Marc S. Sabatine, Christopher P. Cannon
Approach to the Patient with Chest Pain
Acute chest pain is one of the most common reasons for seeking care in the emergency department (ED), and it accounts for approximately 8 million ED visits annually in the United States. Such pain suggests acute coronary syndrome (ACS), but after diagnostic evaluation, only 15% to 25% of patients with acute chest pain actually have ACS.1,2 The difficulty lies in discriminating patients with ACS or other life-threatening conditions from those with noncardiovascular, non–life-threatening chest pain. The diagnosis of ACS is missed in approximately 2% of patients, which can lead to substantial consequences—for example, the short-term mortality in patients with acute myocardial infarction (MI) who are mistakenly discharged from the ED increases twofold over that expected for patients who are admitted to the hospital. For patients with a lower risk for complications, however, these concerns must be balanced against the cost and inconvenience of admission and against the risk for complications from tests and procedures with a low probability of improving patient outcomes.
Several recent advances have enhanced the accuracy and efficiency of evaluation of patients with acute chest pain, including better blood markers for myocardial injury3; decision aids to stratify patients according to their risk for complications; early exercise testing4; radionuclide scanning for lower risk patient subsets5 (see Chapter 16); multislice computed tomography for anatomic evaluation of coronary artery disease (CAD), pulmonary embolism (PE), and aortic dissection6 (see Chapter 18); and the use of chest pain units4 and critical pathways for efficient and rapid evaluation of lower-risk patients.7
Causes of Acute Chest Pain
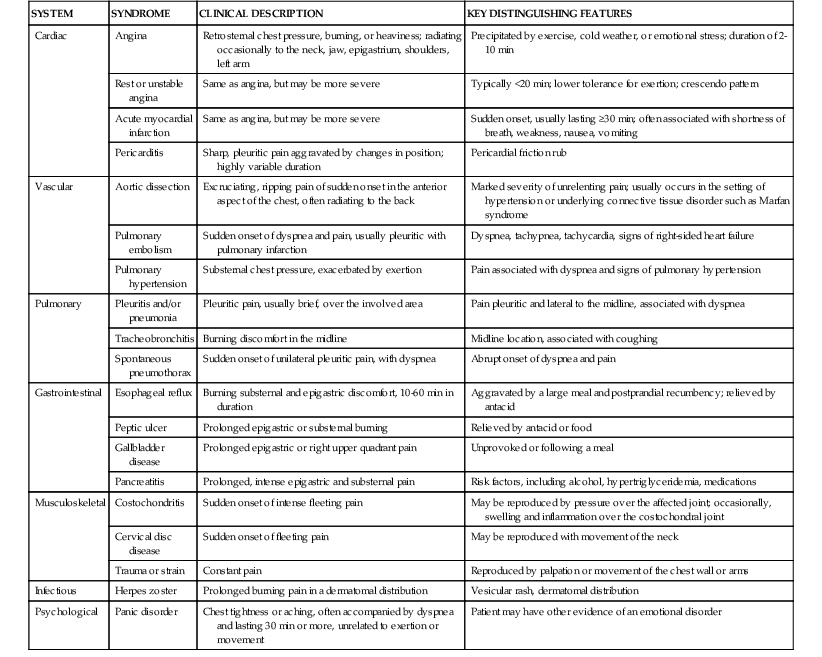
In a typical population of patients undergoing evaluation for acute chest pain in EDs, about 15% to 25% have acute MI or unstable angina.2 A small percentage has other life-threatening problems, such as PE or acute aortic dissection, but most leave the ED without a diagnosis or with a diagnosis of a non–cardiac-related condition. Such noncardiac conditions include musculoskeletal syndromes, disorders of the abdominal viscera (including gastroesophageal reflux disease), and psychological conditions (Table 50-1).
Myocardial Ischemia or Infarction
The most common serious cause of acute chest discomfort is myocardial ischemia or infarction (see Chapters 52 and 53), which occurs when the supply of myocardial oxygen is inadequate for the demand. Myocardial ischemia usually occurs in the setting of coronary atherosclerosis, but it may also reflect dynamic components of coronary vascular resistance. Coronary spasm can occur in normal coronary arteries or, in patients with coronary disease, near atherosclerotic plaque and in smaller coronary arteries (see Chapter 49). Other less common causes of impaired coronary blood flow include syndromes that compromise the orifices or lumina of the coronary arteries, such as coronary arteritis, proximal aortitis, spontaneous coronary dissection, proximal aortic dissection, coronary emboli from infectious or noninfectious endocarditis or thrombus in the left atrium or left ventricle, myocardial bridge, or a congenital abnormality of the coronary arteries (see Chapter 20).
The classic manifestation of ischemia is angina, which is usually described as a heavy chest pressure or squeezing, a burning feeling, or difficulty breathing (see Chapter 11). The discomfort often radiates to the left shoulder, neck, or arm. It typically builds in intensity over a period of a few minutes. The pain may begin with exercise or psychological stress, but ACS most commonly occurs without obvious precipitating factors.
Atypical descriptions of chest pain reduce the likelihood that the symptoms represent myocardial ischemia or injury. The American College of Cardiology (ACC) and American Heart Association (AHA) guidelines list the following as pain descriptions uncharacteristic of myocardial ischemia7:
• Pleuritic pain (i.e., sharp or knifelike pain brought on by respiratory movements or coughing)
• Primary or sole location of the discomfort in the middle or lower abdominal region
• Pain that may be localized by the tip of one finger, particularly over the left ventricular apex
• Pain reproduced with movement or palpation of the chest wall or arms
• Constant pain that persists for many hours
• Very brief episodes of pain that last a few seconds or less
Nevertheless, data from large populations of patients with acute chest pain indicate that ACS occurs in those with atypical symptoms at sufficient frequency that no single factor suffices to exclude the diagnosis of acute ischemic heart disease. Clinicians should be mindful of “angina equivalents” such as jaw or shoulder pain in the absence of chest pain or dyspnea, nausea or vomiting, and diaphoresis. In particular, women, older persons, and individuals with diabetes may be more likely to report atypical symptoms of myocardial ischemia or infarction (see Chapter 77). Data from the National Registry of Myocardial Infarction demonstrate that among patients hospitalized with MI, women—particularly young women—are significantly less likely than men to manifest chest pain. Not surprisingly, patients without chest pain had higher in-hospital mortality.8
Pericardial Disease
The visceral surface of the pericardium is insensitive to pain, as is most of the parietal surface. Therefore, noninfectious causes of pericarditis (e.g., uremia; see Chapter 71) usually cause little or no pain. In contrast, infectious pericarditis almost always involves the surrounding pleura, so patients typically experience pleuritic pain with breathing, coughing, and changes in position. Swallowing may induce the pain because of the proximity of the esophagus to the posterior portion of the heart. Because the central diaphragm receives its sensory supply from the phrenic nerve and the phrenic nerve arises from the third to fifth cervical segments of the spinal cord, pain from infectious pericarditis is frequently felt in the shoulders and neck. Involvement of the diaphragm more laterally can lead to symptoms in the upper part of the abdomen and back, and thus create confusion with pancreatitis or cholecystitis. Pericarditis occasionally causes a steady, crushing substernal pain resembling that of acute MI.9
Vascular Disease
Acute aortic dissection (see Chapter 57) usually causes a sudden onset of excruciating ripping pain, the location of which reflects the site and progression of the dissection. Ascending aortic dissection tends to be manifested as pain in the midline of the anterior aspect of the chest, and posterior descending aortic dissection tends to cause pain in the back of the chest. Aortic dissections are rare, with an estimated annual incidence of 3 per 100,000, and usually occur in the presence of risk factors, including Marfan and Ehlers-Danlos syndromes, bicuspid aortic valve, pregnancy (for proximal dissections), and hypertension (for distal dissections).
Pulmonary emboli (see Chapter 73) often cause a sudden onset of dyspnea and pleuritic chest pain, although they may be asymptomatic. The annual incidence is approximately 1 per 1000, although this number is probably an underestimate. Massive pulmonary emboli tend to cause severe and persistent substernal pain, which is attributed to distention of the pulmonary artery. Smaller emboli that lead to pulmonary infarction can cause lateral pleuritic chest pain. Hemodynamically significant pulmonary emboli may cause hypotension, syncope, and signs of right-sided heart failure. Pulmonary hypertension (see Chapter 74) can result in chest pain similar to that of angina pectoris, presumably because of right-heart hypertrophy and ischemia.
Pulmonary Conditions
Pulmonary conditions that cause chest pain generally produce dyspnea and pleuritic symptoms, the location of which reflects the site of pulmonary disease. Tracheobronchitis tends to be associated with a burning midline pain, whereas pneumonia can produce pain over the involved lung. The pain of pneumothorax begins suddenly and is usually associated with dyspnea. Primary pneumothorax typically occurs in tall, thin young men; secondary pneumothorax occurs in the setting of pulmonary disease such as chronic obstructive pulmonary disease, asthma, or cystic fibrosis. Asthma exacerbations can be accompanied by chest discomfort, typically characterized as tightness.
Gastrointestinal Conditions
Irritation of the esophagus by acid reflux can produce a burning discomfort that may be exacerbated by alcohol, aspirin, and some foods. Symptoms are often worsened by a recumbent position and are relieved by sitting upright and with acid-reducing therapies. Esophageal spasm can produce a squeezing chest discomfort similar to that of angina. Mallory-Weiss tears of the esophagus can occur in patients who have had prolonged vomiting episodes. Severe vomiting can also result in esophageal rupture (Boerhaave syndrome) with mediastinitis. Chest pain caused by peptic ulcer disease usually occurs 60 to 90 minutes after meals and typically responds rapidly to acid-reducing therapies. This pain is generally epigastric in location but can radiate to the chest and shoulders. Cholecystitis produces a wide range of pain syndromes and generally causes right upper quadrant abdominal pain, but chest and back pain caused by this disorder is not unusual. The pain is frequently described as being aching or colicky. Pancreatitis typically causes an intense, aching epigastric pain that may radiate to the back. Relief through acid-reducing therapies is limited.
Musculoskeletal and Other Causes
Chest pain can arise from musculoskeletal disorders involving the chest wall (such as costochondritis), by conditions affecting the nerves of the chest wall (such as cervical disc disease), by herpes zoster, or following heavy exercise. Chest pain secondary to musculoskeletal causes is often elicited by direct pressure over the affected area or by movement of the patient’s neck. The pain itself can be fleeting, or it can be a dull ache that lasts for hours. Panic syndrome is a major cause of chest discomfort in ED patients. The symptoms typically include chest tightness, often accompanied by shortness of breath and a sense of anxiety, and generally last 30 minutes or longer.
Diagnostic Considerations
Clinical Evaluation
When evaluating patients with acute chest pain, clinicians must address a series of issues related to prognosis and immediate management.10 Even before trying to arrive at a definite diagnosis, high-priority questions include the following:
Initial Assessment
Evaluation of a patient with acute chest pain can begin before the physician sees the patient, and thus effectiveness may depend on the actions of the office staff and other nonphysician personnel. Guidelines from the ACC and AHA7 (see Chapters 52 and 53, Guidelines sections) emphasize that patients with symptoms consistent with ACS should not be evaluated solely over the telephone but should be referred to facilities that allow evaluation by a physician and recording of a 12-lead electrocardiogram (ECG).11 These guidelines also recommend strong consideration of immediate referral to an ED or a specialized chest pain unit for patients with suspected ACS who experience chest discomfort at rest for longer than 20 minutes, hemodynamic instability, or recent syncope or near-syncope. Transport as a passenger in a private vehicle is considered an acceptable alternative to an emergency vehicle only if the wait would lead to a delay longer than 20 to 30 minutes.
Guidelines11 recommend that patients with the following chief complaints undergo immediate assessment by triage nurses and be referred for further evaluation:
• Indigestion or heartburn; nausea and/or vomiting associated with chest discomfort
• Persistent shortness of breath
• Weakness, dizziness, lightheadedness, loss of consciousness
For such patients, initial assessment involves taking a history, performing a physical examination, obtaining an ECG and chest radiograph, and measuring biomarkers of myocardial injury.
History
If the patient does not need immediate intervention because of impending or actual circulatory collapse or respiratory insufficiency, the physician’s assessment should begin with a clinical history that captures the characteristics of the patient’s pain, including its quality, location, and radiation; the time and tempo (abrupt or gradual) of onset; the duration of symptoms; provoking or palliating activities; and any associated symptoms, particularly those that are pulmonary or gastrointestinal. ACS is typically described as a diffuse substernal chest pressure that starts gradually, radiates to the jaw or arms, worsens with exertion, and is relieved by rest or nitroglycerin. Because angina tends to be manifested in the same way in a given patient (at least if it is due to ischemia in the same territory), it is useful to compare the current episode with any previous documented episodes of angina. Studies have suggested that response to nitroglycerin may not reliably discriminate cardiac chest pain from non–cardiac-related chest pain.12 In contrast to the tempo of the chest pain in ACS, PE, aortic dissection, and pneumothorax are all characterized by chest pain that is sudden and severe in onset. Moreover, pain that is pleuritic or positional in nature suggests PE, pericarditis, pneumonia, or a musculoskeletal condition. A review of the literature yielded eight factors from the chest pain history with a likelihood ratio for ACS significantly greater than 1 and six factors with a likelihood ratio significantly lower than 1 (Table 50-2).13
TABLE 50-2
Value of Elements of the Chest Pain History for the Diagnosis of Acute Coronary Syndrome
| PAIN DESCRIPTOR | POSITIVE LIKELIHOOD RATIO (95% CI) |
| Increased Likelihood of AMI | |
| Radiation to the right arm or shoulder | 4.7 (1.9-12.0) |
| Radiation to both arms or shoulders | 4.1 (2.5-6.5) |
| Associated with exertion | 2.4 (1.5-3.8) |
| Radiation to the left arm | 2.3 (1.7-3.1) |
| Associated with diaphoresis | 2.0 (1.9-2.2) |
| Associated with nausea or vomiting | 1.9 (1.7-2.3) |
| Worse than previous angina or similar to previous MI | 1.8 (1.6-2.0) |
| Described as pressure | 1.3 (1.2-1.5) |
| Decreased Likelihood of AMI | |
| Described as pleuritic | 0.2 (0.1-0.3) |
| Described as positional | 0.3 (0.2-0.5) |
| Described as sharp | 0.3 (0.2-0.5) |
| Reproducible with palpation | 0.3 (0.2-0.4) |
| Inframammary location | 0.8 (0.7-0.9) |
| Not associated with exertion | 0.8 (0.6-0.9) |
AMI = acute myocardial infarction; CI = confidence interval.
Modified from Swap CJ, Nagurney JT: Value and limitations of chest pain history in the evaluation of patients with suspected acute coronary syndromes. JAMA 294:2623, 2005.
In addition to the characteristics of the acute episode, the presence of risk factors for atherosclerosis (e.g., advanced age, male sex, diabetes) increases the likelihood that the chest pain is resulting from myocardial ischemia. A history of MI is associated not only with a high risk for obstructive CAD but also with an increased likelihood of multivessel disease. Younger patients have a lower risk for ACS but should be screened with greater care for a history of recent cocaine use (see Chapter 68).14
Physical Examination
The initial examination of patients with acute chest pain should endeavor to identify potential precipitating causes of myocardial ischemia (e.g., uncontrolled hypertension), important comorbid conditions (e.g., chronic obstructive pulmonary disease), and evidence of hemodynamic complications (e.g., congestive heart failure, new mitral regurgitation, hypotension).7 In addition to vital signs, examination of peripheral vessels should include assessment for the presence of bruits or absent pulses, which suggest extracardiac vascular disease (see Chapter 68).
For patients whose clinical findings do not suggest myocardial ischemia, the search for noncoronary causes of chest pain should focus first on potentially life-threatening issues (e.g., aortic dissection, PE) and then turn to the possibility of other cardiac diagnoses (e.g., pericarditis) and noncardiac diagnoses (e.g., esophageal discomfort). Aortic dissection is suggested by blood pressure or pulse disparities or by a new murmur of aortic regurgitation accompanied by back or midline anterior chest pain. A friction rub may accompany pericarditis. Differences in breath sounds in the presence of acute dyspnea and pleuritic chest pain raise the possibility of pneumothorax. Tachycardia, tachypnea, and an accentuated pulmonic component of the second heart sound (P2) may be the major manifestations of PE on physical examination.
Electrocardiography
An ECG, a source of decisive data, should be obtained within 10 minutes after arrival for patients with ongoing chest discomfort and as rapidly as possible for patients who have a history of chest discomfort consistent with ACS but whose discomfort has resolved by the time of evaluation so that patients who might benefit from immediate reperfusion therapy (mechanical or pharmacologic) can be identified (see Chapter 12).11 To that end, obtaining a prehospital ECG decreases the door-to-diagnosis time and, for ST-segment elevation MI (STEMI), the door-to-balloon time. Importantly, these gains accrue without any prolongation of scene or transport times and, in fact, with a reduction in scene and transport times for patients identified to have STEMI.15
The ECG aids in both diagnosis and prognosis. New persistent or transient ST-segment abnormalities (≥0.05 mV) that develop during a symptomatic episode at rest and resolve when the symptoms resolve strongly suggest acute ischemia and severe coronary disease. Nonspecific ST-segment and T wave abnormalities are usually defined as lesser amounts of ST-segment deviation or T wave inversion of 0.2 mV or less and are not as helpful for risk stratification. The likelihood ratios for ACS with various findings on the ECG are shown in Table 50-3.16 Completely normal findings on an ECG do not exclude the possibility of ACS; the risk for acute MI is approximately 4% in patients with a history of CAD and 2% in those with no such history.17 Patients with normal or nearly normal findings on an ECG, however, have a better prognosis than do those with clearly abnormal ECGs at initial evaluation. Moreover, a normal ECG has a negative predictive value of 80% to 90%, regardless of whether the patient was experiencing chest pain at the time that the ECG was obtained.18 Diffuse ST-segment elevation and PR-segment depression suggest pericarditis. Right-axis deviation, right bundle branch block, T wave inversions in leads V1 to V4, and an S wave in lead I and Q wave and T wave inversions in lead III suggest PE.
TABLE 50-3
Value of ECG Findings for the Diagnosis of Acute Coronary Syndrome
| ECG FINDING | POSITIVE LIKELIHOOD RATIO (95% CI WHERE AVAILABLE) |
| New ST-segment elevation ≥1 mm | 5.7-53.9 |
| New Q wave | 5.3-24.8 |
| Any ST-segment elevation | 11.2 (7.1-17.8) |
| New conduction defect |