Atrial Septal Defects
Ritu Sachdeva
Introduction
Atrial septal defects (ASDs) originate at particular sites in the atrial septum and are named according to their embryonic origin. The most common occurs in the central part of the atrial septum in the region of fossa ovalis (secundum ASD). Others include those in the region of endocardial cushion (primum ASD), in the sinus venosus septum (sinus venosus ASD), and in the region of ostium of coronary sinus (CS) (CS ASD). In this chapter, we also address patent foramen ovale (PFO), which is a normal interatrial communication present in fetal life that may persist in adults. Ostium primum ASD is discussed in Chapter 29 since it is a part of the spectrum of atrioventricular septal defects (AVSD).
Incidence
ASDs constitute 8% to 10% of congenital heart defects in children. The incidence of ASDs has been estimated to be 56 per 100,000 live births (1). The recent estimates are much higher (100 per 100,000 live births), likely due to increased recognition of ASDs in this era of the common use of echocardiography (2). The female:male ratio for secundum ASDs is 2:1, but for the sinus venosus ASDs it is 1:1 (3,4). Secundum ASDs constitute approximately 75% of ASDs, followed by ostium primum ASD (20%) and sinus venosus ASD (5%) (5). CS ASDs more often are seen in association with heterotaxy syndromes and systemic venous anomalies, and isolated CS ASDs are rare (<1%).
Genetic and Environmental Risk Factors
Although most ASDs occur sporadically, familial modes of inheritance have been reported. The risk of congenital heart disease in offspring of a woman with a sporadic ASD is estimated to be 8% to 10% (6). ASDs may be related to mutations in either regulatory genes or their target sarcomeric genes. Heterozygous mutations in the transcription factor NKX2.5/CSX were among the first found in families with autosomal dominant transmission of secundum ASDs (7). Mutations in other transcription factors such as TBX5, GATA4, GATA6, and TBX20 also have been associated with secundum ASDs (7,8,9,10,11). TBX5 mutations also are responsible for Holt–Oram syndrome, which is an autosomal dominant syndrome characterized by secundum ASD, anomalies of the upper extremities, and atrioventricular (AV) conduction delay (12). A locus on chromosome 14q12 with a missense mutation in alpha-myosin heavy chain (MYH6), a structural protein expressed at high levels in the developing atria, has been linked to dominantly inherited ASD (13). Secundum ASDs also have been reported in association with cardiomyopathies that have resulted from mutations in sarcomeric genes (14,15). In ASDs associated with prolonged AV conduction, an autosomal dominant pattern of inheritance has been reported (16). Secundum ASDs also are associated with other syndromes such as Noonan, Down, Klinefelter, Williams, Kabuki, Goldenhar, and Ellis–van Creveld. In addition to genetic factors, maternal diseases and exposure to environmental risk factors may play a role in development of ASDs (17). These factors include pregestational diabetes, phenylketonuria, influenza and exposure to retinoids, nonsteroidal anti-inflammatory drugs, anticonvulsants, thalidomide, smoking, and alcohol (17,18,19).
Pathogenesis and Anatomic Features
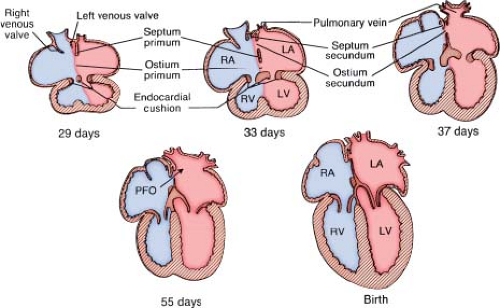
During embryogenesis, the primitive atrium undergoes a complex septation process (Fig. 28.1) (20). In the fourth week of embryonic life, the septum primum appears as a thin-walled sagittal fold in the middle of the common atrium and grows inferiorly toward the endocardial cushion. The opening between the leading edge of the septum primum and the endocardial cushion is called the ostium primum. Before complete closure of the ostium primum, tissue reabsorption occurs in the superior portion of the septum primum resulting in another opening called the ostium secundum. This occurs during the fifth and sixth week of embryonic life. Concurrently, an anterosuperior infolding of the atrial roof develops to the right of the septum primum, called the septum secundum that is concave shaped with a superior and inferior limb. The inferior limb fuses with the lowermost part of the atrial septum to join the endocardial cushion, thus separating the inferior portions of the two atria. The thick muscular ridge of the superior limb forms an incomplete
partition that overlies the ostium secundum resulting in an opening called the foramen ovale. The septum secundum thus forms the concave-shaped superior margin of the fossa ovalis, called the limbus of fossa ovalis (annulus ovalis) and the septum primum forms the valve of fossa ovalis. During fetal life, inferior vena caval flow from the placenta is deflected toward the foramen ovale by the eustachian valve, and then blood is directed from the right atrium to the left atrium via the foramen ovale. This fetal interatrial communication (the PFO) normally closes after birth as a result of fusion of the septum primum and septum secundum. However, it may persist in 25% to 30% of adults where it is probe patent with a competent valve. In some cases, the valve of fossa ovalis is incompetent, either congenitally or acquired due to elevated right or left atrial pressures allowing interatrial shunting across the foramen.
partition that overlies the ostium secundum resulting in an opening called the foramen ovale. The septum secundum thus forms the concave-shaped superior margin of the fossa ovalis, called the limbus of fossa ovalis (annulus ovalis) and the septum primum forms the valve of fossa ovalis. During fetal life, inferior vena caval flow from the placenta is deflected toward the foramen ovale by the eustachian valve, and then blood is directed from the right atrium to the left atrium via the foramen ovale. This fetal interatrial communication (the PFO) normally closes after birth as a result of fusion of the septum primum and septum secundum. However, it may persist in 25% to 30% of adults where it is probe patent with a competent valve. In some cases, the valve of fossa ovalis is incompetent, either congenitally or acquired due to elevated right or left atrial pressures allowing interatrial shunting across the foramen.
The atrial septal anatomy and location of various types of ASDs are shown in Figure 28.2. Secundum ASDs occur in the central part
of the atrial septum (fossa ovalis) as a result of deficient valve tissue, ectopic or excessive resorption of septum primum, or deficient growth of septum secundum (Fig. 28.3). Such defects result in an enlarged ostium secundum. These defects usually are single, but rarely can occur as multiple atrial septal fenestrations.
of the atrial septum (fossa ovalis) as a result of deficient valve tissue, ectopic or excessive resorption of septum primum, or deficient growth of septum secundum (Fig. 28.3). Such defects result in an enlarged ostium secundum. These defects usually are single, but rarely can occur as multiple atrial septal fenestrations.
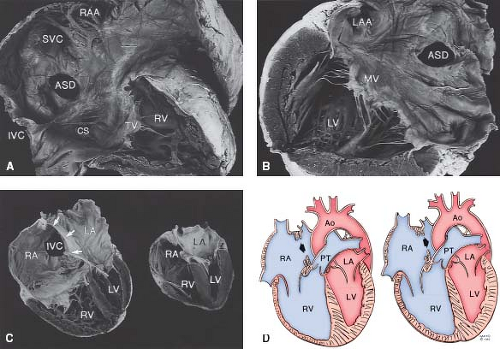
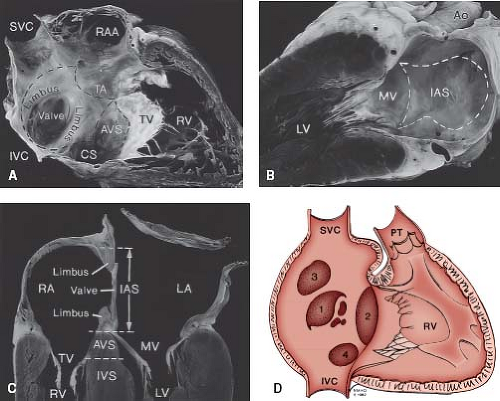 Figure 28.2 A: Anatomy of the atrial septum. View from the right atrial side. B: View from the left atrial side. The interatrial septum (IAS) is outlined by the dotted lines. It is formed by the limbus of the fossa ovalis on the right atrial side and the valve of the fossa ovalis toward the left. The aortic root indents the right atrial free wall anterosuperiorly as the torus aorticus (TA). C: Cross section of the heart in a four-chamber view. The IAS formed by the limbus and valve of fossa ovalis lies between the right and left atria (RA, LA). The atrioventricular septum (AVS) lies between the RA and left ventricle (LV). D: Schematic diagram showing the location of various types of ASDs: (1) secundum; (2) primum; (3) sinus venosus; (4) coronary sinus. IVC, inferior vena cava; IVS, interventricular septum; MV, mitral valve; PT, pulmonary trunk; RAA, right atrial appendage; RV, right ventricle; SVC, superior vena cava; TV, tricuspid valve. (Reprinted by permission of the Mayo Foundation.) |
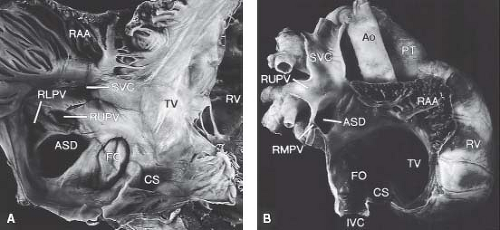
Sinus venosus defects occur outside the margins of the fossa ovalis, in relation to the venous connections of the right atrium (Fig. 28.4). The right horn of sinus venosus incorporates the right superior vena cava (SVC) and inferior vena cava (IVC) into the right atrium. Ectopic or incomplete resorption of the sinus venosus
results in deficiency of the wall that separates the right pulmonary veins from the SVC, IVC, and the right atrium resulting in a sinus venosus ASD (21). Some argue that these should be termed partial anomalous pulmonary venous return to the SVC. The interatrial communication in these defects is, in fact, the orifice of the unroofed right pulmonary vein and is not a true defect in the atrial septum per se (21). Most commonly, the sinus venosus ASDs are related to the SVC where blood from the right upper and/or middle pulmonary veins is directed into SVC or the right atrium. Less frequently, a similar defect can occur inferior to the fossa ovalis in relation to the IVC and the right lower pulmonary venous orifice. This defect often has been termed an IVC-type sinus venosus ASD, although direct involvement of the IVC almost never occurs. Hence, the term sinus venosus defect of right atrial type is preferred (21).
results in deficiency of the wall that separates the right pulmonary veins from the SVC, IVC, and the right atrium resulting in a sinus venosus ASD (21). Some argue that these should be termed partial anomalous pulmonary venous return to the SVC. The interatrial communication in these defects is, in fact, the orifice of the unroofed right pulmonary vein and is not a true defect in the atrial septum per se (21). Most commonly, the sinus venosus ASDs are related to the SVC where blood from the right upper and/or middle pulmonary veins is directed into SVC or the right atrium. Less frequently, a similar defect can occur inferior to the fossa ovalis in relation to the IVC and the right lower pulmonary venous orifice. This defect often has been termed an IVC-type sinus venosus ASD, although direct involvement of the IVC almost never occurs. Hence, the term sinus venosus defect of right atrial type is preferred (21).
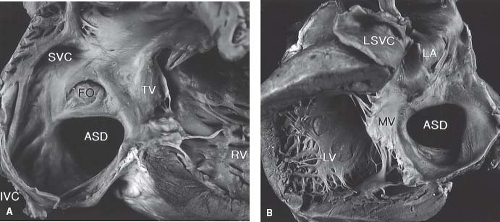
The left horn of the sinus venosus forms the CS. The CS defect (unroofed CS) results from failure of the wall between the left atrium and CS to develop. There may be complete or partial unroofing of the CS resulting in direct communication with the left atrium (Fig. 28.5). Almost always, this anomaly is associated with a left SVC. A rare variation consists of complete absence of the CS rather than unroofing, along with a defect in the expected location of the ostium of CS. In such defects, blood from the left SVC directly enters the left atrium. Kirklin and Barratt-Boyes (5) classified the unroofed CS defects as type I, completely unroofed with LSVC; type II, completely unroofed without LSVC; type III, partially unroofed midportion; and type IV, partially unroofed terminal portion.
Associated Cardiovascular Anomalies
Interatrial communications can occur in isolation, but often are associated with other congenital heart defects. The presence of an interatrial communication may be crucial for survival in some such defects such as hypoplastic left heart syndrome, D-transposition of great arteries, tricuspid atresia, and total anomalous pulmonary venous return. Partial anomalous pulmonary venous return is present in almost 90% of patients with sinus venosus ASDs and, more rarely, can occur with secundum ASDs. Although valvular pulmonary stenosis frequently has been associated with ASDs, the increased gradient across the pulmonary valve may be flow related and not necessarily due to an abnormal valve per se. CS ASDs commonly are associated with a persistent left SVC. Aside from congenital heart defects, secundum ASDs also have been reported in association with noncompaction and apical hypertrophic cardiomyopathy (14,15).
Lutembacher Syndrome
Lutembacher syndrome is the association of an ASD with mitral stenosis. Unlike the original description by Lutembacher, where the mitral stenosis was considered to be congenital in origin, the current consensus is that it is of rheumatic origin (22). As a result of the mitral stenosis, the left-to-right shunting across the ASD is augmented. The ASD usually is large and unrestrictive. Therefore, the magnitude of the atrial level shunt is directly related to the degree of obstruction at the mitral valve. Rarely, when the ASD is restrictive, the left-to-right shunt is continuous due to significant pressure difference across the atrial septum during the entire cardiac cycle.
Pathophysiology
During fetal life, the majority of the blood reaching the left atrium comes via the foramen ovale since there is minimal flow to the lungs. After birth, the lungs expand and the pulmonary blood flow increases. The increased pulmonary venous return to the left atrium results in the left atrial pressure exceeding the right atrial pressure causing functional closure of the foramen ovale. Intrauterine physiology is not altered in the presence of an ASD, but unlike the PFO, the ASD does not close with the hemodynamic changes that occur following birth. The physiologic consequences of an ASD depend on the magnitude and duration of the shunt and its interaction with the pulmonary vascular bed. The primary determinant of the magnitude and direction of the shunt is the relative compliance of the
ventricles. During the neonatal transition period as the pulmonary vascular resistance drops and the right ventricular wall becomes thinner and hence more compliant than that of the left ventricle, there is an increase in left-to-right shunting. Catheter-based studies on flow dynamics in ASD provide insight into the circulatory pattern during the various phases of cardiac cycle. Maximum left-to-right shunting occurs during diastole when all four cardiac chambers are in communication. Atrial contraction further augments this shunt. A small right-to-left shunt, predominantly from IVC blood can occur during early diastole or during onset of systole. The magnitude and amount of shunting varies with the respiratory cycle. During inspiration when the intrathoracic pressure is decreased, there is a decrease in the left-to-right shunt across the ASD. Conversely, during expiration, when the intrathoracic pressure is increased, there is an increase in the left-to-right shunt.
ventricles. During the neonatal transition period as the pulmonary vascular resistance drops and the right ventricular wall becomes thinner and hence more compliant than that of the left ventricle, there is an increase in left-to-right shunting. Catheter-based studies on flow dynamics in ASD provide insight into the circulatory pattern during the various phases of cardiac cycle. Maximum left-to-right shunting occurs during diastole when all four cardiac chambers are in communication. Atrial contraction further augments this shunt. A small right-to-left shunt, predominantly from IVC blood can occur during early diastole or during onset of systole. The magnitude and amount of shunting varies with the respiratory cycle. During inspiration when the intrathoracic pressure is decreased, there is a decrease in the left-to-right shunt across the ASD. Conversely, during expiration, when the intrathoracic pressure is increased, there is an increase in the left-to-right shunt.
Moderate-to-large left-to-right shunts across an ASD result in volume overload and dilation of the right atrium and ventricle (see Fig. 28.3). The tricuspid and pulmonary annuli may dilate and become incompetent. The volume-overloaded right ventricle alters the diastolic configuration of the left ventricle with septal bowing toward the left. Occasionally, the abnormal left ventricular geometry may result in prolapse of the mitral valve or superior systolic motion of the mitral leaflet (23). As a result of the increased flow into the lungs, the pulmonary arteries, capillaries, and the veins are dilated and there can be flow-related pulmonary artery hypertension. Over time this can lead to medial hypertrophy of pulmonary arteries and muscularization of the arterioles resulting in pulmonary vascular obstructive disease (24,25). With severe pulmonary vascular obstructive disease, patients develop Eisenmenger syndrome as the atrial level shunt becomes right-to-left, resulting in cyanosis (see Fig. 28.3).
Clinical Presentation
History
Most patients with ASD are asymptomatic and may remain undiagnosed until later in life. They may come to medical attention due to abnormal auscultatory findings or diagnostic studies such as ECG, chest radiograph, or echocardiogram. Very rarely, some infants with ASD may present with features of pulmonary overcirculation, recurrent respiratory infections, and failure to thrive. The mechanism of heart failure in these infants is not well understood since the hemodynamics are quite similar to those who are asymptomatic. Some have proposed rapid remodeling and thinning of the pulmonary vascular bed to be the reason for this early presentation (26). Additionally, one should carefully evaluate mitral anatomy and function since mitral stenosis or regurgitation can augment atrial shunting. Despite repair of ASD in these patients, there may not be any significant improvement in their symptoms (27). Older children may present with symptoms of mild fatigue and dyspnea that may worsen with age. In adults, worsening of clinical condition has been attributed to various factors such as a decrease in left ventricular compliance secondary to coronary artery disease and hypertension, which, in turn, results in increased left-to-right shunt, right ventricular failure, atrial arrhythmias, and elevated pulmonary artery pressure. Patients who develop Eisenmenger syndrome (reversal of the left-to-right shunt due to pulmonary hypertension) may present with cyanosis and syncope with exertion.
Physical Examination
Phenotypic features of various syndromes associated with ASD may be notable. The most common syndrome known to be associated with ostium secundum ASD is the Holt–Oram syndrome. In this syndrome, the thumb is hypoplastic and in some cases may be rudimentary or absent. Additionally, the metacarpals may be small or absent and the radius may be absent. The thumb may resemble a finger.
In patients with long-standing large left-to-right shunt, there is a left precordial bulge. Palpation reveals a prominent right ventricular impulse felt along the lower left sternal border and the subcostal area. In normal persons, splitting of S2 has a normal variation with respiration. During inspiration, negative intrathoracic pressure causes increased venous return into the right side of the heart, which in turn causes the pulmonary valve to stay open for a longer duration in ventricular systole causing a normal delay in the pulmonary valve closure component of S2. During expiration, the positive intrathoracic pressure reduces the venous return to the right side of the heart, resulting in an earlier closure of pulmonary valve. The auscultatory hallmark of ASD is wide, fixed splitting of the second heart sound (S2). This means that the aortic and pulmonary components of S2 are widely separated during expiration and demonstrate little or no variation in degree of splitting during inspiration or with Valsalva maneuver. S2 is “widely split” due to a delay in closure of the pulmonary valve resulting from prolonged emptying of the volume-overloaded right ventricle and increased pulmonary vascular capacitance leading to low pulmonary impedance and, therefore, a long “hangout interval” after the end of right ventricular systole. The S2 is “fixed” since the increased right ventricular stroke volume does not vary much with respiration.
A systolic ejection murmur can be heard in the left upper parasternal area. The murmur is due to increased flow across the pulmonary valve. This murmur begins shortly after S1 and is crescendo–decrescendo, reaching its peak in early to midsystole and ending before S2. When this murmur is loud, it can indicate a large shunt or associated pulmonary valve stenosis (a systolic ejection click usually is present when the pulmonary valve is truly stenotic). When there is a large left-to-right shunt, a mid-diastolic murmur can be heard due to excessive flow across the tricuspid valve. This murmur is short, soft, low to medium in frequency, and localized to the left lower parasternal area. Rarely, a diastolic murmur may result from pulmonary regurgitation as a result of an exceptionally large pulmonary trunk that dilates the valve annulus.
Most patients are acyanotic. However, cyanosis can be seen in those with pulmonary hypertension, significant right ventricular outflow tract obstruction, or in rare cases of a large eustachian valve directing IVC blood into the left atrium via the ASD. Patients with pulmonary hypertension and a right-to-left atrial level shunt are cyanotic and have auscultatory findings that are different from those of patients with an ASD without pulmonary hypertension. The jugular venous pulse has a dominant “A” wave resulting from increased force of right atrial contraction. The large “A” waves result in presystolic distention of the right ventricle resulting in a fourth heart sound. The pulmonary component of S2 is loud and prominent. The wide fixed splitting of S2 and tricuspid flow murmur disappear and the midsystolic pulmonary flow murmur is replaced by a softer and shorter murmur. A high-frequency early diastolic murmur (Graham Steell murmur) of pulmonary regurgitation can be heard due to pulmonary valve incompetence resulting from pulmonary hypertension. Also, a holosystolic S1 coincident murmur of tricuspid regurgitation heard best at the right lower sternal border can develop.
Diagnostic Evaluation
Electrocardiogram and Electrophysiology
ECG findings depend on the type and size of the ASD. In patients with a small left-to-right shunt and no right atrial or ventricular
dilation, the ECG is normal. With a significant left-to-right shunt, an rSR’ pattern occurs in the right precordial leads indicating right ventricular volume overload (Fig. 28.6). There often is slight prolongation of the QRS complex with slurring of the terminal R’, and the terminal forces are directed to the right, superiorly and anteriorly. Other features include right axis deviation and tall P waves reflecting right atrial enlargement. In almost 50% of patients with sinus venosus ASD, a frontal plane P wave axis of <30 degrees is seen (4). In most patients with ASD, the frontal plane QRS axis is between +95 and +170 degrees. Ostium primum ASD can be distinguished from other forms of ASDs by the presence of a counterclockwise loop and left axis deviation. Rhythm usually is sinus in children. However, older patients, usually beyond the third decade of life, can have junctional rhythm or atrial arrhythmias such as atrial fibrillation or flutter (28). With the advent of pulmonary hypertension, the rSR’ pattern in the right precordial leads is replaced by Q waves and tall monophasic R waves with deeply inverted T waves.
dilation, the ECG is normal. With a significant left-to-right shunt, an rSR’ pattern occurs in the right precordial leads indicating right ventricular volume overload (Fig. 28.6). There often is slight prolongation of the QRS complex with slurring of the terminal R’, and the terminal forces are directed to the right, superiorly and anteriorly. Other features include right axis deviation and tall P waves reflecting right atrial enlargement. In almost 50% of patients with sinus venosus ASD, a frontal plane P wave axis of <30 degrees is seen (4). In most patients with ASD, the frontal plane QRS axis is between +95 and +170 degrees. Ostium primum ASD can be distinguished from other forms of ASDs by the presence of a counterclockwise loop and left axis deviation. Rhythm usually is sinus in children. However, older patients, usually beyond the third decade of life, can have junctional rhythm or atrial arrhythmias such as atrial fibrillation or flutter (28). With the advent of pulmonary hypertension, the rSR’ pattern in the right precordial leads is replaced by Q waves and tall monophasic R waves with deeply inverted T waves.
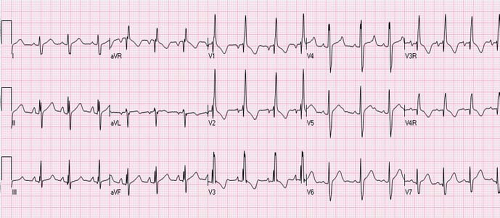 Figure 28.6 ECG in a 4-year-old patient with large secundum ASD showing rSR’ pattern in lead V1 and V2 and terminal widening on S in lead V6 indicating right ventricular volume overload. |
Electrophysiologic studies have demonstrated a significant age-related incidence of sinus node dysfunction that may begin in early childhood (29,30). These patients have prolonged corrected sinus node recovery times and sinoatrial conduction times (31). However, these findings are not significant clinically and the resting and ambulatory ECGs in these patients are normal. AV node dysfunction is less common than sinus node dysfunction. Electrophysiologic studies in such cases show a prolonged A-H interval or an AV node Wenckebach periodicity at slow atrial pacing rates. First-degree AV block can occur in older individuals as a result of intraatrial H-V conduction delay and in patients with a rare autosomal dominant form of secundum ASDs (16).
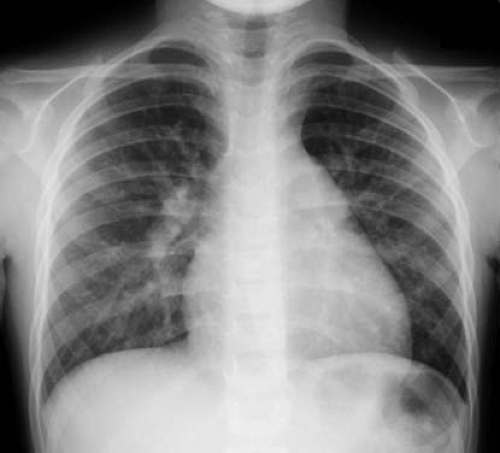
Chest X-Ray
A small shunt across the ASD generally results in a normal-appearing chest x-ray. Cardiomegaly, due to right atrial and right ventricular enlargement, and increased pulmonary vascular markings extending to the periphery are seen in patients with significant shunts. (Fig. 28.7). The dilated right ventricle occupies the apex and forms an acute angulation with the left hemidiaphragm in the anteroposterior projection and obliterates the retrosternal space in the lateral view. Dilation of the right ventricular outflow tract may cause smooth continuity with the enlarged main pulmonary artery. The proximal branch pulmonary arteries, especially the right pulmonary artery, also are dilated. The left atrial and left ventricular sizes are normal. If pulmonary hypertension develops, the increased peripheral pulmonary arterial vascularity is replaced by oligemic lung fields.
Echocardiogram
The echocardiogram is instrumental in defining the type of ASD, its size, the degree of shunting, its effect on the right-sided chambers of the heart, associated lesions, and estimations of right ventricular pressure. These echocardiographic findings help in determining the appropriate intervention.
Transthoracic Echocardiography
Two-Dimensional Imaging and M-Mode
An ASD can be visualized from the subcostal, parasternal, and apical views. Subcostal views provide the best profile of the atrial septum since the ultrasound beam is perpendicular to it. However, subcostal windows may be suboptimal in older or obese patients. In apical views, a “drop-out” may be seen in the thin septal region of the fossa ovalis since the ultrasound beam is parallel to it. This can give a false appearance of an ASD.
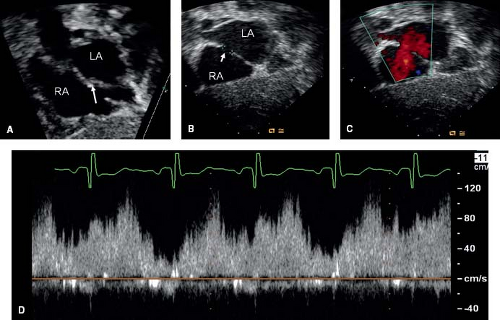
The type of ASD can be determined by defining its location. PFO and secundum ASDs are located in the region of fossa ovalis, that is, the midatrial septal region (Fig. 28.8). A PFO is guarded by a flap valve on the left side and limbus of fossa ovalis to the right. A PFO can be differentiated from a secundum ASD by this overlap of septal tissue. Ostium primum ASDs are located between the anteroinferior margin of the fossa ovalis and AV valves. Sinus venosus defects are seen superiorly at the junction of SVC with the right atrium
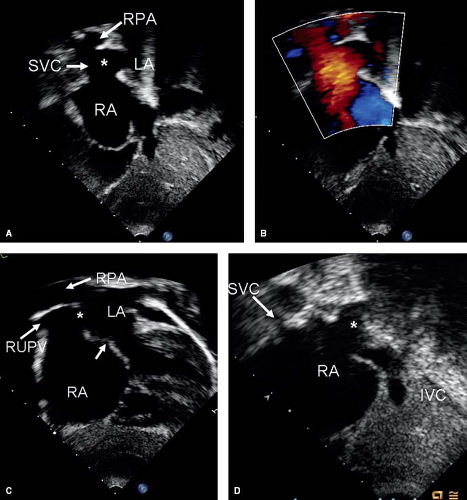
(Fig. 28.9). These defects are best seen from the subcostal short-axis or high right parasternal views as a communication between the atria where the right upper pulmonary vein and SVC are usually seen. The right pulmonary artery is seen in cross section immediately above the ASD. Subcostal long-axis and parasternal short-axis views are useful in evaluating sinus venosus ASDs of the right atrial type which appear as a posteroinferior atrial defect with a deficient posterior margin (Fig. 28.9). Anomalous drainage of right middle and lower pulmonary vein can be seen best in the parasternal short-axis view. For CS defects, a large ostium of the CS is seen as an inferior
interatrial communication, just above and anterior to the entry of IVC into the right atrium. Absence of the CS indicates complete unroofing. In partial unroofing, parts of the wall between the CS and the left atrium can be identified.
(Fig. 28.9). These defects are best seen from the subcostal short-axis or high right parasternal views as a communication between the atria where the right upper pulmonary vein and SVC are usually seen. The right pulmonary artery is seen in cross section immediately above the ASD. Subcostal long-axis and parasternal short-axis views are useful in evaluating sinus venosus ASDs of the right atrial type which appear as a posteroinferior atrial defect with a deficient posterior margin (Fig. 28.9). Anomalous drainage of right middle and lower pulmonary vein can be seen best in the parasternal short-axis view. For CS defects, a large ostium of the CS is seen as an inferior
interatrial communication, just above and anterior to the entry of IVC into the right atrium. Absence of the CS indicates complete unroofing. In partial unroofing, parts of the wall between the CS and the left atrium can be identified.
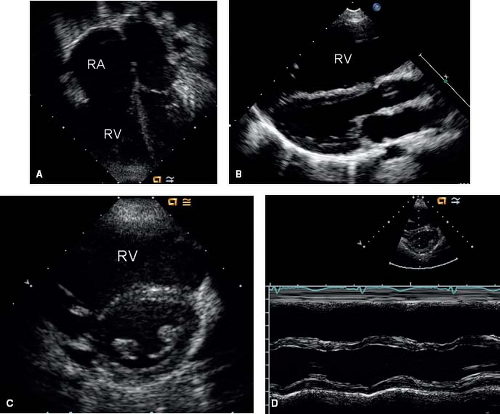
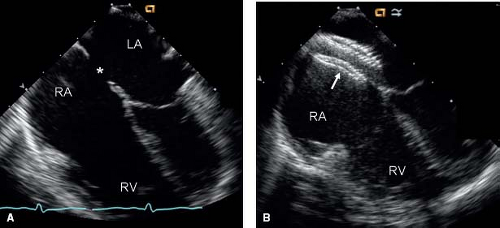
The enlarged right atrium, right ventricle, and pulmonary arteries also are seen on 2-D imaging. The volume-overloaded right ventricle causes diastolic flattening and paradoxical motion of the interventricular septum (Fig. 28.10). Associated anomalies such as pulmonary stenosis, mitral valve prolapse, and anomalous pulmonary venous return also should be evaluated using 2-D imaging. M-mode imaging of the ventricles will show an enlarged right ventricle and paradoxical septal motion (Fig. 28.10).
Doppler
Using color Doppler, one can visualize the shunt across the ASD (see Fig. 28.8). This shunt usually is left-to-right, but in patients with elevated pulmonary artery pressure, a bidirectional shunt or a right-to-left shunt can be seen. Pulsed-wave Doppler shows interatrial shunting in late systole and early diastole. Since the pressure gradient across the atrial septum is minimal when these defects are nonrestrictive, low-velocity flow is noted using Doppler. Commonly, a qualitative assessment of the shunt across the ASD is done by direct visualization of the shunt using color Doppler and its effect on the right-sided cardiac chambers. However, a quantitative assessment of the pulmonary to systemic blood flow ratio (Qp:Qs) also can be made. For this, the time velocity integrals obtained by tracing the pulsed-wave Doppler of pulmonary and aortic outflow are multiplied by the area of pulmonary and aortic valve, respectively. This has been shown to have a close correlation with the Qp:Qs measured invasively by oximetry during cardiac catheterization (32). Presence of right ventricular outflow tract obstruction, semilunar valve insufficiency, and patent ductus arteriosus limit the use of this method (33).
A large left-to-right shunt may result in a flow-related peak gradient of as much as 30 mm Hg across the pulmonary valve. However, with higher gradients, one must suspect associated pulmonary valvular stenosis. Progressive tricuspid regurgitation resulting from tricuspid annular dilation and lack of coaptation of leaflets can be seen with significant right ventricular dilation. Doppler assessment for estimating pulmonary artery pressure can be performed by measuring the tricuspid and pulmonary regurgitant jets and applying the modified Bernoulli equation to calculate transvalve gradients and adding estimated right atrial pressure and right ventricular
end-diastolic pressure, respectively. Development of pulmonary hypertension results in worsening of tricuspid and pulmonary regurgitation. In addition, the main and proximal pulmonary arteries further dilate. The right ventricle becomes hypertrophied, and its systolic function starts deteriorating.
end-diastolic pressure, respectively. Development of pulmonary hypertension results in worsening of tricuspid and pulmonary regurgitation. In addition, the main and proximal pulmonary arteries further dilate. The right ventricle becomes hypertrophied, and its systolic function starts deteriorating.
Transesophageal Echocardiography
A transesophageal echocardiogram (TEE) is useful for evaluation of ASD when transthoracic images are inconclusive due to poor windows or unusual location of the defect. TEE can be particularly useful to detect a sinus venosus ASD, which easily can be missed using transthoracic imaging. In addition, it frequently is used as a monitoring adjunct for operative and percutaneous closure of ASD. Due to the close proximity of the transducer to the cardiac structures, transesophageal imaging allows better spatial resolution and superior images of the atrial septum compared with transthoracic imaging (Fig. 28.11).
Contrast Echocardiography
Intravenous contrast using injection of agitated saline during transthoracic or transesophageal echocardiographic imaging can be used to confirm the shunting across an ASD or a PFO. A left-to-right shunt is seen as a negative contrast washout into the right atrium, when the right atrium is opacified with contrast. A right-to-left shunt is detected by the presence of microbubbles in the left atrium and ventricle, and this effect can be augmented by performing a simultaneous Valsalva maneuver. In the presence of an unroofed CS, injection of contrast in the left arm will result in microbubbles in the left atrium before it opacifies the right atrium.
Three-Dimensional Echocardiography
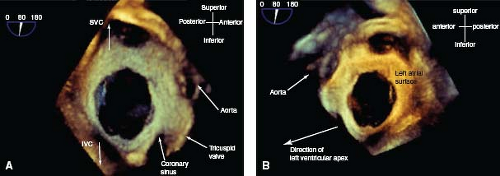
Imaging the ASD using 2-D echocardiography is based on limited number of orthogonal planes and could result in underestimation of the size of the ASD and its surrounding rims. With the availability of real-time 3-D echocardiography, an en face view of the entire atrial septum can be obtained, thus allowing better morphologic delineation of the ASD and its surrounding structures (34). This information is helpful in determining whether or not closure of the ASD can be accomplished with a transcatheter device (35). Furthermore, 3-D transesophageal echo now is being used not only to delineate anatomic features of ASD but also for guiding device closure during the procedure (Fig. 28.12) (36,37,38).
Intracardiac Echocardiography
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree