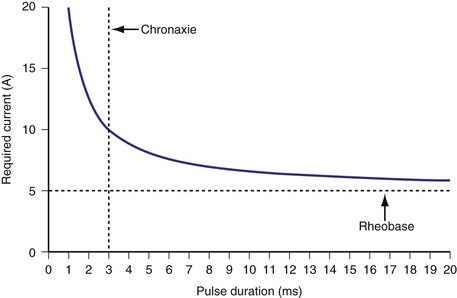
Similar to pacing impulses, defibrillation waveforms also have a strength-duration relationship3–11 (Fig. 16-1). However, important differences exist. In pacing, the strength-duration curve, marking the necessary current strength for a particular pulse duration that would elicit myocardial capture, separates the two-dimensional graph plane into two zones, one where capture consistently occurs and the other where there is no capture. These two zones are sharply demarcated by the strength-duration curve. That is, the probability of capture rises extremely steeply as one crosses the strength-duration curve from one zone to the other, so steeply that it appears to be almost an all-or-none phenomenon (Fig. 16-2).
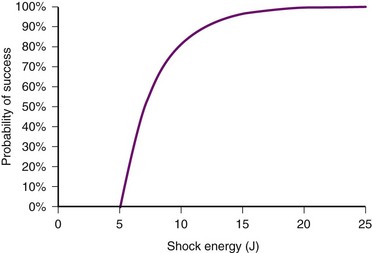
In contrast, the strength-duration relationship for defibrillation, described for monophasic waveforms or for the first phase of biphasic waveforms, is more probabilistic.12–14 For a particular pulse width, the probability of defibrillation changes over a much wider range of current strengths. Rather than a narrow line forming the strength-interval curve for pacing, this is a wide, fuzzy band where, at a particular pulse width, the probability of defibrillation at the lower end of the band is near zero and at the upper end of the band, near 100%. Alternatively, the strength-duration relation of defibrillation can be seen as a family of strength-duration curves having varying probabilities of defibrillation. For example, one curve can define the strength-duration of a waveform that would achieve a 50% successful defibrillation rate (DFT50). Another curve, located higher on the graph, would represent 90% successful defibrillation rate (DFT90).
The differences between the pacing and defibrillation strength-duration curves can be attributed to several factors. Pacing occurs during diastole, where the resting membrane state is quite stable. Defibrillation, in contrast, occurs when the myocardium is in a highly inhomogeneous state of depolarization and repolarization. Multiple wavefronts may exist, the paths of which vary over time. Thus, the distribution of such depolarizing and repolarizing wavefronts is continuously changing. During pacing, the impulse needs to elicit an action potential in only a small volume of myocardium directly under the electrode, whereas the defibrillation shock needs to extinguish all or a critical number of wavefronts throughout the myocardium. It has been demonstrated that a minimum gradient of around 5 V/cm needs to be achieved throughout the myocardium to defibrillate at 80% effectiveness with a monophasic waveform, to obtain a high probability of terminating VF.15 The postulated mechanisms by which defibrillation shocks terminate VF have changed over the years. Multiple factors influence DFT, including size of the heart, location of the right ventricular (RV) electrode, polarity of the first phase, waveform durations, system impedance, heterogeneity of conduction and repolarization, number of propagating wavelets,16 virtual electrode effect, and non-RV electrode locations. Several reports have detailed these factors, primarily antiarrhythmic drugs17–21 (Table 16-1); a thorough discussion is beyond the scope of this chapter.
TABLE 16-1 Drugs and Clinical Conditions Affecting Defibrillation Threshold (DFT)
| Drugs/Condition | Effect on DFT | |
|---|---|---|
| Drugs | Atropine, azimilide, diltiazem, fentanyl, halothane, isoflurane, lidocaine, mexiletine, moricizine, phenylephrine, sildenafil (IV), verapamil | Increase |
| Sotalol, dofetilide, ibutilide, β-blockers | Decrease | |
| Amiodarone,* epinephrine, flecainide, isoproterenol | Variable | |
| Clinical/Other | High shock impedance, intracellular hypercalcemia, pneumopericardium, pneumothorax | Increase |
| Hyperkalemia, atrial natriuretic peptide (ANP) | Decrease | |
| Electrode polarity,* electrode position | Variable |
 Shock Waveform
Shock Waveform
Although early implantable defibrillators tended to have larger capacitors, experimental studies demonstrate that lower capacitance values of 60 to 90 microfarad (µF) offer more efficient defibrillation energies, thus minimizing use of battery power.22–27 Technology limits the maximum voltage that capacitors can be charged, and concerns regarding the adequacy of small capacitor pulse width in the presence of low impedance have resulted in most defibrillators now having capacitance in the 90-µF range. This capacitance allows an adequate pulse width of the first phase at typical impedances seen in human implants and still preserves enough charge for delivery during the second phase.
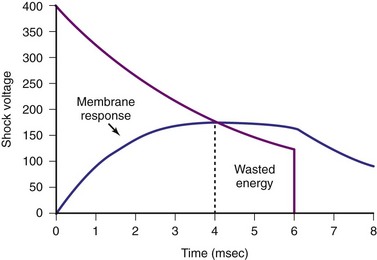
Pulse width is important because the effect of a shock delivered to the myocardium is to depolarize the cell membrane. When the myocardial cell is exposed to a sudden voltage gradient generated by the shock, the cell membrane responds by changing the transmembrane voltage. The rate of such change in transmembrane voltage is not instantaneous but exponential. Experimental studies have defined the ranges of time constants of such depolarization.28,29 These time constants indicate that charging of the cell membranes would essentially be near maximum with pulse widths of 4 to 5 msec. Thus, pulse widths beyond 5 to 7 msec probably do not add much to the defibrillation effect30,31 (Fig. 16-3). In fact, very long pulse widths (>10 msec) may have detrimental effects in defibrillation.32,33 Capacitors larger than 90 µF can easily deliver waveforms with optimal duration, but would leave a large residual charge in the capacitor at the end of a shock, wasting battery power. In addition, because the capacitors occupy the largest volume in an ICD generator, these larger capacitors would result in larger ICD generators. Much of the reduction in current generator size has resulted from the ability to reduce the capacitor size used in earlier ICDs.
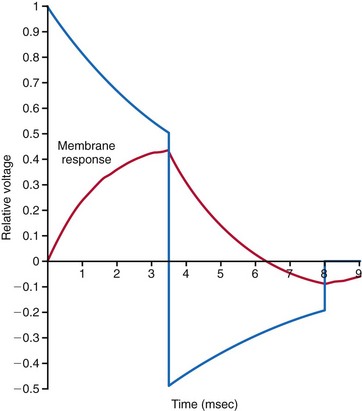
Since the early 1990s, biphasic waveforms have become standard in all implantable defibrillators. Early work on the biphasic waveform33 led to experimental34–38 and then human39,40 studies that demonstrated the superiority of the biphasic waveforms over the monophasic in achieving lower DFTs.34–40 However, the mechanism of this superiority was postulated theoretically and demonstrated experimentally much later. The “charge burping” theory was postulated in 1994 to explain this superiority.41 Essentially, this theory proposed that the residual charge on the myocardial cell membrane at the end of a monophasic shock is proarrhythmic, because it could initiate new waves to continue VF. Thus, the second phase of the biphasic waveform served to discharge the residual transmembrane polarization so as to eliminate this proarrhythmic effect (Fig. 16-4).
Clinical testing supported the predictions of the charge-burping theory.42 Experimentally, the phenomenon of the second-phase effects corroborated this theory and provided additional insight into how polarity matters. Recordings of actual transmembrane voltage changes during the shock waveform became possible once technology to record transmembrane voltages using optical techniques became available. Before this, transmembrane voltages could be measured only by direct electrical recordings. Such recordings, in the millivolt range, are completely overwhelmed by the electrical artifacts of the shock, in the hundreds of volts, three to five orders of magnitude larger. The optical techniques essentially utilize voltage-sensitive dyes impregnated into the cell membrane, whose florescence changes depending on the transmembrane voltage. This fluorescence can be measured optically; thus the electrical artifacts of the shock would not affect such measurements. Until the development of these optical techniques, the transmembrane effects of the shock waveform could not be directly observed.43 A series of experimental studies using optical techniques then provided direct observational data on the potential mechanisms of the superiority of biphasic waveforms.44 Furthermore, these studies also demonstrated how anodal shocks, especially with monophasic waveforms, could be more effective.
During delivery of the shock, the cardiac cell membrane surrounding the region of the shock electrode does not depolarize or hyperpolarize in a uniform manner. This nonuniform depolarization pattern has been labeled the virtual electrode effect and was predicted to occur based on computer modeling of cardiac tissue response to an externally applied, single-source (or point-source) voltage gradient.45 Interestingly, the nonhomogeneous pattern consists of a depolarization area near the electrode, surrounded by areas of hyperpolarization in the case of a cathodal shock, and vice versa with an anodal shock. At the end of a cathodal shock, the depolarized area in the center of the virtual electrode effect enhances propagation of a new wavefront toward surrounding tissue of hyperpolarized membranes. This increases the propensity to generate new wavefronts that would perpetuate VF. With an anodal shock, the favored propagation is from the surrounding areas toward the center of the virtual electrode effect. This direction of propagation favors the extinguishing of wavefronts as they collide toward the center, thus reducing the propensity to generate new wavefronts that would perpetuate VF. These same effects explain why cathodal pacing has a somewhat lower threshold than anodal pacing.
These observations also provide a mechanistic explanation of why monophasic shocks with an anodal polarity have a lower DFT. Observations of biphasic shocks using optical recordings of transmembrane voltages show that reentrant wavefronts generated at the end of a monophasic shock cannot propagate when the second phase reduces (cancels out) the virtual electrode effect. Thus, the basic mechanism of the “burping” hypothesis was experimentally confirmed.46 Theoretically, a biphasic waveform in which the second phase appropriately cancels out the remaining transmembrane voltages at the end of the first phase should demonstrate no difference in DFT between anodal and cathodal shocks.
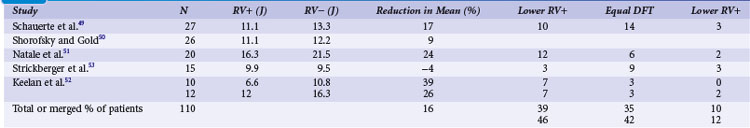
Clinical studies with the monophasic waveforms consistently demonstrate that anodal shocks are more effective than cathodal shocks.47,48 With the biphasic waveform, anodal shocks have been more effective in a majority of clinical studies,49–52 although one study reported slightly improved cathodal (9.9 J) versus anodal (9.5 J) DFT53 (Table 16-2). Experimental animal studies using more optimized first-phase and second-phase waveforms, in which more thorough testing of DFT is feasible, have shown no difference in anodal versus cathodal first-phase shocks.54 The occasional patient who demonstrates a better cathodal then anodal DFT may be explained in two ways. First, clinical testing necessarily limits the number of defibrillation trials during an implantation. Thus, based on the probabilistic nature of defibrillation, an occasional patient may show a defibrillation at slightly lower shock strength with a cathodal shock than with an anodal shock, especially if the biphasic waveform is near ideal, such that the two waveforms would be comparable. Second, but less likely, when the second phase delivers an excessive amount of charge, a cathodal-type virtual electrode effect would occur at the end of the second phase if the first phase was anodal, thus making the shock less effective than the reverse polarity. Kroll et al.55 explored first-phase shock polarity and advocated for routine programming of an anodal first phase.
In practice, it is difficult to determine exactly how to optimize the first and second phases so as to achieve the best cancellation of the virtual electrode effect across the entire myocardium. Thus, programming the first phase as an anodal shock is generally the best practice. Calculations have been reported of the “optimal” first and second phases to provide the proper charging and then “burping.”56,57 In devices where the pulse width of the two phases can be programmed, optimizing these parameters can sometimes yield an acceptable DFT when the “standard” pulse widths do not provide an adequate DFT safety margin.
 Postimplant Testing
Postimplant Testing
Arrhythmia Detection
Implantable defibrillators are generally tested at implantation or generator replacement for appropriate detection, pacing, and therapy effectiveness. Once the generator has been attached to the new or existing lead(s), sensing parameters of intrinsic cardiac rhythm (if present) are usually tested first. In some devices, lead testing is performed using a pacing analyzer before attachment to the ICD generator. R waves of 5 mV or greater are usually considered the minimum necessary amplitude, but in some clinical situations, lower amplitudes may be acceptable. During tachyarrhythmias, in particular VF, ventricular detection amplitudes may be substantially reduced, which is why ICD generators contain “autogain” amplifiers.58 This function automatically increases the sensitivity of the device to detect R waves of lower amplitude. In most cases, it is desirable to position the ICD lead where the greatest R-wave amplitude is observed, to minimize underdetection. ICD manufacturers provide additional programmable detection functions to improve or reduce these sensitivity settings for specific clinical situations. For example, patients with long QT syndrome may exhibit oversensing caused by T-wave detection outside the usual detection “window” where various T-wave rejection algorithms are active. This can result in inappropriate shocks caused by “double counting.” Reprogramming the parameters affecting the autogain/autothreshold functions of the ICD after a sensed R wave may help prevent this problem.
Defibrillation Testing
There is some controversy as to whether defibrillation testing should be part of a new implant or generator change. We favor such testing for the reasons listed here, provided the patient’s clinical condition makes this a low-risk test. Defibrillation testing usually consists of induction of VF by various means: T-wave shocks, delivery of a low-amplitude direct current (DC) through the lead, or more rarely, rapid burst pacing. The T-wave shock method is based on the presence of a vulnerable window surrounding the peak of the T wave within which a low-amplitude shock will induce VF59 (Fig. 16-5
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree