Cardiac Malpositions and Abnormalities of Atrial and Visceral Situs
Patrick W. O’Leary
Muhammad Yasir Qureshi
Donald J. Hagler
Cardiac malposition describes an abnormal or anomalous location of the heart. The discussion of cardiac malposition includes abnormalities of overall cardiac position (dextrocardia, mesocardia, levocardia [isolated]), the heterotaxy syndromes, pericardial defects, and ectopia cordis (1,2). Cardiac malpositions often are integral components of complicated cardiac anomalies involving abnormal visceral and atrial situs (sidedness). Due to the complex nature of these anomalies, pathologists and clinicians have advocated using a logical, systematic, and consistent method for describing the anatomy associated with these lesions. This method of analysis is described as “the sequential, segmental approach to the diagnosis of congenital heart disease,” and is worth reviewing before embarking on a description of the individual anomalies that include abnormal organ positions. This approach begins with a description of the asymmetrically arranged organ systems of the abdomen and thorax (determination of situs), proceeds to define the cardiac position and orientation within the chest, and concludes with a detailed description of the venous, cardiac, and arterial segmental anatomy, as well as the spatial relationships of the segment components and the connections of adjacent segments to one another. We will begin with a review of this approach and conclude by discussing the wide variety of cardiovascular anomalies and problems that can be encountered in patients with abnormal cardiac positioning.
Definitions
Understanding of the complex malformations associated with abnormal cardiac position is facilitated by a clear definition of the terminology used to describe them.
The classification of overall cardiac position is determined by the cardiac base–apex axis as illustrated in Figure 51.1. Dextrocardia has been defined classically and most consistently as the location of the heart in the right hemithorax with the apex pointing (base–apex axis) inferiorly and to the right. Dextrocardia may occur with atrial situs solitus, situs inversus, or situs ambiguus. When it occurs with situs solitus, it has also been called isolated dextrocardia. Although the normal heart can be “shifted” into the right chest by a variety of extracardiac processes (diaphragmatic hernia, tumors, or tension pneumothorax), these situations, in which the cardiac base–apex axis is still oriented to the left, are better described as “dextroposition,” rather than dextrocardia.
Mesocardia occurs when the location of the heart is essentially in the midline, with the cardiac base–apex axis pointing directly inferior (neither to the right nor left). The ventricular apices are equally spaced, near the xiphoid process, to the right and the left side of the sternum. This anatomic cardiac malposition is often overlooked as being infrequent and atypical. However, many cases accepted as having dextrocardia may be described more accurately as mesocardia, because of a midline cardiac position. Most cases of mesocardia have been described with situs solitus. However, mesocardia also has been described with situs inversus and situs ambiguus.
Levocardia describes the normal position of the heart and should only be considered a cardiac malposition when it occurs in conjunction with situs inversus or situs ambiguus of the atria. In these situations, the cardiac position may be described as “isolated levocardia.” Thus, despite abnormal atrial and visceral situs, the heart (ventricles) is in the left hemithorax with the apex (base–apex axis) pointing inferiorly and to the left.
Situs (or sidedness) is a term used to describe the spatial relation of an asymmetrically arranged organ system (or group of related organs). For our discussion, the situs of the abdominal organs (stomach, liver, bowel, and spleen) and the atria will receive the most attention. Situs solitus describes the normal site or position of the abdominal organs or atria. Typically, they must be independently considered and described for a precise segmental description. Situs inversus describes an inversion or right/left reversal of visceral or atrial positions, that is, a mirror image of the normal situation. In cases of atrial situs inversus, the right atrium will lie to the left and the anatomic left atrium to the right. Abdominal situs inversus consists of a right-sided stomach and spleen, and a left-sided liver. Situs ambiguus describes an uncertain or indeterminate visceral or atrial position, usually because the structures have taken on a symmetrical or indeterminate anatomic form bilaterally. A common example of this symmetry is seen in patients with abdominal situs ambiguus. These individuals will have a liver that is “bilateral” (equally occupying both the right and left upper quadrants) and is in a midline position. Both lobes of these bilaterally symmetrical livers will resemble a “normal” right lobe and these patients commonly will have absence of the spleen (asplenia). Figure 51.2 illustrates the normal situation and three types of abdominal organ positions (visceral situs).
Heterotaxia is the term used to describe a constellation of anomalous viscera situs and related groups of complex cardiovascular malformations. These “syndromes” are often seen with a loss of the typical left–right organ asymmetry usually seen in the abdomen, lungs and the heart. Variations from the normal (situs solitus) in heterotaxy are expressed as randomization (situs ambiguus), bilateral symmetry (mirror images), or as complete reversal of visceral position (situs inversus). In 1993, Casey et al. (1,2) localized a genetic defect in familial heterotaxy. They found an X-linked recessive trait associated with the inability of the developing embryo to establish normal left–right asymmetry. They later localized this deletion to X q26.2. They further reported that familial heterotaxy occurs with autosomal dominant, recessive,
and X-linked inheritance. Kato et al. (3) and Peeters et al. (4) have reported de novo balanced translocations involving chromosome band 6q21 in a patient with heterotaxia and left isomerism. Genes implicated in human heterotaxy include Zic3, LeftyA, Cryptic, and Acvr2B (5). Both situs ambiguus and situs inversus can appear in some families with heterotaxy.
and X-linked inheritance. Kato et al. (3) and Peeters et al. (4) have reported de novo balanced translocations involving chromosome band 6q21 in a patient with heterotaxia and left isomerism. Genes implicated in human heterotaxy include Zic3, LeftyA, Cryptic, and Acvr2B (5). Both situs ambiguus and situs inversus can appear in some families with heterotaxy.
Sequential Segmental Approach to Cardiac Diagnosis
A segmental approach implies a systematic and therefore sequential review of all structures involved in the congenital cardiovascular anomalies. This approach would determine the pathologic status of the viscera, the atria, the atrioventricular (AV) connection(s), the ventricles, the ventriculoarterial (VA) connection(s), and the great arteries. Van Praagh included 10 cardiac segments from an embryologic standpoint: the sinus venosus, the primitive atrium, the common pulmonary vein, the AV canal, the primitive ventricle (proampulla), the proximal bulbus cordis (meta-ampulla), the infundibulum (conus), the truncus arteriosus, the aortic sac, and the arterial arches (6). However, from a practical clinical approach, some embryologic segments have little relevance or application for anatomic or pathologic examination.
TABLE 51.1 Segmental Approach to Evaluation of Complex Congenital Heart Disease | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Therefore, a more practical clinical and pathologic segmental analysis begins with definition of major organ positions (visceral situs, atrial situs, and cardiac position/orientation), followed by a detailed description of four segments and the three connections between them. The seven components of this system are (a) the venous segment (systemic and pulmonary veins, plus the coronary sinus), (b) the venoatrial connections, (c) the atria and the atrial septum, (d) the AV connection(s), (e) the ventricles and the ventricular septum, (f) the VA connection(s), and (g) the great arteries (aorta, pulmonary arteries, coronary arteries, and ductus arteriosus). Table 51.1 illustrates a basic strategy for segmental diagnosis including many criteria and anatomic features that must be determined for accurate diagnosis. Additional anatomic landmarks are outlined in Table 51.2. This approach should be applied regardless of the method of examination being used (any clinical imaging technique or a pathologic examination).
TABLE 51.2 Anatomic Features of Cardiac Segments | |||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Visceral and Atrial Situs
Abdominal Organs
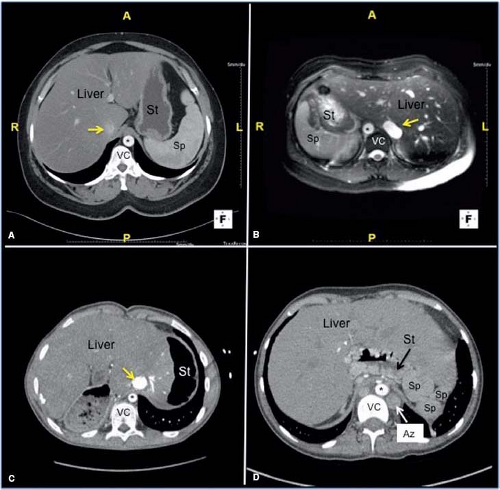
As illustrated in Table 51.1, the evaluation begins with definition of visceral situs. Visceral (abdominal) situs is determined by the positions of the liver and stomach (see Fig. 51.1). The pancreas and spleen are generally located on the same side of the vertebral column as the stomach. Atrial and visceral situs often are considered together because they are usually concordant (the atrial and visceral situs are the same). However, exceptions occur, and venous connections and visceral situs may not necessarily agree with the apparent atrial situs. These features can be particularly variable in situs ambiguus. Situs ambiguus may be best defined as an uncertain or indeterminate situs (organ positions do not fit into any standard category). However, in many cases, the right and left components appear very similar to each other, and have been described as mirror images. Such cases also have been referred to as “isomeric” or as having bilateral right or left sidedness.
In visceral situs ambiguus with right isomerism (bilateral right sidedness), the spleen is usually absent (asplenia) and the liver is centrally located, symmetrically straddling the midline (see Fig. 51.1). In this situation, two mirror-image morphologic right hepatic lobes are present. In contrast to the normal arrangement, the inferior vena cava (IVC) and abdominal aorta will be found on the same side of the vertebral column. In addition, these patients almost always will have malrotations of the bowel.
Situs ambiguus with polysplenia has been described as bilateral left sidedness or left isomerism (5,6,7,8,9,10,11,12). However, the degree of right/left symmetry is less pronounced in these patients, compared to that seen in asplenia and bilateral right sidedness. In fact, the most common arrangement of the abdominal viscera in polysplenic patients is situs inversus. As a teaching tool, the concept of isomerism (mirror-image sidedness) is an attempt to simplify the typical features of complex anomalies in which multiple abnormalities tend to occur together. Thus, the asplenia syndrome (Ivemark syndrome) appears as a pathologic grouping of features emphasizing right sidedness, such as bilateral right bronchi and bilateral right (trilobed) lungs, bilateral right atria, and a symmetrical liver. Ivemark described the implications of asplenia on the pathogenesis of conotruncal anomalies in 1955. Van Mierop et al. (12) pointed out that the asplenia syndrome is associated with pulmonary stenosis or atresia, whereas in polysplenia syndrome, presence of single ventricle physiology and obstruction to pulmonary arterial blood flow are less common. In contrast, interruption of the IVC with azygos continuation of the inferior venous system and anomalous, bilateral
pulmonary venous connection of each lung to the ipsilateral atrium are common in polysplenia, but rare in asplenia. Tables 51.3 and 51.4 outline the frequency of both extracardiac and cardiac malformations in cases of heterotaxia. The frequencies represent a compilation of cases reported by multiple investigators (13,14,15,16).
pulmonary venous connection of each lung to the ipsilateral atrium are common in polysplenia, but rare in asplenia. Tables 51.3 and 51.4 outline the frequency of both extracardiac and cardiac malformations in cases of heterotaxia. The frequencies represent a compilation of cases reported by multiple investigators (13,14,15,16).
TABLE 51.3 Frequency of Extracardiac Findings in Heterotaxy Syndromes | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Although splenic anomalies are often associated with complex congenital heart disease and with abnormalities of visceral situs, they are not the cause. Embryologically, the spleen, in contrast to the other thoracoabdominal viscera, does not initially develop as a midline structure. Rather, the splenic anlage is left sided from its inception. As a result, in the case of bilateral right-sided symmetry, the spleen would not be expected to develop—hence the association of right isomerism with asplenia. In contrast, left isomerism is frequently associated with polysplenia, although multiple spleens are usually found on only one side of the vertebral column (along the dorsal aspect of the stomach).
Cardiac Atria
Clinically, the identification of the morphologic right atrium is important for establishing atrial situs. This is due to the fact that much of the anatomic left atrium is derived from the embryonic common pulmonary vein. As a result, its walls are relatively smooth and lack distinctive features for identification by clinical imaging modalities. In contrast, the anatomic right atrium is derived from the primitive atrium and has several muscular structures that make it more recognizable. Its most reliable intrinsic anatomic hallmark is the relatively thick superior limbus of the fossa ovalis, which can be observed by direct inspection at operation, at autopsy, or by tomographic imaging techniques (echocardiographic, CT, or cardiac MR scanning). In cases in which the atrial septum is absent, distorted, or cannot be visualized, atrial morphology may be inferred by inspection of the free atrial wall. In contrast to a left atrium, a morphologic right atrium will have a large pyramidal appendage, a crista terminalis, and pectinate muscles (14). In situs ambiguus and other complex cases, the atrial septum can essentially be absent, resulting in a common atrium. In these cases, assignment of atrial “identity” can be challenging and often must rely on secondary, associated features. The presence of pectinate muscles and the crista is helpful in anatomic specimens. However, these are not always clearly defined by cardiac imaging. In these situations, the connections of the coronary sinus and/or the suprahepatic segment of the IVC provide the most reliable and readily recognizable markers identifying the position of the morphologic right atrium (16,17). Anatomic determinants of the other cardiac segments also have been described and are summarized in Table 51.2.
TABLE 51.4 Frequency of Cardiac Findings in Heterotaxy Syndromes | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
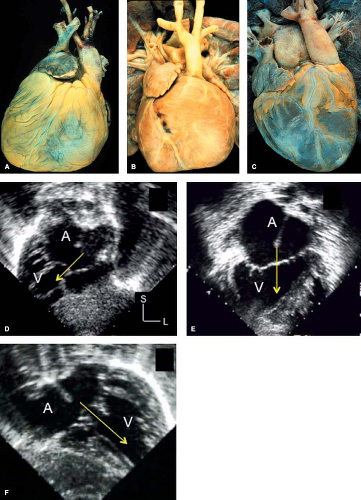
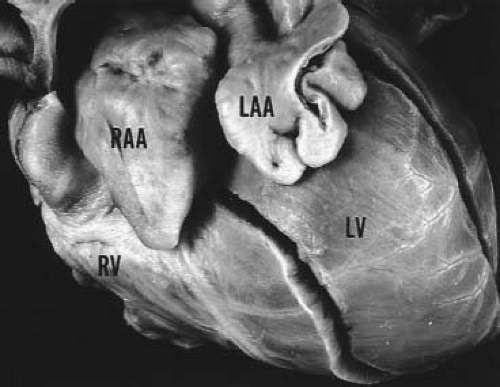
One can encounter cases of truly bilateral right atria (or right atrial isomerism). In these patients the appearance of both atrial appendages and the atrial free walls will have the characteristics of a right atrium. The coronary sinus is usually absent in these cases and IVC connection can be ambiguous as well (bilateral or midline hepatic veins). Even more rarely, in polysplenia syndrome with left atrial isomerism, both atrial appendages and free walls may resemble a left atrial appendage. The left atrial appendage usually has a narrow base and a tubular, hooked shape (Fig. 51.3) (18). However, in most cases with asplenia or polysplenia, the atrial anatomy is poorly defined, with a large common atrium, a thin rudimentary remnant of atrial septum, anomalies of systemic and pulmonary venous connections, and abnormal, but similarly shaped bilateral appendages.
Van Praagh et al. (16) have argued that the concept of bilateral or two right atria and bilateral or two left atria is anatomically unrealistic. These researchers and also De la Cruz and Nadal-Ginard (17) emphasized the importance of the suprahepatic segment of the IVC as a reliable marker for the morphologic right atrium, because embryologically, the sinus venosus is incorporated into the right atrium, and IVC-to-right atrium concordance is diagnostically accurate, even when there is visceroatrial discordance. With incorporation of the sinus venosus, the coronary sinus also can be recognized as a reliable marker for the morphologic right atrium. The morphologic right atrium, then, can be determined by a normally formed coronary sinus, a larger and more anterior appendage, and connection of the suprahepatic portion of the IVC. This approach allows clinical assignment of atrial situs in most cases, although the most important clinical issues are often related to the position and spatial arrangement of the venoatrial connections rather than the morphologic identities of the atria themselves.
The Lungs
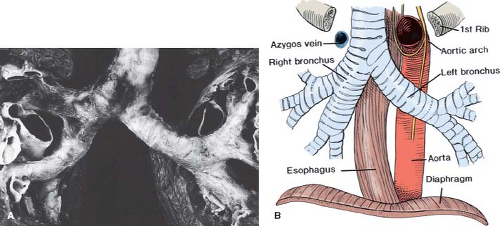
Pulmonary situs is determined by the sidedness of the morphologic right and left lungs. Clinically, this is defined by the relationship of the pulmonary arteries to their adjacent bronchi, and not by the number of lobes in each lung. In a morphologic right lung, the pulmonary artery travels anterior to the upper lobe and intermediate bronchi. In contrast, a morphologic left lung is characterized by a pulmonary artery that courses over the main bronchus and posterior to the upper lobe bronchus. The distance from the carina to the origin of the upper lobe bronchus is 1.5 to 2 times greater for the morphologic left lung than for the right lung (19). These relative distances may be used clinically to infer pulmonary situs. The simplest method is by examination of the air bronchograms on a plain chest x-ray. This ratio applies regardless of the sidedness of the aortic arch (i.e., regardless of the bronchus over which the aorta travels) (Fig. 51.4). In the setting of pulmonary isomerism (both lungs having the same morphology), however, the ratio approaches unity, because the lengths of the two main bronchi are similar. In these cases, bilateral trilobed (right) lungs should suggest the diagnosis of situs ambiguus with the asplenia syndrome (right isomerism), and bilateral bilobed (left) lungs the polysplenia syndrome (left isomerism). However, lung isomerism does not exist in all cases of cardiac isomerism and pulmonary isomerism can occur in patients without features of cardiac heterotaxia.
The cardiac and vascular segments must be evaluated not only according to morphology, but also in terms of spatial orientation. For example, atria and ventricles should be described both by their morphology and their right or left sidedness. In general, the position and orientation of the ventricular septum is used to describe the location of the ventricular chambers. In hearts with levocardia with normally positioned ventricles, the right ventricle (RV) is to the right and anterior, while the left ventricle (LV) is left and posterior to the septum. In contrast, in situs inversus totalis the usual ventricular arrangement is the LV–right posterior and the RV–left anterior. Mesocardia is characterized by a vertical midline septum with side-by-side ventricles. Rarely, the ventricular septum is horizontal rather than vertical and results in superoinferior (over-and-under or “upstairs–downstairs”) ventricles.
The position of the aorta is described relative to the pulmonary trunk. Normal hearts are characterized by a right posterior aorta. After the morphology and position of the cardiac segments are determined, the manner in which they connect to one another is evaluated. The three forms of connections to be investigated are venoatrial, AV, and VA.
The difficulties encountered in the accurate definition of atrial situs based on atrial anatomy alone emphasize the importance of accurate determination of systemic and pulmonary venous connections, as described in Tables 51.1 and 51.2. Regardless of the designation of a right or left atrium or the shape of the atrial appendages, the localization of the superior vena cava (SVC), IVC, hepatic veins, coronary sinus, and pulmonary venous connections should be determined individually and recognized as unilateral or bilateral. This determination is particularly important for surgical management and should be a routine part of any imaging study.
Atrioventricular Connections and Valve Morphology
The AV connection defines which atrium is connected or aligned with which ventricle. The normal AV connection, or right atrium-to-RV connection (and left atrium-to-LV), defines a concordant AV connection. Whereas a discordant connection is the abnormal relationship of the right atrium to the morphologically LV, while the left atrium connects to the morphologically RV. In hearts manifesting complex positional abnormalities, both common atrial to ventricular connection (common AV valve related to both atria) and univentricular AV connections are not unusual. In fact, most heterotaxy patients with univentricular physiology will demonstrate a common AV valve and connection.
Ventricular Positions and Morphology
Complete assessment of this segment includes an assessment of the cardiac base–apex axis to describe the three basic possibilities of levocardia, dextrocardia, or mesocardia as detailed previously. As covered elsewhere in this chapter, an evaluation of this segment should incorporate descriptions of the ventricular locations, relationships, function, and morphology, as well as the anatomy of the ventricular septum and any septal defects.
Great Artery Relations
The great arterial connections, the spatial relationships between the arteries and their subsequent distribution/branching patterns, must be determined as described in Table 51.1. Eight basic types of great artery relationship are possible based on the aortic and pulmonary valve positions at the level of the semilunar valves. They can be best described by the aortic location as follows:
Right posterior aorta (normally related)
Right lateral aorta (side by side)
Right anterior aorta (d-malposed)
Directly anterior aorta
Left anterior aorta (l-malposed)
Left lateral aorta (left side by side)
Left posterior aorta (inverted normal)
Directly posterior aorta
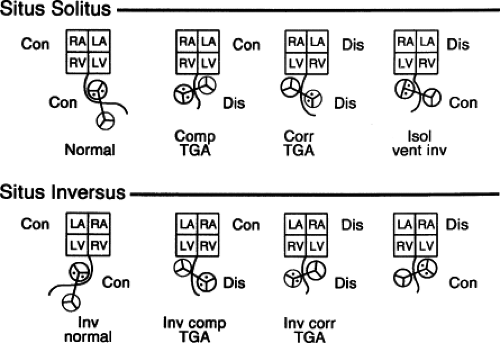
This type of segmental approach to complex cardiovascular malformations allows prediction of all possible cardiac conditions that could be encountered. Its flexibility depends upon the sequential addition of each cardiac segment and relevant connections, accounting for all possible combinations of atrial, ventricular, and great arterial anomalies. Figure 51.5 schematically illustrates four typical clinical examples of segmental variations occurring in both atrial situs solitus and situs inversus.
Cardiac Imaging: Use of the Segmental Approach
Transthoracic echocardiography is a convenient and noninvasive technique that can provide most of the clinically relevant information concerning the cardiovascular anatomy and physiology, especially in young patients. In situations where a transthoracic examination is inadequate (i.e., patients with poor transthoracic images, large body size, or postoperative bandaging, etc.) or impossible (during an operation), cardiac magnetic resonance (cMR), computed tomography (CT), or transesophageal echocardiographic scans can be used to obtain additional anatomic and functional information. We will primarily use echocardiographic examples to illustrate the segmental approach, but the concepts are applicable to, and should be used in all tomographic imaging modalities.
The imaging approach to patients with cardiac malpositions should follow the same sequential segmental pattern used by the anatomist/pathologist as outlined earlier in this chapter and in many reports (20,21,22,23). The following section and accompanying images of normal and malpositioned hearts are included to illustrate the tomographic and three-dimensional anatomy associated with these malformations and to assist the reader in correlating the imaging findings with anatomic examples of these complex hearts.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree